Abstract
A systematic review of the literature and its critical appraisal shows that there is ample evidence for benefits gained by dedicated arm rehabilitation therapy in terms of improvement of active motor control (impairment) and activities among stroke victims. Self-perceived usefulness of the affected arm as well as disability has been shown to improve with some interventions. Overall, the immediate benefits from various interventions are moderate and long-term effects were less systematically assessed. If applied appropriately, the interventions show little risk to harm and are by and large very acceptable to stroke survivors. The question of feasibility to deliver evidence-based arm rehabilitation therapy rests largely on access to and the availability of skilled human therapeutic resources. The use of technical devices requires additional resources and therapeutic skills.
This chapter gives insight into the current evidence for therapy to improve (sensori)motor control and function of the affected arm and hand. Early after stroke, recovery of function is often the main goal, while in the chronic stage therapeutic goals more often focus on activities. Further, therapy, and therefore choice of interventions, for patients with severe arm paresis is quite different from those with mild-to-moderate arm paresis. Hence, separate evidence-based recommendations are provided for the acute and subacute and the chronic phase and for patients with severe or mild-to-moderate paresis.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Stroke is the leading cause of long-term disability among adults. Even with appropriate acute care and neurorehabilitation, recovery of motor function after stroke is usually incomplete (Ward and Cohen 2004). More than 60% of stroke survivors suffer from persistent neurological deficits with impaired motor function compromising their independence with activities of daily living (Feigin et al. 2003; Levin et al. 2009). Motor function of the affected arm can explain up to 50% of the variance in functional autonomy in stroke patients (Mercier et al. 2001). Further, both arm impairment (i.e. the ability to move the arm and its segments selectively) and arm activities (i.e. the ability to handle objects successfully) as assessed at discharge from rehabilitation are associated with the level of activity and participation 6 months later, that is, the degree of difficulty and help needed in daily life and instrumental activities, and in social roles (Desrosiers et al. 2003).
Thus, arm paresis after stroke and its therapeutic management is a key element of stroke rehabilitation. Treatment of arm paresis has been ranked among the top ten research priorities relating to life after stroke by stroke survivors, caregivers, and health professionals (Pollock et al. 2014a). A wide variety of therapeutic interventions exist that aim to improve arm impairment, activity limitations, and the amount of actual use of the affected arm in daily life (Pollock et al. 2014b). Therapeutic decisions need to be individualized based on factors such as individual therapeutic goals, time post-stroke, and severity of paresis. Indeed, arm paresis after stroke shows a bimodal distribution: there are many people with either mild or severe arm activity limitations, but fewer with moderate activity limitations (Nakayama et al. 1994). And the course and expected level of recovery are different for patients with more or less profound arm paresis initially after stroke (Kwah and Herbert 2016). Therefore, subgroups with different severity of arm paresis do have different therapeutic needs.
Based on a systematic search of the best evidence available, i.e. randomized controlled trials (RCTs) and systematic reviews (SRs), with or without meta-analysis, and their critical appraisal, a best evidence synthesis was performed for treatment of arm paresis post-stroke, and clinical practice recommendations are given based on this synthesis.
2 Methods for the Best Evidence Synthesis
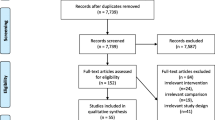
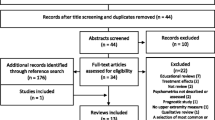
Within the framework of stroke rehabilitation guideline development of the German neurorehabilitation society (DGNR) (Platz 2017), systematic PubMed searches for RCTs and SRs addressing rehabilitative therapy for arm paresis after stroke had been performed in 2006, 2013, 2016, and on 23.07.2017 (PubMed and Cochrane Library). Search terms had been (((Cerebrovascular Accident OR Stroke OR cerebrovascular disorders)) AND (Upper Extremity OR arm)) AND (Rehabilitation OR Physical Therapy Modalities OR Biofeedback OR Durable Medical Equipment OR Occupational therapy OR exercise therapy OR physiotherapy OR therapy); few additional reports (3 SRs, 1 RCT) were included based on hand search. Retrieved abstracts and, if necessary, full-text references were checked for defined eligibility criteria by two independent raters and any disagreement was resolved by a third author. Overall, a total of 411 RCT and 114 SR reports were eligible and selected for the best evidence synthesis provided below. The following steps were taken from critical appraisal of evidence to the provision of recommendations (Chap. 2):
-
I.
For each included source (original paper, systematic review), the following was performed:
-
1.
Standardized evaluation of the methodology (internal validity with regard to study design issues and risk of bias).
-
2.
Classification of evidence level (1a to 5 according to the “Oxford Center for Evidence-Based Medicine—Levels of Evidence”, last version from March 2009, http://www.cebm.net/Oxford-centre-evidence-based-medicine):levels-evidence, March-2009 /).
-
3.
summarizing the results, conclusions, and deriving recommendations as applicable to the individual source,
-
1.
-
II.
Based on data from all sources on a specific therapy method (across original papers, systematic reviews, and meta-analyses), the following steps were taken:
-
4.
rating the overall quality of evidence of the sources included and thereby the confidence in the estimation of the effect strength (of a therapy) and,
-
5.
provision of graded recommendations.
-
4.
The quality of evidence is grouped into four categories according to the “GRADE” system (“Grades of Recommendation, Assessment, Development and Evaluation, GRADE”, www.gradeworkinggroup.org) (Schünemann et al. 2013):
-
High quality: further research is unlikely to affect our confidence in the estimation of the (therapeutic) effect or prognosis.
-
Medium quality: further research is likely to affect our confidence in the estimation of the (therapeutic) effect or prognosis and may alter the estimate.
-
Low quality: further research will most likely influence our confidence in the estimation of the (therapeutic) effect or prognosis and will probably change the estimate.
-
Very low quality: any estimation of the (therapy) effect or prognosis is very uncertain.
The grading of the recommendations according to GRADE (Schünemann et al. 2013) corresponds to the categories “ought to” (A) (strong recommendation) and “should” (B) (weak recommendation); for the purpose of this practice recommendation project, we added the category “can” (0) (option) (Platz 2017; Chap. 2). Recommendation category A is granted for clinically effective interventions with high-quality evidence support; with medium-quality evidence category B; and with low- or very low-quality evidence category 0 can be appropriate. Grading of recommendations is based on both quality of evidence including risk of bias, imprecision or heterogeneity of results, publication bias and other relevant factors for clinical decision-making such as clinical relevance of the approach, effect sizes reported, the benefit-risk ratio, scientific plausibility, and ease to transfer an approach to routine clinical practice.
3 Assessment
A systematic Cochrane overview performed a data synthesis across all Cochrane reviews for interventions targeting upper limb function after stroke. It describes a large variety of assessment tools for arm/hand impairment and function (activity limitations) (Pollock et al. 2014b). Some of the more frequently used measures that can be considered as valid, reliable, and with a clinically important focus are presented below. An indication is given whether the tests can be used for patients with mild, moderate, or severe arm paresis. These measures can be recommended for clinical use when the construct measured is of importance for clinical assessment, monitoring, or decision-making. It should be noted that no single measure is right or wrong, but has its role depending on the specific intention for assessment.
3.1 Measures of Impairment
3.1.1 Active Motor Control
-
Fugl-Meyer Assessment of Sensorimotor Recovery after Stroke (upper limb section) (FM Arm) (Fugl-Meyer et al. 1975) (mild-to-severe paresis).
-
Wolf Motor Function Test (WMFT) (Wolf et al. 2001) (mild-to-moderate paresis).
-
Rivermead Motor Assessment, arm section (RMA arm) (Lincoln and Leadbetter 1979) (mild-to-severe paresis).
-
Motricity Index (MI) (Demeurisse et al. 1980) (mild-to-severe paresis).
-
Dynamometer scores (including Jamar) (Bohannon 1997) (mild-to-moderate paresis).
The arm section of the Fugl-Meyer test is among the most widely used assessments in post-stroke rehabilitation research; it measures selective motion capacity with its motor subscale (range 0–66), somatosensory function (range 0–24), and passive joint motion/pain (range 0–44). Individual items are ordinally scaled (0–2). The Wolf Motor function test includes selective movement tasks and handling object tasks, each item ordinally scaled (0–5). In addition, time to complete tasks is measured. The Rivermead Motor Assessment arm section has 15 movement tasks with dichotomous evaluation. It is a hierarchical test (range 0–38). The Motricity Index is a simple three-item measure of degree of arm paresis (range 1–100); it is based on a weighted grading of MRC strength grades (Medical Research Council 1975). Dynamometer measure strength for a given movement, e.g. grip, numerically.
3.1.2 Spasticity/Resistance to Passive Movement
Ashworth Scale (AS) (Ashworth 1964), or Modified Ashworth Scale (MAS) (Bohannon and Smith 1987) or Resistance to Passive movement Scale (REPAS) (Platz et al. 2008).
Spasticity as clinically assessed with resistance to passive joint movements has most frequently been measured with the ordinally scaled Ashworth or Modified Ashworth Scale (range 0–4). The REPAS is a summary rating scale across joints based on the Ashworth scale and provides detailed instructions for performance and scoring to support reliability.
3.2 Measures of Arm, Hand, and/or Finger Function
-
Action Research Arm Test (ARAT) (Lyle 1981) (mild-to-moderate paresis).
-
Box and Block Test (BBT) (Desrosiers et al. 1994) (mild-to-moderate paresis).
-
Nine-Hole Peg Test (NHPT) (Kellor et al. 1971) (mild paresis).
-
Wolf Motor Function Test (WMFT) (Wolf et al. 2001) (mild-to-moderate paresis).
The ARAT is a hierarchical assessment of the ability to grasp, handle, and transport larger and smaller objects with different grips, which consists of four subtests and a total of 19 tasks (score ranges from 0 to 57). It has widely been used in stroke rehabilitation research. The BBT is a measure of manual dexterity. The number of one-inch cubes grasped and transported from one compartment of a box to the adjacent compartment within 1 min is measured. The NHPT measures finger dexterity by the time needed to grasp and place nine pegs in their holes.
3.3 Measure of Self-Perceived Usefulness of the Affected Arm in Daily Life
-
Motor Activity Log, MAL (Taub et al. 1993) (mild-to-moderate paresis).
The MAL is a structured interview intended to examine how much and how well people use their more-affected arm outside of the laboratory setting. Participants are asked standardized questions about the amount of use of their more-affected arm (amount of use, AOU) and the quality of their movement (QOM) during the 30 different functional activities indicated. Each item and subscale are rated between 0 (not used) and 5 (as before stroke) with half scores when indicated, i.e. 0.5, 4.5.
4 Therapy
The best evidence synthesis was grouped in the following sections: training, technology-supported training, and medication. Only a selection of relevant references has been cited. The complete list of references is presented online (www.clinical-pathways.org) for further information.
4.1 Training
A large variety of training strategies without the need for specific technical support is available for stroke survivors with arm paresis.
4.1.1 Dosage of the Therapeutic Time Prescribed and Organizational Forms of Therapy
A simple dose–response relationship does not exist for arm rehabilitation. In addition, it seems that a dedicated active arm motor training is a prerequisite to achieve further improvements when dosages are increased. Such a training provided for 2.5 h (Kwakkel et al. 1999) to 4–5 h (Winstein et al. 2004) per week over 4–20 weeks can accelerate arm motor recovery in the acute and subacute phase post-stroke. Two hours of training per week (Sehatzadeh 2015) or a total of more than 15 h of training seem to be necessary to achieve measurable effects on arm motor recovery (Pollock et al. 2014). Increasing therapy time up to 2 or 3 h per day has been shown to generate an additional benefit in the subacute phase post-stroke (Han et al. 2013). Improvement in the chronic phase was demonstrated after prolonged specific active motor training of ≥3 h per week (Cauraugh et al. 2011; Corti et al. 2012), while that was not the case for more passive “mobilization and tactile stimulation” therapy (Hunter et al. 2011). Of note, there was no significant effect for additional time spent in exercise therapy for very high intensity (6 h per day) vs. moderately high intensity (up to 3 h per day) constraint-induced movement therapy (Kwakkel et al. 2015).
Aside from one-to-one therapy, treatment can be organized as arm rehabilitation circuit training (Blennerhassett and Dite 2004; Pang et al. 2006) or as supervised home training (e.g. Harris et al. 2009; Almhdawi et al. 2016). For the latter, dedicated manuals for patients with mild, moderate, or severe paresis with repetitive impairment- and activity-oriented training seem to support therapeutic success in the subacute phase and client-centred individualized functional goal-oriented practice in the chronic phase.
An alternative form of organizing rehabilitation service is telerehabilitation that with limited evidence has been shown to be feasible (Wolf et al. 2015) and equally effective as institution-based arm therapy (Laver et al. 2013). Caregiver-mediated exercises have not been shown to be beneficial for arm rehabilitation post-stroke (Vloothuis et al. 2016).
In case an individual has very poor predicted recovery (complete arm paralysis with diagnosed severe corticospinal tract damage; i.e. loss of motor evoked potentials (MEP) with transcranial magnetic stimulation (TMS), posterior limb of internal capsule damage on MRI diffusion tensor imaging (DTI)), a therapeutic focus on prevention of secondary complications (only) for the plegic arm and teaching compensatory strategies already early after stroke can be effective and show little risk to miss out a recovery potential (Stinear et al. 2017).
4.1.2 “Schools” of Therapy
Bobath therapy (neurodevelopmental therapy, NDT) and other “schools” of therapy have not been shown to specifically support arm motor recovery post-stroke (Langhorne et al. 2009).
4.1.3 Type of Feedback Given
Extrinsic feedback has its role in clinical practice. Proof of clinical effectiveness is, however, rather limited (e.g. Israely and Carmeli 2016).
4.1.4 Bilateral Training
Quite a few RCTs compared bilateral to unilateral training approaches. Overall, equivocal or inferior benefits after bilateral training were reported. Among mildly affected chronic stroke survivors, activities and actual amount of use were better promoted with unilateral training: a Cochrane review (Pollock et al. 2014) indicated better arm function (6 trials, 375 participants; standardized mean difference, SMD 0.20, 95% confidence interval, 95% CI 0.00 to 0.41) and ADL improvement (3 trials, n = 146) after unilateral compared to bilateral training. Effects at impairment level had been comparable (4 trials, n = 228).
When repetitive bilateral symmetrical active–passive movements were, however, used as priming before active rehabilitation of the affected limb, recovery could be accelerated in subacute stroke patients (Stinear et al. 2014).
4.1.5 Impairment-Oriented Training
One single-centre (n = 60) and two multicentre RCTs (n = 60 and n = 148, resp.) demonstrated a superior effect of the impairment-oriented arm training, i.e. the arm basis training for severe arm paresis and the arm ability training for mild arm paresis. The arm basis training had a bigger effect on selective motion capacity (FM Arm) with incomplete severe arm paresis compared to therapeutic time equivalent control therapies, i.e. Bobath therapy or “best conventional” therapy (Platz et al. 2005; Platz et al. 2009). The graded training enhances selective motion capacities by a systematic repetitive training of individual joint motions without gravity influence to start with, with gravity influence next, and finally multi-joint movements in a progressive training scheme. The arm ability training improved sensorimotor efficiency with arm activities (TEMPA; Desrosiers et al. 1993) with a long-term effect (Platz et al. 2001) and superiority compared to therapeutic time-equivalent “best conventional” therapy (Platz et al. 2009). It specifically trains speed and accuracy of abilities such as fast finger movements, aiming, visuomotor tracking, steadiness, and dexterity as well as endurance. Taken together, the training techniques provide a comprehensive modular approach for arm paresis after stroke with a moderate differential beneficial effect compared to an active control arm therapy of the same therapeutic time (SMD 0.47, 95% CI 0.12 to 0.81; two studies, number of participants analyzed by individual patient data meta-analysis = 135) (Platz et al. 2015/in preparation).
4.1.6 Task-Specific Training
Task-specific training where training tasks resemble an activity received substantial attention in RCTs with variable results, mostly indicating no or a comparable effect to control therapies both for subacute and chronic patients and a lack of dose–response relationship. A Cochrane review summarized that there is low-quality evidence that repetitive task training (RTT) improves arm function (SMD 0.25, 95% CI 0.01 to 0.49; 11 studies, number of participants analyzed = 749), hand function (SMD 0.25, 95% CI 0.00 to 0.51; eight studies, number of participants analyzed = 619) with significant differences between groups up to 6 months post-treatment (SMD 0.92, 95% CI 0.58 to 1.26; three studies, number of participants analyzed = 153), but not after 6 months; effects were not modified by intervention type, dosage of task practice, or time post-stroke and were no longer significant when studies with high or unclear risk of bias were removed from the meta-analysis (French et al. 2016).
A stronger benefit had been substantiated in one RCT included in the meta-analysis where patients received training tasks that resemble a meaningful activity while the therapist nevertheless focused on motor control issues (impairment level) as therapeutic target (Arya et al. 2012). This was similarly demonstrated for task-oriented mirror therapy (Arya et al. 2015).
4.1.7 Constraint-Induced Movement Therapy (CIMT)
The constraint-induced movement therapy (CIMT) is a response to the observed behaviour that some stroke survivors who suffer from arm paresis learn to cope with their non-affected arm. Even when their affected arm recovers, the learnt non-use behaviour persists. The psychologist Edward Taub, Ph.D. provided a therapeutic approach that reverses the learnt non-use behaviour by restraint of the non-affected arm and massed practice of the affected arm with gradual increase of motor challenges, i.e. “shaping” (Taub et al. 1993). The original CIMT includes 6 h of “shaping” as one-to-one therapy and 90% of waking hours restraint of the non-affected arm. Modified versions, i.e. mCIMT, use up to 3 h therapy sessions with massed practice and less than 90% of waking hours restraint (if any), e.g. a couple of hours per day. The approach (CIMT and mCIMT) is suitable for individuals with learnt non-use and only moderate arm paresis with some preserved hand function and no severe pain or spasticity problem in their affected arm.
CIMT is the arm rehabilitation technique with the broadest evidence base including >50 RCTs and many systematic reviews.
A Cochrane review (Corbetta et al. 2015) (systematic search up to January 2015) with 42 RCTs (1453 participants) and 40 RCTs being used for meta-analysis assessed effects of CIMT and mCIMT or “Forced Use, FU” (restraint only) compared to different control interventions. A high risk for bias by small trials was noted.
Immediate post-training effects were non-significant for ADL activities (Functional Independence Measure, FIM or Barthel Index, BI; 11 studies, 344 participants; SMD 0.24, 95% CI −0.05 to 0.52). A small significant effect on arm motor function (ARAT, WMFT, Emory Motor Function Test, Manual Function Test, RMA; 28 studies, 858 participants; SMD 0.34, 95% CI 0.12 to 0.55) was documented. A moderate significant effect was found for dexterity (Peg Tests, BBT; 4 studies, 113 participants; SMD 0.42, 95% CI 0.04 to 0.79), a significant effect on perceived arm motor function (MAL quality of movement, 24 studies, 891 participants; +0.68 points, 95% CI 0.47 to 0.88; MAL amount of use, 23 studies, 851 participants; +0.79 points, 95% CI 0.50 to 1.08), a moderate to large statistically significant effect on arm motor impairment (FM arm, Chedoke McMaster Impairment Inventory, grip strength, isometric strength; 16 studies, 372 participants; SMD 0.82, 95% CI 0.31 to 1.34), and a non-significant effect on quality of life, QoL (SIS, 3 studies, 96 participants; mean difference, MD +6,54 points, 95% CI −1.2 to 14.28). Three studies involving 125 participants explored disability after a few months of follow-up and found no significant difference (FIM or BI; SMD −0.20, 95% CI −0.57 to 0.16), yet numerically in favour of conventional treatment.
Overall, small effects on arm motor function and perceived arm motor function, moderate effects on dexterity, and larger effects on arm motor impairment could be substantiated after training, while effects on disability and QoL remained non-significant; evidence for long-term effects is rather limited.
Time post-stroke and type of CIMT (mCIMT vs. CIMT) seem not to be critical factors for its effectiveness (Kwakkel et al. 2015) with the exception that early after stroke mCIMT seems preferable over CIMT (Nijland et al. 2011).
A restraint outside therapeutic sessions promotes actual amount of use (only) when combined with a “transfer package”, i.e. an individualized critical reflection of “obstacles” to use the affected arm in daily life and how to overcome them individually (Taub et al. 2013).
4.1.8 Strength Training
Strengthening exercises, at least when offered as one component of training, were shown to have a positive effect on grip strength (SMD 0.97, 95% CI 0.05 to 1.85; 6 studies, 306 participants) and arm function (SMD 0.21, 95% CI 0.03 to 0.39; 11 studies, 465 participants), but not on disability (ADL) for patients with (mild to) moderate arm paresis (subacute and chronic phase) as indicated by a systematic review (Harris and Eng 2010).
In a large multicentre RCT recruiting acute/subacute stroke patients with significant, yet incomplete arm paresis (288 participants, 2 to 60 days post-stroke) received up to 1.5 h daily training for 6 weeks either as functional strength training (FST) or movement performance therapy (MPT), each combined with conventional therapy. FST consisted of progressive resistive exercise during functional tasks and MPT of “hands on” guidance for smooth and accurate movements. No significant intergroup differences were observed at the end of intervention (ARAT, WMFT, hand and pinch grip force) (Pomeroy et al. 2018).
4.1.9 Mirror Therapy
During mirror therapy a patient performs certain movements with their non-affected arm while looking into a mirror. The mirror is placed in the patient’s midsagittal plane and therefore reflects the non-paretic side as if it were the affected side that moves. It is assumed that by the virtual visual image of the normally moving affected arm, the brain network subserving its control is activated. If this therapy is performed for about 30 min every day for several weeks, motor function has improved and disability reduced in both subacute and chronic phases.
A Cochrane review (Thieme et al. 2018) included 62 studies (ten studies addressing the lower limb) with a total of 1982 participants that compared mirror therapy with other interventions. When compared with all other interventions, mirror therapy for the arm had a significant effect on motor function (activity level) (post-intervention data: SMD 0.46, 95% CI 0.23 to 0.69; 31 studies, 1048 participants). In addition, mirror therapy improved the capability for selective movements (post-intervention data: FM, arm motor score MD 4.32, 95% CI 2.46 to 6.19; 28 studies, 898 participants). The authors found a significant positive effect on pain (SMD −1.10; 95% CI −2.10 to −0.09) which is influenced by patient population and a non-significant positive effect on visuospatial neglect (SMD 1.06; 95% CI −0.10 to 2.23). The effects on motor function could not be substantiated at follow-up assessment after 6 months. The therapeutic effects on motor function were statistically significant when mirror therapy was compared with a sham intervention where the affected arm was invisible, but not when it could be seen.
Therefore, mirror therapy could be applied as an additional intervention in the rehabilitation of patients after stroke. This includes supervised training, including home training for selected patients with a high degree of compliance (Tyson et al. 2015). It is still unclear whether mirror therapy can replace other interventions for improving motor function of the arm. It might especially be suitable as additional training for patients with more severe arm paresis who could not train their affected arm actively by themselves.
4.1.10 Mental Practice
During mental practice (MP) a subject repeatedly mentally rehearses an action or task without physically performing it. In rehabilitation trials, MP was typically used after an actual training session with the paretic arm, ranging from twice a week to daily sessions, each lasting 10 to 60 min for 3–10 weeks.
According to two Cochrane reviews, MP in combination with other treatment appeared more effective in improving upper extremity (impairment and) activity than the other treatment alone (5 studies, 105 participants; SMD 1.37, 95% CI 0.60 to 2.15; Barclay-Goddard et al. 2011; 7 studies, 197 participants; SMD 0.62, 95% CI 0.05 to 1.19; Pollock et al. 2014b). Effects on disability (ADL) were not substantiated. Due to small numbers in each group, subgroup analyses based on time since stroke and dosage of MP were not feasible.
4.1.11 Action Observation
The evidence reviewed is too weak and inconsistent to serve as a basis for clinical practice recommendations.
4.1.12 Music Therapy and Rhythmic Auditory Stimulation
A Cochrane review (Magee et al. 2017) examined the effects of music interventions on upper extremity function. The review included nine studies with a total of 308 participants and found no evidence of effect for music interventions for general upper extremity function, range of motion (shoulder flexion), hand function, upper limb strength, and manual dexterity; two studies indicated that music therapy improved timed performance somewhat.
Further, the addition of rhythmic auditory stimulation or rhythmic auditory cueing with repetitive training of reaching movements might generate some benefit (Yoo and Kim 2016).
4.2 Technology-Supported Training
4.2.1 Passive Devices for Repetitive Arm and Hand Training
A variety of passive devices have been developed for repetitive practice in arm and hand rehabilitation. They include a “reha-slide” the “Bilateral Arm Training with Rhythmic Auditory Cueing, BATRAC”, an “Upper Limbs’ Encircling Motion, ULEM” apparatus, an arm ergometer (bidirectional hand cycle commonly used for upper body aerobic exercise), and hand orthoses that passively assist finger extension. The devices have been tested in a few RCTs with subacute and chronic stroke patients with moderate-to-severe arm paresis without big effects or superiority; they are an option, e.g. when integrated in a circuit class approach.
4.2.2 Trunk Restraint
Trunk restraint has been used when reaching movements are trained and accompanied by excess trunk movements as compensation for limited shoulder flexion. It has a moderate effect on reduction of upper extremity impairment (improvement of FM Arm score) and increase of active shoulder flexion during reaching as shown for chronic stroke patients with (mild to) moderate upper extremity impairment (FM Arm: SMD 0.54, 95% CI 0.06 to 1.01; 3 trials with 71 participants; active shoulder flexion: SMD 0.45, 95% CI 0.11 to 0.79; 5 trials with 138 patients) (Wee et al. 2014).
4.2.3 Splints and Strapping
Using splints and other orthoses to immobilize joints, e.g. the wrist of the severely affected arm, or shoulder strapping (glenohumeral, sacapulo-thoracal) does not facilitate motor recovery (Veerbeek et al. 2014). It can have a positive therapeutic and prophylactic effect on pain of the joint treated (wrist orthosis and shoulder strapping) (Appel et al. 2014; Bürge et al. 2008).
4.2.4 Arm Rehabilitation Using Virtual Reality (VR) Applications
Both VR games using a commercially available gaming consoles and purpose-built VR-based rehabilitation systems have been used clinically and in clinical trials for arm rehabilitation post-stroke.
Laver et al. (2017) presented in an updated Cochrane review a meta-analysis, which indicated no differential effect on arm function and activities when comparing virtual reality to conventional therapy (SMD 0.07, 95% CI −0.05 to 0.20; 22 studies, 1038 participants, low-quality evidence). However, when virtual reality was used in addition to usual care (providing a higher dose of therapy for those in the intervention group), there was a statistically significant difference between groups in favour of VR (SMD 0.49, 95% CI 0.21 to 0.77; 10 studies, 210 participants, low-quality evidence). These results were mainly based on FM Arm scores (16 trials with 599 participants; MD 2.85, 95% CI 1.06 to 4.65). In subgroup analyses, beneficial effects were corroborated for trials providing more than 15 h of intervention (trend) and when using a specialized rehabilitation training system rather than a commercially available gaming console; comparisons between subgroups were, however, not significant. Time after stroke and severity of arm paresis did not modify the effects significantly.
In one of these non-robot purpose-built VR intervention systems, the therapist’s arm movement with an object as shown in the VR (“end effector”-based training) has to be mimicked by a patient. The system provides both “knowledge of performance” (quality of the arm movement) and “knowledge of result” (precision of the movement) based on movement tracking with an electromagnetic system. It has been tested in subacute to chronic stroke patients with mild-to-moderate arm paresis with 1 h VR training plus 1 h traditional arm training five times a week for 4 weeks compared to 2 h per day traditional training. This consistently showed some differential beneficial effect on selective motion capacity and disability (e.g. Kiper et al. 2014). For subjects with more severe arm paresis, a system that integrates arm weight support can be used; the therapy is equally effective as time-equivalent conventional therapy (e.g. Prange et al. 2015).
4.2.5 EMG- and Neuro-Biofeedback
Only low-quality evidence comparing EMG biofeedback with physiotherapy is available with some suggestion that biofeedback may have some beneficial impact (Pollock et al. 2014).
Neurofeedback therapy activation of cerebral motor areas can either be used for direct feedback training, e.g. as target domain during motor imagery or as signal to induce electrical stimulation or robot-assisted movements of the paretic limb. Data from first clinical trials suggest that neurofeedback can have a therapeutic effect (e.g. Mihara et al. 2013; Ramos-Murguialday et al. 2013). Currently, the technology is seen as investigational.
4.2.6 Neuromuscular Electrical Stimulation (NMES)
NMES either can stimulate nerves and muscles cyclically independent of a person’s volition to move (cNMES) or triggered by an EMG signal induced by voluntary muscle activity (EMG-triggered NMES, EMG-NMES), controlled by voluntary movements of the non-affected side (“contralaterally controlled FES, CCFES”) or can be used within a functional task as functional electrical stimulation, FES, e.g. when single- or multi-channel NMES is used for grasp and release activities.
License and restrictions for use need to be considered for each device used as provided by its manufacturer.
Veerbeek et al. 2014 have carried out a series of meta-analyses of RCT data related to electrical stimulation (49 RCTs, n = 1521).
Their meta-analyses indicated that cNMES of wrist and finger flexors and extensors (22 trials, 894 participants) (but not cNMES of wrist and finger extensors) improved selective movement capacity (modified FM Arm: SMD 0.91, 95% CI 0.29 to 1.53; 41 participants) and hand function (Jebsen-Taylor hand function test, JTHFT: SMD 0.88, 95% CI 0.05 to 1.71; 23 participants), but not arm-hand activities. EMG-NMES of wrist and finger extensors (25 trials, 492 participants) improved active range of motion (SMD 1.16, 95% CI 0.28 to 2.03; 61 participants), selective movements (FM Arm, Chedoke-McMaster stroke assessment, CMMSA: SMD 0.58, 95% CI 0.04 to 1.12; 49 participants), arm-hand activities (ARAT, BBT, JTHFT, 10CMT (10-cup moving test), and FTHUE (Functional test for the hemiplegic upper extremity: SMD 0.72, 95% CI 0.41 to 1.03; 162 participants), but had no significant effect on muscle strength and muscle tone. Subgroup analyses revealed no significant differences between post-stroke phases. No evidence revealed a beneficial effect of transcutaneous electrical nerve stimulation (TENS) (four trials, 484 participants).
cNMES of shoulder muscles decreased shoulder subluxation (x-ray: SMD 0.58, 95% CI 0.15 to 1.01; 190 participants) while it did not significantly affect motor function, ROM, or pain; the effect might be restricted to the subacute phase (Vafadar et al. 2015).
For a selection of patients, FES can be used to facilitate high-intensity self-training, e.g. when hand opening is induced by EMG-NMES integrated in an FES orthosis for patients with severe finger extension paresis, and thereby motor function improved (effect size for intergroup differences of change scores: FM Arm-distal d = 1.16, 95% CI 0.21 to 2.10; 24 participants) (Shindo et al. 2011).
4.2.7 Arm Robot Therapy
Arm robots have been developed to repetitively train shoulder and elbow movements (e.g. MIT-manus, MIME, NeReBot, InMotion Linear Robot, UL-EX07, ReoGo), forearm and wrist movements (e.g. NeReBot, Bi-Manu-Track, Hand Mentor, UL-EX07), or finger movements (e.g. Reha-Digit, Amadeo, Hand Mentor, Gloreha). They have the advantage that with assistance by the robot (as needed) stroke survivors can repetitively train active movements even when they have severe arm paresis. High repetition rates for selective movements can thus be achieved during training sessions.
Mehrholz et al. (2018) included 45 trials (involving 1615 participants) in their updated Cochrane review. In these trials, therapy has usually been applied five times a week for 20–105 min, mostly for 2–6 weeks.
Electromechanical and robot-assisted arm training improved activities of daily living scores (SMD 0.31, 95% CI 0.09 to 0.52; 24 studies, 957 participants, high quality of evidence), arm function (SMD 0.32, 95% CI 0.18 to 0.46; 41 studies, 1452 participants, high quality of evidence), and arm muscle strength (SMD 0.46, 95% CI 0.16 to 0.77; 23 studies, 826 participants, high quality of evidence). Electromechanical and robot-assisted arm training did not increase the risk of participant drop-out (RD 0.00, 95% CI −0.02 to 0.02; 45 studies, 1615 participants, high quality of evidence), and adverse events were rare. The test for subgroup differences between a subgroup of participants who received mainly training for the distal arm and the hand (finger, hand, and radioulnar joints) and a subgroup of participants who received training mainly of the proximal arm (shoulder and elbow joints) revealed no significant difference for either arm function or activities of daily living scores at the end of intervention. Effects on ADL could be substantiated for the subgroup treated during the acute and subacute phase, but not for the subgroup with participants in the chronic phase (i.e. more than 3 months after stroke).
Taken together, people who receive electromechanical and robot-assisted arm and/or hand training during the acute/subacute phase after stroke improve their activities of daily living, arm and hand function, and muscle strength while effects in the chronic phase are uncertain.
Another systematic review with meta-analyses excluding RCTs of “insufficient quality” corroborated a small effect of robot therapy on motor function (FM Arm: MD 2.23, 95% CI 0.87 to 3.59; 28 RCTs, 884 participants) and (only) at trend level for activities of daily living (SMD 0.27, 95% CI −0.05 to 0.59; 14 studies with 427 participants) (Veerbeek et al. 2017). In dose-matched trials (i.e. equal therapeutic time allocated), a differential benefit of robot-assisted therapy on motor recovery was still observed (FM Arm: MD 2.28, 95% CI 0.89 to 3.68; 26 RCTs, 808 participants).
The high number of selective repetitive movements generated during robot therapy is probably the main reason for its therapeutic effect.
A specifically task-oriented robot therapy used by high functioning stroke survivors did not achieve a superior benefit at the activity level compared to task-oriented training without robot (Timmermans et al. 2014).
4.2.8 Repetitive Transcranial Magnetic Stimulation (rTMS)
rTMS has been applied during the acute, post-acute, and chronic post-stroke phases to improve motor recovery in stroke patients having upper and/or lower limb paresis. The rationale has been that priming the arm motor cortex by an excitatory stimulation of the lesioned hemisphere or by an inhibitory stimulation of the non-lesioned hemisphere (that itself might inhibit the ipsilesional motor cortex) can promote arm motor recovery.
The following “inhibitory” (I) or “excitatory” (E) types of rTMS have been used in arm motor rehabilitation:
Low-frequency (LF) rTMS (I) of the contralesional primary motor cortex (M1),
Continuous theta burst stimulation (cTBS) (I) of the contralesional M1,
High-frequency (HF) rTMS (E) of the ipsilesional M1, and,
Intermittent theta burst stimulation (iTBS) (E) of the ipsilesional M1.
Thirty-four studies with 904 participants were included in the systematic review by Zhang et al. (2017). Upper limb function was measured by grip force, movement accuracy, keyboard, tapping, pinch and lift force, or complex hand movements. Pooled estimates show that rTMS significantly improved short-term (SMD 0.43, 95% CI 0.30 to 0.56) and long-term (SMD 0.49, 95% CI 0.29 to 0.68) manual dexterity. The mean effect size for the acute subgroup was 0.69 (95% CI 0.41 to 0.97), for subacute stroke 0.43 (95% CI 0.16 to 0.70), and for chronic stroke 0.34 (95% CI 0.00 to 0.69; P = 0.048), respectively. The mean effect size for the high-frequency rTMS subgroup was 0.45 (95% CI 0.22 to 0.69) versus 0.42 (95% CI 0.26 to 0.58) for the low-frequency rTMS subgroup. The mean effect size was significant for intermittent theta burst stimulation (iTBS) subgroup at 0.60 (95% CI 0.10 to 1.10), but was only at a trend level for continuous theta burst stimulation (cTBS) at 0.35 (95% CI −0.11 to 0.81; n.s.). The pooled effect size for the subcortical subgroup was 0.66 (95% CI 0.36 to 0.95); the mean effect size for non-specified subgroup was 0.39 (95% CI 0.24 to 0.54). In addition, 25 studies were divided into four subgroups based on the numbers of sessions of the treatment: 1 session, 5 sessions, 10 sessions, and 15 to 16 sessions, with the mean effect sizes as follows: 0.55 (95% CI 0.29 to 0.81) for 1 session; 0.67 (95% CI 0.41 to 0.92) for 5 sessions; 0.20 (95% CI −0.06 to 0.41; n.s.) for 10 sessions; and 0.08 (95% CI −0.36 to 0.51; n.s.) for 15–16 sessions. Only three studies reported mild adverse events such as headache and increased anxiety.
In summary, the data showed a moderate short- and long-term benefit and only rarely reported mild adverse events, i.e. harm. Contralesional LF rTMS and ipsilesional HF rTMS or iTBS were effective with a maximum effect for trials applying 5 sessions; the situation is less clear for contralesional cTBS. The modifiers of treatment effect are to be taken into account with bigger benefits earlier after stroke and with patients suffering from subcortical stroke. Trials frequently included patients with mild-to-moderate arm paresis.
The systematic review by Xiang et al. 2019 including 42 RCTs with a total of 1168 participants reported qualitatively similar results.
Evidence for combined treatments (bilateral HF and LF rTMS within sessions or across series of sessions) also showed benefits. E.g., 2 weeks contralesional LF rTMS, followed by ipsilesional iTBS, had superior effects to the reverse order and sham stimulation in subacute stroke survivors with moderate-to-severe arm paresis (Wang et al. 2014). The database for such combinations is, however, still limited.
Although rTMS appears generally safe for stroke patients, it is necessary to follow the safety guidelines to prevent or minimize the risk of side effects (Rossi et al. 2009). In addition, characteristics of the medical product used for rTMS (including licensing) need to be taken into consideration.
4.2.9 Repetitive Peripheral Magnetic Stimulation (rPMS)
A Cochrane review documented one RCT (63 participants with spastic paresis) with no effect of rPMS in addition to rehabilitation treatment on arm function (FM Arm), while there was some effect on elbow spasticity (Momosaki et al. 2017).
4.2.10 Transcranial Direct Current Stimulation (tDCS)
Compared to rTMS, tDCS is a less focal non-invasive method. tDCS is used to modulate cortical excitability by applying a direct current to the brain; tDCS using anodal stimulation (a-tDCS) might lead to increased cortical excitability (e.g. of the lesioned hemisphere), whereas cathodal stimulation (c-tDCS) might lead to decreased excitability (e.g. of the non-lesioned hemisphere); in addition, anodal stimulation and cathodal stimulation may be applied simultaneously (dual-tDCS).
A systematic Cochrane review including 12 trials with a total of 431 participants measured upper extremity function at the end of a tDCS intervention period and revealed no evidence of an effect in favour of tDCS (a-tDCS, c-tDCS, dual-tDCS) compared to any type of placebo or passive control intervention (SMD 0.01, 95% CI −0.48 to 0.50 for studies presenting absolute values (low-quality evidence) and SMD 0.32, 95% CI −0.51 to 1.15 (low-quality evidence) for studies presenting change values) (Elsner et al. 2016). Regarding the effects of tDCS on upper extremity function at the end of follow-up, four studies with a total of 187 participants (absolute values) showed no evidence of an effect (SMD 0.01, 95% CI −0.48 to 0.50; low-quality evidence). Similarly, a network meta-analysis failed to substantiate an effect of tDCS on arm function among stroke survivors (16 RCTs, 302 participants) (Elsner et al. 2017).
4.2.11 Somatosensory Stimulation
Somatosensory stimulation that can be provided as tactile, thermal, vibratory, pneumatic, or electrical stimulation might have a potential to enhance recovery not only in the somatosensory but also in the motor domain. Overall, the efficacy data from clinical trials is not yet convincing regarding its effects on motor recovery (Veerbeek et al. 2014). Moderate-quality evidence, arising from a small RCT (29 participants), showed that sensory stimulation (alternating cold and “hot” thermal stimulation) combined with active movement efforts had a beneficial effect on arm activities and impairment, when compared to no treatment in acute and subacute stroke patients with moderate-to-severe arm paresis (Modified Motor Assessment Scale: MD 1.58, 95% CI 0.98 to 2.18; Brunnstrom Score: MD 0.19, 95% CI 0.09 to 0.29; 29 participants) (Chen et al. 2005). Similar results (Stroke Rehabilitation Assessment of Movement, ARAT) of thermal stimulation were reported for mostly chronic patients (23 participants; Wu et al. 2010).
4.2.12 Acupuncture
The evidence for acupuncture in arm rehabilitation post-stroke—more frequently assessed for the subacute phase—is considered of low quality due to design issues, heterogeneity, or lack of differential effects in clinical trials. A systematic review highlights the big issues of risk of bias in the published data (Cai et al. 2017). A bigger RCT randomizing 295 subacute stroke patients with moderate-to-severe arm paresis documented acupuncture for 30 min six times a week for 4 weeks to be equally effective as physiotherapy (60 min neurodevelopmental therapy) and occupational therapy (45 min) or the combination of both acupuncture and physiotherapy as well as occupational therapy (Zhuang et al. 2012).
4.2.13 Investigational Devices
Clinical evidence for post-stroke arm rehabilitation with other investigational devices such as the brain–machine interface-supported therapy, vagal nerve stimulation, or epidural electrical motor cortex stimulation is too limited for a recommendation for their use outside study protocols.
4.3 Medication
4.3.1 Botulinum Neurotoxin A (BoNT A)
Treatment of spasticity is covered in the corresponding chapter of this book. There is considerable evidence that BoNT A reduces spasticity, pain associated with spasticity, and consequently difficulties to integrate a plegic severely spastic arm in daily activities (Andringa et al. 2019); there is, however, less evidence to suggest that BoNT A improves active arm function even though this can be the case individually (e.g. Foley et al. 2013).
4.3.2 Drugs to Enhance Recovery
Medication thought to modify neuroplasticity and motor recovery post-stroke has not been investigated extensively. Any data regarding arm rehabilitation is regarded preliminary, e.g. for l-dopa, donepezil, d-amphetamine, fluoxetine, or cerebrolysin and not yet sufficient to recommend its routine clinical use. Individualized treatment decisions (e.g. for l-dopa, fluoxetine, or cerebrolysin) are at the discretion of the physician in charge, mostly as “off-label” treatment.
5 Clinical Pathway and Practice Recommendations
5.1 General Comments
As presented in the paragraph “therapy”, there is ample evidence for benefits gained by dedicated arm rehabilitation therapy in terms of improvement of active motor control (impairment) and activities. Self-perceived usefulness of the affected arm as well as disability has been shown to improve with some interventions. Overall, the immediate benefits from various interventions are moderate, long-term effects were less systematically assessed. If applied appropriately, the interventions show little risk to harm and are by and large very acceptable for patients. The question of feasibility to apply these clinically effective therapies rests largely on access to and the availability of skilled human therapeutic resources. The use of technical devices such as arm rehabilitation “robots”  (), NMES ($) requires additional investment (high $$, moderate $) and therapeutic skills. Our knowledge of cost-effectiveness, i.e. the change in costs to the change in therapeutic effects, is limited.
(), NMES ($) requires additional investment (high $$, moderate $) and therapeutic skills. Our knowledge of cost-effectiveness, i.e. the change in costs to the change in therapeutic effects, is limited.
Arm rehabilitation therapy is always embedded in the overall goal-oriented rehabilitation plan. As such, there is no tight link between an individual arm impairment or activity limitations and a certain therapeutic intervention and strategy. Rather, arm rehabilitation needs individually to be tailored and therapeutic decisions need to be weighed with a reflection of the overall rehabilitation goals and their priority.
In addition, therapeutic decisions in arm rehabilitation rest on underlying therapeutic principles, e.g. what are the next steps of motor control improvement to be taken. They might well be achievable by alternative therapeutic options as locally available. Teams might want to use the recommendations given below to structure their service and local “arm rehabilitation pathway” in such a way that the diverse patients’ needs can effectively be addressed. As stated above, this is not primarily a matter of technical devices: Evidence-based smart and skilled therapeutic approaches do not have to imply huge “costs” for equipment. Conversely, technical devices can reduce the workload for human resources and can be cost-effective.
This chapter gives insight into the currently best available evidence when improvement of (sensori)motor control, i.e. body functions, or activities performed with the affected arm and hand is a therapeutic goal. Therapeutic affordances will tentatively differ early after stroke, when functional restauration might be a prevailing goal, while in the chronic stage therapeutic goals might more frequently focus on activities primarily. Further, therapeutic steps to be taken by patients with severe arm paresis are quite different from those with mild-to-moderate arm paresis and therapeutic interventions need to be chosen accordingly. Hence, the evidence-based recommendations provided below will be given for the acute, subacute, and the chronic phase and for patients with severe or mild-to-moderate paresis separately. On an individual basis, there might be good reasons to take different decisions based on a patient’s values and preferences and a healthcare professional’s expertise.
5.2 Dosage and Organization of Treatment
5.2.1 Acute and Subacute Phase After Stroke
A dedicated active arm motor control training of at least 2 h per week over several weeks ought to be provided when acceleration of arm motor recovery is intended post-stroke (level of evidence 1a, quality of evidence high, A+). Increasing the therapy time up to 3 h per day can create a benefit in the subacute stage and can be considered individually (level of evidence 1b, quality of evidence moderate, 0).
Depending on individual circumstances (patient and service) (I) one-to-one therapy, (II) circuit class training that can integrate the use of passive training apparatuses, and (III) intermittently supervised home training, the latter ideally with dedicated training manuals for patients with mild, moderate, or severe paresis, resp., with a focus on repetitive impairment- and activity-oriented training, and documentation of the training should be entertained as therapeutic options (level of evidence 1b, quality of evidence moderate, B+).
In cases with an individually very poor prediction (complete paralysis with diagnosed severe corticospinal tract damage (loss of motor evoked potentials (MEP) with transcranial magnetic stimulation (TMS), posterior limb of internal capsule damage on MRI diffusion tensor imaging (DTI)), a therapeutic focus on prevention of secondary complications (only) for the plegic arm and teaching compensatory strategies with the non-paretic arm can be considered already early after stroke (level of evidence 2b, quality of evidence low [imprecision], 0).
5.2.2 Chronic Phase After Stroke
At least 3 h per week of dedicated active arm motor rehabilitation (including circuit class approaches and the use of training apparatuses or home practice with intermittent supervision) with regular evaluation of therapeutic progress are recommended for prolonged therapy in the chronic stage when improvements at impairment or activity level are intended and can be observed (level of evidence 1b, quality of evidence moderate, B+).
When improved performance of the affected arm in individually valued functional tasks is the therapeutic goal, intensified home practice with intermittent supervision for a couple of weeks is recommended; the training should then focus on client-centred individualized functional goal-oriented practice (level of evidence 1b, quality of evidence moderate, B+).
5.3 Therapeutic Options (Table 1)
5.3.1 Therapeutic Options for Stroke Survivors with Severe Paresis
Daily arm basis training should be considered in the acute and subacute phase when improvement of selective motion capacity is the therapeutic goal for patients with incomplete severe arm paresis (level of evidence 1b, quality of evidence moderate, B+); it is an option in the chronic phase (0).
If available, arm robot therapy should be offered on a daily base especially to increase dosage/intensity of repetitive selective movements in the acute/subacute phase when selective movement capacity recovery is therapeutic goal (level of evidence 1a, quality of evidence high, B+ [unclear long-term effects]); it is also an option in the chronic phase (level of evidence 1a, quality of evidence low [uncertain effects], 0).
Daily mirror therapy as additional training for several weeks, e.g. as supervised self-training, should be considered when motor improvement at impairment and/or activity level is intended (and likely to be achieved) (level of evidence 1a, quality of evidence moderate, B+); patients with neuropathic pain or neglect might have an additional benefit.
Bilateral training is a therapeutic option (level of evidence 1a, quality of evidence moderate, 0).
cNMES of wrist and finger flexors and extensors, or when EMG-triggering is voluntarily possible, EMG-NMES of wrist and finger extensors, in selected cases as FES can be used to enhance selective motion capacity and arm activities (level of evidence 1a, quality of evidence low, 0). cNMES of shoulder muscles can be applied to treat subluxation (level of evidence 1a, quality of evidence low, 0).
Purpose-built virtual reality therapy systems with arm weight support can be used to improve selective motion capacity and arm activities with severe incomplete arm paresis (level of evidence 1b, quality of evidence low, 0).
Somatosensory (especially thermal) stimulation is an option as adjunct therapy (level of evidence 1b, quality of evidence moderate, 0), in the subacute phase also acupuncture (level of evidence 1b, quality of evidence low, 0).
Using splints and other orthoses to intermittently immobilize joints, e.g. the wrist of the severely affected arm or shoulder strapping (glenohumeral, scapulo-thoracal), does not facilitate motor recovery and should not be used for this therapeutic goal (level of evidence 1a, quality of evidence moderate, B−). It can be used to prevent or treat pain associated with severe paresis at these joints (level of evidence 1a, quality of evidence low, 0).
Substantial difficulties to integrate a plegic and severely spastic arm in daily activities and spasticity-associated pain should trigger the evaluation of a botulinum neurotoxin A treatment of the affected muscle groups (level of evidence 1a, quality of evidence moderate, B+).
5.3.2 Therapeutic Options for Stroke Survivors with Moderate and Mild Paresis
A three-week course of daily arm ability training should be considered when improvement of sensorimotor skilfulness (e.g. dexterity) is the therapeutic goal for patients with mild-to-moderate arm paresis (level of evidence 1b, quality of evidence moderate, B+).
Repetitive task training is a therapeutic option when improvement of arm activities is the therapeutic goal (level of evidence 1a, quality of evidence moderate, 0).
Strengthening exercises can be an element of individualized therapy (level of evidence 1a, quality of evidence moderate, 0).
Purpose-built virtual reality therapy systems (and gaming consoles) can be used to improve selective motion capacity (level of evidence 1a, quality of evidence low, 0).
For patients with moderate arm paresis showing compensatory trunk displacement during reaching, using a trunk restraint while training reaching movements can be considered (level of evidence 1a, quality of evidence low, 0).
Daily mirror therapy as additional training for several weeks, e.g. as supervised self-training, should be considered when motor improvement at impairment and/or activity level is intended (level of evidence 1a, quality of evidence moderate, B+); patients with neuropathic pain or neglect might have an additional benefit.
Mental practice is an alternative option after actual motor training sessions with the paretic arm (level of evidence 1a, quality of evidence moderate, 0).
Contralesional LF rTMS or ipsilesional HF rTMS or iTBS of the primary hand motor cortex (e.g. five sessions) can be considered as adjunct therapy by experienced personnel when available and when used within safety guidelines, preferably early after stroke (level of evidence 1a, quality of evidence moderate, 0 [resource implications]).
Patients showing learnt non-use while having only mild-to-moderate paresis with some preserved hand function and no major problems with pain or spasticity in their affected arm should be offered mCIMT or CIMT (the latter when beyond the acute phase) when the actual amount of use of the affected arm is the therapeutic target, i.e. to reverse learnt non-use (level of evidence 1a, quality of evidence high, B+); wearing a restraint of the non-affected arm outside therapeutic sessions is recommended when a “transfer package” is offered during therapy (level of evidence 1b, quality of evidence moderate, B+).
5.3.3 Therapeutic Options Independent of Stage of Disease or Severity of Paresis
For Bobath therapy (neurodevelopmental therapy, NDT), no recommendation can be given (level of evidence 1a, quality of evidence moderate).
The use of tDCS (a-tDCS, c-tDCS, dual-tDCS) to improve upper limb motor function is discouraged outside study protocols (level of evidence 1a, quality of evidence low, B−). The same holds true for rPMS (level of evidence 1a, quality of evidence low, B−).
References
Almhdawi KA, Mathiowetz VG, White M, delMas RC (2016) Efficacy of occupational therapy task-oriented approach in upper extremity post-stroke rehabilitation. Occup Ther Int 23:444–456
Andringa A, van de Port I, van Wegen E, Ket J, Meskers C, Kwakkel G (2019) Effectiveness of Botulinum toxin treatment for upper limb spasticity Poststroke over different ICF domains: a systematic review and meta-analysis. Arch Phys Med Rehabil 100:1703–1725
Appel C, Perry L, Jones F (2014) Shoulder strapping for stroke-related upper limb dysfunction and shoulder impairments: systematic review. NeuroRehabilitation 35:191–204
Arya KN, Verma R, Garg RK, Sharma VP, Agarwal M, Aggarwal GG (2012) Meaningful task-specific training (MTST) for stroke rehabilitation: a randomized controlled trial. Top Stroke Rehabil 19:193–211
Arya KN, Pandian S, Kumar D, Puri V (2015) Task-based Mirror therapy augmenting motor recovery in poststroke hemiparesis: a randomized controlled trial. J Stroke Cerebrovasc Dis 24:1738
Ashworth B (1964) Preliminary trial of carisoprodol in multiple sclerosis. Practitioner 192:540–542
Barclay-Goddard RE, Stevenson TJ, Poluha W, Thalman L (2011) Mental practice for treating upper extremity deficits in individuals with hemiparesis after stroke. Cochrane Database Syst Rev (5):CD005950
Blennerhassett J, Dite W (2004) Additional task-related practice improves mobility and upper limb function early after stroke: a randomised controlled trial. Aust J Physiother 50(4):219–224
Bohannon RW (1997) Reference values for extremity muscle strength obtained by hand-held dynamometry from adults aged 20 to 79 years. Arch Phys Med Rehabil 78(1):26–32. https://doi.org/10.1016/s0003-9993(97)90005-8
Bohannon RW, Smith MB (1987) Inter-rater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther 67:206–207
Bürge E, Kupper D, Finckh A, Ryerson S, Schnider A, Leemann B (2008) Neutral functional realignment orthosis prevents hand pain in patients with subacute stroke: a randomized trial. Arch Phys Med Rehabil 89:1857–1862
Cai Y, Zhang CS, Liu S, Wen Z, Zhang AL, Guo X, Lu C, Xue CC (2017) Electroacupuncture for poststroke spasticity: a systematic review and meta-analysis. Arch Phys Med Rehabil 98:2578–2589
Cauraugh JH, Naik SK, Lodha N, Coombes SA, Summers JJ (2011) Long-term rehabilitation for chronic stroke arm movements: a randomized controlled trial. Clin Rehabil 25:1086–1096
Chen JC, Liang CC, Shaw FZ (2005) Facilitation of sensory and motor recovery by thermal intervention for the hemiplegic upper limb in acute stroke patients: a single-blind randomized clinical trial. Stroke 36:2665–2669
Corbetta D, Sirtori V, Castellini G, Moja L, Gatti R (2015) Constraint-induced movement therapy for upper extremities in people with stroke. Cochrane Database Syst Rev (10):CD004433
Corti M, McGuirk TE, Wu SS, Patten C (2012) Differential effects of power training versus functional task practice on compensation and restoration of arm function after stroke. Neurorehabil Neural Repair 26:842–854
Demeurisse G, Demol O, Robaye E (1980) Motor evaluation in vascular hemiplegia. Eur Neurol 19:382–389
Desrosiers J, Hebert R, Dutil E, Bravo G (1993) Development and reliability of an upper extremity function test for the elderly: the TEMPA. Can J Occup Ther 60:9–16
Desrosiers J, Bravo G, Herbert R, Dutil E, Mercier L (1994) Validation of the box and block test as a measure of dexterity of elderly people: reliability, validity, and norms studies. Arch Phys Med Rehabil 75:751–755
Desrosiers J, Malouin F, Bourbonnais D, Richards CL, Rochette A, Bravo G (2003) Arm and leg impairments and disabilities after stroke rehabilitation: relation to handicap. Clin Rehabil 17:666–673
Elsner B, Kugler J, Pohl M, Mehrholz J (2016) Transcranial direct current stimulation (tDCS) for improving activities of daily living, and physical and cognitive functioning, in people after stroke. Cochrane Database Syst Rev (3):CD009645
Elsner B, Kwakkel G, Kugler J, Mehrholz J (2017) Transcranial direct current stimulation (tDCS) for improving capacity in activities and arm function after stroke: a network meta-analysis of randomised controlled trials. J Neuroeng Rehabil 14:95
Feigin VL, Lawes CM, Bennett DA, Anderson CS (2003) Stroke epidemiology: a review of population-based studies of incidence, prevalence, and case-fatality in the late 20th century. Lancet Neurol 2:43–53
Foley N, Pereira S, Salter K, Fernandez MM, Speechley M, Sequeira K, Miller T, Teasell R (2013) Treatment with Botulinum toxin improves upper-extremity function post stroke: a systematic review and meta-analysis. Arch Phys Med Rehabil 94:977–989
French B, Thomas LH, Coupe J, McMahon NE, Connell L, Harrison J, Sutton CJ, Tishkovskaya S, Watkins CL (2016) Repetitive task training for improving functional ability after stroke. Cochrane Database Syst Rev (11):CD006073
Fugl-Meyer AR, Jaasko L, Leyman IL, Olsson S, Steglind S (1975) The post-stroke hemiplegic patient. I A method for evaluation of physical performance. Scand J Rehabil Med 7:13–31
Han C, Wang Q, Pp M, Mz Q (2013) Effects of intensity of arm training on hemiplegic upper extremity motor recovery in stroke patients: a randomized controlled trial. Clin Rehabil 27:75–81
Harris JE, Eng JJ, Miller WC, Dawson AS (2009) A self-administered Graded Repetitive Arm Supplementary Program (GRASP) improves arm function during inpatient stroke rehabilitation: a multi-site randomized controlled trial. Stroke 40(6):2123–2128
Harris JEH, Eng JJ (2010) Strength training improves upper-limb function in individuals with stroke. a meta-analysis. Stroke 41:136–140
Hunter SM, Hammett L, Ball S, Smith N, Anderson C, Clark A et al (2011) Dose-response study of mobilisation and tactile stimulation therapy for the upper extremity early after stroke: a phase I trial. Neurorehabil Neural Repair 25(4):314–322
Israely S, Carmeli E (2016) Error augmentation as a possible technique for improving upper extremity motor performance after a stroke—a systematic review. Top Stroke Rehabil 23:116–125
Kellor M, Frost J, Silberberg N (1971) Hand strength and dexterity. Am J Occup Ther 25:77–83
Kiper P, Agostini M, Luque-Moreno C, Tonin P, Turolla A (2014) Reinforced feedback in virtual environment for rehabilitation of upper extremity dysfunction after stroke: preliminary data from a randomized controlled trial. Biomed Res Int 2014:752128
Kwah LK, Herbert RD (2016) Prediction of walking and arm recovery after stroke: a critical review. Brain Sci 6:4
Kwakkel G, Veerbeek JM, van Wegen EEH, Wolf SL (2015) Constraint-induced movement therapy after stroke. Lancet Neurol 14:224–234
Kwakkel G, Wagenaar RC, Twisk JW, Lankhorst GJ, Koetsier JC (1999) Intensity of leg and arm training after primary middle-cerebral-artery stroke: a randomised trial. Lancet 354:191–196
Langhorne P, Coupar F, Pollock A (2009) Motor recovery after stroke: a systematic review. Lancet Neurol 8:741–754
Laver KE, Schoene D, Crotty M, George S, Lannin NA, Sherrington C (2013) Telerehabilitation services for stroke. Cochrane Database Syst Rev (12):CD010255
Laver KE, Lange B, George S, Deutsch JE, Saposnik G, Crotty M (2017) Virtual reality for stroke rehabilitation. Cochrane Database Syst Rev 11:CD008349
Levin MF, Kleim JA, Wolf SL (2009) What do motor “recovery” and “compensation” mean in patients following stroke? Neurorehabil Neural Repair 23:313–319
Lincoln NB, Leadbetter D (1979) Assessment of motor function in stroke function. Physiotherapy 65:48–51
Lyle RC (1981) A performance test for assessment of upper limb function in physical rehabilitation treatment and research. Int J Rehabil Res 4:483–492
Magee WL, Clark I, Tamplin J, Bradt J (2017) Music interventions for acquired brain injury. Cochrane Database Syst Rev 1(1):CD006787
Medical Research Council (1975) Aids to the investigation of peripheral nerve injuries. Medical Research Council, HMSO, London
Mehrholz J, Pohl M, Platz T, Kugler J, Elsner B (2018) Electromechanical and robot-assisted arm training for improving activities of daily living, arm function, and arm muscle strength after stroke. Cochrane Database Syst Rev 9(9):CD006876
Mercier L, Audet T, Herbert R, Rochette A, Dubois MF (2001) Impact of motor, cognitive, and perceptual disorders on the ability to perform activities of daily living after stroke. Stroke 32:2602–2608
Mihara M, Hattori N, Hatakenaka M, Yagura H, Kawano T, Hino T, Miyai I (2013) Near-infrared spectroscopy-mediated Neurofeedback enhances efficacy of motor imagery-based training in Poststroke victims: a pilot study. Stroke 44:1091–1098
Momosaki R, Yamada N, Ota E, Abo M (2017) Repetitive peripheral magnetic stimulation for activities of daily living and functional ability in people after stroke. Cochrane Database Syst Rev 6(6):CD011968
Nakayama H, Jorgensen HS, Raaschou HO, Olsen TS (1994) Recovery of upper extremity function in stroke patients: the Copenhagen study. Arch Phys Med Rehabil 75:394–398
Nijland R, Kwakkel G, Bakers J, van Wegen E (2011) Constraint-induced movement therapy for the upper paretic limb in acute or sub-acute stroke: a systematic review. Int J Stroke 6:425–433
Pang MY, Harris JE, Eng JJ (2006) A community-based upper-extremity group exercise program improves motor function and performance of functional activities in chronic stroke: a randomized controlled trial. Arch Phys Med Rehabil 87(1):1–9
Platz T (2017) Practice guidelines in neurorehabilitation. Neurol Int Open 1:E148–E152
Platz T, Winter T, Müller N, Pinkowski C, Eickhof C, Mauritz K-H (2001) Arm ability training for stroke and traumatic brain injury patients with mild arm paresis. A single-blind, randomized, controlled trial. Arch Phys Med Rehabil 82:961–968
Platz T, Eickhof C, van Kaick S, Engel U, Pinkowski C, Kalok S, Pause M (2005) Impairment-oriented training or Bobath therapy for arm paresis after stroke: a single blind, multi-centre randomized controlled trial. Clin Rehabil 19:714–724
Platz T, Vuadens P, Eickhof C, Arnold P, Van Kaick S, Heise K (2008) REPAS, a summary rating scale for resistance to passive movement: item selection, reliability and validity. Disabil Rehabil 30:44–53
Platz T, van Kaick S, Mehrholz J, Leidner O, Eickhof C, Pohl M (2009) Best conventional therapy versus modular impairment-oriented training (IOT) for arm paresis after stroke: a single blind, multi-Centre randomized controlled trial. Neurorehabil Neural Repair 23:706–716
Platz T, Elsner B, Mehrholz J (2015) Arm basis training and arm ability training: two impairment-oriented exercise training techniques for improving arm function after stroke (protocol). Cochrane Database Syst Rev (9):CD011854
Pollock A, Farmer SE, Brady MC, Langhorne P, Mead GE, Mehrholz J et al (2014) Interventions for improving upper limb function after stroke. Cochrane Database Syst Rev Issue 11. Art.No.: CD010820
Pollock A, St George B, Fenton M, Firkins L (2014a) Top 10 research priorities relating to life after stroke—consensus from stroke survivors, caregivers, and health professionals. Int J Stroke 9:313–320
Pollock A, Farmer SE, Brady MC, Langhorne P, Mead GE, Mehrholz J, van Wijck F (2014b) Interventions for improving upper limb function after stroke. Cochrane Database Syst Rev 2014(11):CD010820
Pomeroy VM, Hunter SM, Johansen-Berg H, Ward NS, Kennedy N, Chandler E, Weir CJ, Rothwell J, Wing A, Grey M, Barton G, Leavey N (2018) Functional strength training versus movement performance therapy for upper limb motor recovery early after stroke: a RCT. NIHR Journals Library, Southampton
Prange GB, Kottink AI, Buurke JH, Eckhardt MM, van Keulen-Rouweler BJ, Ribbers GM, Rietman JS (2015) The effect of arm support combined with rehabilitation games on upper-extremity function in subacute stroke: a randomized controlled trial. Neurorehabil Neural Repair 29:174–182
Ramos-Murguialday A, Broetz D, Rea M, Läer L, Yilmaz Ö, Brasil FL, Liberati G, Curado MR, Garcia-Cossio E, Vyziotis A, Cho W, Agostini M, Soares E, Soekadar S, Caria A, Cohen LG, Birbaumer N (2013) Brain-machine Interface in chronic stroke rehabilitation: a controlled study. Ann Neurol 74:100–108
Rossi S, Hallett M, Rossini PM, Pascual-Leone A (2009) Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol 120:2008–2039
Schünemann H, Brożek J, Guyatt G, Oxman A, editors (2013) GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group. Available from http://www.guidelinedevelopment.org/handbook
Sehatzadeh S (2015) Effect of Increased Intensity of Physiotherapy on Patient Outcomes After Stroke: An Evidence-Based Analysis. Ont Health Technol Assess Ser 15(6):1–42
Shindo K, Fujiwara T, Hara J, Oba H, Hotta F, Tsuji T, Hase K, Liu M (2011) Effectiveness of hybrid assistive neuromuscular dynamic stimulation therapy in patients with subacute stroke: a randomized controlled pilot trial. Neurorehabil Neural Repair 25:830–837
Stinear CM, Petoe MA, Anwar S, Barber PA, Byblow WD (2014) Bilateral priming accelerates recovery of upper limb function after stroke: a randomized controlled trial. Stroke 45:205–210
Stinear CM, Byblow WD, Ackerley SJ, Barber PA, Smith MC (2017) Predicting recovery potential for individual stroke patients increases rehabilitation efficiency. Stroke 48:1011–1019
Taub E, Miller NE, Novack TA, Cook EW, Fleming WC, Nepomuceno CS et al (1993) Technique to improve chronic motor deficit after stroke. Arch Phys Med Rehabil 74:347–354
Taub E, Uswatte G, Mark VW, Morris DM, Barman J, Bowman MH, Bryson C, Delgado A, Bishop-McKary S (2013) Method for enhancing real-world use of a more affected arm in chronic stroke: transfer package of constraint-induced movement therapy. Stroke 44:1383–1388
Thieme H, Morkisch N, Mehrholz J, Pohl M, Behrens J, Borgetto B, Dohle C (2018) Mirror therapy for improving motor function after stroke. Cochrane Database Syst Rev 7(7):CD008449
Timmermans AA, Lemmens RJ, Monfrance M, Geers RP, Bakx W, Smeets RJ et al (2014) Effects of task-oriented robot training on arm function, activity, and quality of life in chronic stroke patients: a randomized controlled trial. J Neuroeng Rehabil 11:45
Tyson S, Wilkinson J, Thomas N, Selles R, McCabe C, Tyrrell P et al (2015) Phase II pragmatic randomized controlled trial of patient-led therapies (mirror therapy and lower-limb exercises) during inpatient stroke rehabilitation. Neurorehabil Neural Repair 29:818–826
Vafadar AK, Côté JN, Archambault PS (2015) Effectiveness of functional electrical stimulation in improving clinical outcomes in the upper arm following stroke: a systematic review and meta-analysis. Biomed Res Int 2015:729768
Veerbeek JM, van Wegen E, van Peppen R, van der Wees PJ, Hendriks E et al (2014) What is the evidence for physical therapy poststroke? A systematic review and meta-analysis. PLoS One 9(2):e87987
Veerbeek JM, Langbroek-Amersfoort AC, van Wegen EE, Meskers CG, Kwakkel G (2017) Effects of robot-assisted therapy for the upper limb after stroke. Neurorehabil Neural Repair 31:107–121
Vloothuis JD, Mulder M, Veerbeek JM, Konijnenbelt M, Visser-Meily JM, Ket JC, Kwakkel G, van Wegen EE (2016) Caregiver-mediated exercises for improving outcomes after stroke. Cochrane Database Syst Rev 12(12):CD011058
Wang CP, Tsai PY, Yang TF, Yang KY, Wang CC (2014) Differential effect of conditioning sequences in coupling inhibitory/facilitatory repetitive transcranial magnetic stimulation for poststroke motor recovery. CNS. Neurosci Ther 20:355–363
Ward NS, Cohen LG (2004) Mechanisms underlying recovery of motor function after stroke. Arch Neurol 61:1844–1848
Wee SK, Hughes AM, Warner M, Burridge JH (2014) Trunk restraint to promote upper extremity recovery in stroke patients: a systematic review and meta-analysis. Neurorehabil Neural Repair 28:660–677
Winstein CJ, Rose DK, Tan SM, Lewthwaite R, Chui HC, Azen SP (2004) A randomized controlled comparison of upper-extremity rehabilitation strategies in acute stroke: A pilot study of immediate and long-term outcomes. Arch Phys Med Rehabil 85(4):620–628
Wolf SL, Catlin PA, Ellis M, Archer AL, Morgan B, Piacentino A (2001) Assessing Wolf Motor function test as outcome measure for research in patients after stroke. Stroke 32:1635–1639
Wolf SL, Sahu K, Bay RC, Buchanan S, Reiss A, Linder S et al (2015) The HAAPI (home arm assistance progression initiative) trial: a novel robotics delivery approach in stroke rehabilitation. Neurorehabil Neural Repair 29:958–968
Wu HC, Lin YC, Hsu MJ, Liu SH, Hsieh CL, Lin JH (2010) Effect of thermal stimulation on upper extremity motor recovery 3 months after stroke. Stroke 41:2378–2380
Xiang H, Sun J, Tang X, Zeng K, Wu X (2019) The effect and optimal parameters of repetitive transcranial magnetic stimulation on motor recovery in stroke patients: a systematic review and meta-analysis of randomized controlled trials. Clin Rehabil 33:847–864
Yoo GE, Kim SJ (2016) Rhythmic auditory cueing in motor rehabilitation for stroke patients: systematic review and meta-analysis. J Music Ther 53:149–177
Zhang L, Xing G, Fan Y, Guo Z, Chen H, Mu Q (2017) Short- and long-term effects of repetitive transcranial magnetic stimulation on upper limb motor function after stroke: a systematic review and meta-analysis. Clin Rehabil 31:1137–1153
Zhuang LX, Xu SF, D’Adamo CR, Jia C, He J, Han DX, Lao LX (2012) An effectiveness study comparing acupuncture, physiotherapy, and their combination in poststroke rehabilitation: a multicentered, randomized, controlled clinical trial. Altern Ther Health Med 18:8–14
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits any noncommercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if you modified the licensed material. You do not have permission under this license to share adapted material derived from this chapter or parts of it.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this chapter
Cite this chapter
Platz, T., Schmuck, L., Roschka, S., Burridge, J. (2021). Arm Rehabilitation. In: Platz, T. (eds) Clinical Pathways in Stroke Rehabilitation. Springer, Cham. https://doi.org/10.1007/978-3-030-58505-1_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-58505-1_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-58504-4
Online ISBN: 978-3-030-58505-1
eBook Packages: MedicineMedicine (R0)