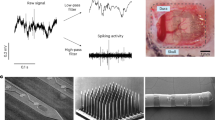
Abstract
The possibility to substitute or extend the brain capabilities has always fascinated the scientific community and, currently, commercial and experimental efforts are proposing cutting-edge prototypal solutions. In a typical scenario, the brain is seen as a complex mechanistic machinery able to execute functions and to process information. These functions are virtually transferred to exogenous actors such as artificial devices that consider a variety of apparatuses variably applicable in complex neuropathological contexts. The applications can vary from functional substitution of lesioned brain regions, to dynamic supplementarity, to support “weakened” functions, to functional enhancement, or even (in a not far future) operative additionality of natural properties.
However, the interface between the biological substrate and their artificial complement represents one of the most crucial elements in the plot. Indeed, these “junction points” reflect the most struggling limitations that, so far, have prevented from a full development in the field. On one hand, best brain-machine-interfaces (BMIs) are the result of a physical communication between the nervous system and the artificial device. Current available solutions are mostly invasive and include, for instance, subdural implanted microelectrode matrices or chronic macroelectrode stereotactic positioning into due brain targets. On the other hand, noninvasive BMIs, such as transcranical magnetic/current stimulations, propose surgical-free activations that however do not match the best requirements both for gross spatial resolution and because of the unreliable conveyed information. Invasive BMI, though partially supervening those problems, typically suffer, however, from other issues preventing their efficient long-term effectiveness. In fact, glial immunoreactions provoke, for instance, glial probe coatings degrading the electrodynamical features. In this review, we discuss the problems related to the diverse applications of BMI; a repertoire of recent cutting-edge insights in the BMI world will be analyzed, however, in a critical key throughout neurophysiological and neurobiological perspectives.
“Que sçay-je?” (ancient French—What do I know?)
Michel de Montaigne
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Andersen RA, Hwang EJ, Mulliken GH (2010) Cognitive neural prosthetics. Annu Rev Psychol 61(1):169–190. https://doi.org/10.1146/annurev.psych.093008.100503
Bair W, Koch C (1996) Temporal precision of spike trains in extrastriate cortex of the behaving macaque monkey. Neural Comput 8(6):1185–1202. https://doi.org/10.1162/neco.1996.8.6.1185
Bastos AM, Vezoli J, Bosman CA, Schoffelen J-M, Oostenveld R, Dowdall JR, De Weerd P, Kennedy H, Fries P (2015) Visual areas exert feedforward and feedback influences through distinct frequency channels. Neuron 85(2):390–401. https://doi.org/10.1016/j.neuron.2014.12.018
Benabid AL, Costecalde T, Eliseyev A, Charvet G, Verney A, Karakas S, Foerster M et al (2019) An exoskeleton controlled by an epidural wireless brain-machine Interface in a Tetraplegic patient: a proof-of-concept demonstration. Lancet Neurol 18(12):1112–1122. https://doi.org/10.1016/S1474-4422(19)30321-7
Bialek W, de Ruyter Van Steveninck RR, Rieke F, Warland D (1999) Spikes: exploring the neural code. MIT Press, Cambridge, MA
Biella G, Sotgiu ML (1995) Evidence that inhibitory mechanisms mask inappropriate Somatotopic connections in the spinal cord of Normal rat. J Neurophysiol 74(2):495–505
Boettiger CA, D’Esposito M (2005) Frontal networks for learning and executing arbitrary stimulus-response associations. J Neurosci 25(10):2723–2732. https://doi.org/10.1523/JNEUROSCI.3697-04.2005
Britten KH, Shadlen MN, Newsome WT, Movshon JA (1992) The analysis of visual motion: a comparison of neuronal and psychophysical performance. J Neurosci 12(12):4745–4765. https://doi.org/10.1523/JNEUROSCI.12-12-04745.1992
Buch VP, Richardson AG, Brandon C, Stiso J, Khattak MN, Bassett DS, Lucas TH (2018) Network brain-computer Interface (NBCI): an alternative approach for cognitive prosthetics. Front Neurosci 12(November):790. https://doi.org/10.3389/fnins.2018.00790
Bundy DT, Szrama N, Pahwa M, Leuthardt EC (2018) Unilateral, 3D arm movement kinematics are encoded in Ipsilateral human cortex. J Neurosci 38(47):10042–10056. https://doi.org/10.1523/JNEUROSCI.0015-18.2018
Canolty RT, Ganguly K, Kennerley SW, Cadieu CF, Koepsell K, Wallis JD, Carmena JM (2010) Oscillatory phase coupling coordinates anatomically dispersed functional cell assemblies. Proc Natl Acad Sci U S A 107(40):17356–17361. https://doi.org/10.1073/pnas.1008306107
Canolty RT, Ganguly K, Carmena JM (2012) Task-dependent changes in cross-level coupling between single neurons and oscillatory activity in multiscale networks. PLoS Comput Biol 8(12):e1002809. https://doi.org/10.1371/journal.pcbi.1002809
Carmena JM (2013) Advances in Neuroprosthetic learning and control. PLoS Biol 11(5):1–4. https://doi.org/10.1371/journal.pbio.1001561
Chiang C-C, Shivacharan RS, Wei X, Gonzalez-Reyes LE, Durand DM (2019) Slow periodic activity in the longitudinal hippocampal slice can self-propagate non-synaptically by a mechanism consistent with ephaptic coupling. J Physiol 597(1):249–269. https://doi.org/10.1113/JP276904
del Ama AJ, Koutsou AD, Moreno JC, de-los-Reyes A, Gil-Agudo A, Pons JL (2012) Review of hybrid exoskeletons to restore gait following spinal cord injury. J Rehab Res Dev 49(4):497. https://doi.org/10.1682/JRRD.2011.03.0043
El Hady A, Machta BB (2015) Mechanical surface waves accompany action potential propagation. Nat Commun 6(1):6697. https://doi.org/10.1038/ncomms7697
Engelbrecht J, Peets T, Tamm K, Laasmaa M, Vendelin M (2018) On the complexity of signal propagation in nerve fibres. Proc Estonian Acad Sci 67(1):28. https://doi.org/10.3176/proc.2017.4.28
Faber DS, Pereda AE (2018) Two forms of electrical transmission between neurons. Front Mol Neurosci 11:427. https://doi.org/10.3389/fnmol.2018.00427
Fisahn C, Aach M, Jansen O, Moisi M, Mayadev A, Pagarigan KT, Dettori JR, Schildhauer TA (2016) The effectiveness and safety of exoskeletons as assistive and rehabilitation devices in the treatment of neurologic gait disorders in patients with spinal cord injury: a systematic review. Global Spine J 6(8):822–841. https://doi.org/10.1055/s-0036-1593805
Fultz NE, Bonmassar G, Setsompop K, Stickgold RA, Rosen BR, Polimeni JR, Lewis LD (2019) Coupled electrophysiological, hemodynamic, and cerebrospinal fluid oscillations in human sleep. Science 366(6465):628–631. https://doi.org/10.1126/science.aax5440
Ghazanfar AA, Stambaugh CR, Nicolelis MA (2000) Encoding of tactile stimulus location by somatosensory thalamocortical ensembles. J Neurosci 20(10):3761–3775. https://doi.org/10.1523/JNEUROSCI.20-10-03761.2000
Grol MJ, de Lange FP, Verstraten FAJ, Passingham RE, Toni I (2006) Cerebral changes during performance of overlearned arbitrary visuomotor associations. J Neurosci 26(1):117–125. https://doi.org/10.1523/JNEUROSCI.2786-05.2006
Herculano-Houzel S (2009) The human brain in numbers: a linearly scaled-up primate brain. Front Hum Neurosci 3(November):31. https://doi.org/10.3389/neuro.09.031.2009
Histed MH, Bonin V, Clay Reid R (2009) Direct activation of sparse, distributed populations of cortical neurons by electrical microstimulation. Neuron 63(4):508–522. https://doi.org/10.1016/J.NEURON.2009.07.016
Houweling AR, Brecht M (2008) Behavioural report of single neuron stimulation in somatosensory cortex. Nature 451(7174):65–68. https://doi.org/10.1038/nature06447
Houweling AR, Doron G, Voigt BC, Herfst LJ, Brecht M (2010) Nanostimulation: manipulation of single neuron activity by Juxtacellular current injection. J Neurophysiol 103(3):1696–1704. https://doi.org/10.1152/jn.00421.2009
Irwin ZT, Schroeder KE, Vu PP, Bullard AJ, Tat DM, Nu CS, Vaskov A et al (2017) Neural control of finger movement via Intracortical brain–machine Interface. J Neural Eng 14(6):066004. https://doi.org/10.1088/1741-2552/aa80bd
Jansen O, Grasmuecke D, Meindl RC, Tegenthoff M, Schwenkreis P, Sczesny-Kaiser M, Wessling M, Schildhauer TA, Fisahn C, Aach M (2018) Hybrid assistive limb exoskeleton HAL in the rehabilitation of chronic spinal cord injury: proof of concept; the results in 21 patients. World Neurosurg 110(February):e73–e78. https://doi.org/10.1016/j.wneu.2017.10.080
Jin Y, Chen J, Zhang S, Chen W, Zheng X (2019) Invasive brain machine interface system. In: Neural interface: frontiers and applications. Springer, Singapore, pp 67–89. https://doi.org/10.1007/978-981-13-2050-7_3
Laiwalla F, Nurmikko A (2019) Future of neural interfaces. Adv Exp Med Biol 1101:225–241. https://doi.org/10.1007/978-981-13-2050-7_9
Li Z, O’Doherty JE, Hanson TL, Lebedev MA, Henriquez CS, Nicolelis MAL (2009) Unscented Kalman Filter for brain-machine interfaces. PLoS One 4(7):e6243. https://doi.org/10.1371/journal.pone.0006243
Li C, Jia T, Xu Q, Ji L, Yu P (2019) Brain-computer interface channel-selection strategy based on analysis of event-related Desynchronization topography in stroke patients. J Healthc Eng 2019(August):1–12. https://doi.org/10.1155/2019/3817124
Mirabella G, Pani P, Ferraina S (2008) Context influences on the preparation and execution of reaching movements. Cogn Neuropsychol 25(7–8):996–1010. https://doi.org/10.1080/02643290802003216
Mirabella G, Pani P, Ferraina S (2011) Neural correlates of cognitive control of reaching movements in the dorsal premotor cortex of rhesus monkeys. J Neurophysiol 106(3):1454–1466. https://doi.org/10.1152/jn.00995.2010
Mohan H, de Haan R, Mansvelder HD, de Kock CPJ (2018) The posterior parietal cortex as integrative hub for whisker sensorimotor information. Neuroscience 368(January):240–245. https://doi.org/10.1016/j.neuroscience.2017.06.020
Nicolelis MA, Chapin JK (1994) Spatiotemporal structure of somatosensory responses of many-neuron ensembles in the rat ventral posterior medial nucleus of the thalamus. J Neurosci 14(6):3511–3532. https://doi.org/10.1523/JNEUROSCI.14-06-03511.1994
Nixon P, Lazarova J, Hodinott-Hill I, Gough P, Passingham R (2004) The inferior frontal Gyrus and phonological processing: an investigation using RTMS. J Cogn Neurosci 16(2):289–300. https://doi.org/10.1162/089892904322984571
Olshausen BA, Field DJ (2004) Sparse coding of sensory inputs. Curr Opin Neurobiol 14(4):481–487. https://doi.org/10.1016/J.CONB.2004.07.007
Pasupathy A, Miller EK (2005) Different time courses of learning-related activity in the prefrontal cortex and striatum. Nature 433(7028):873–876. https://doi.org/10.1038/nature03287
Pesaran B, Musallam S, Andersen RA (2006) Cognitive neural prosthetics. Curr Biol 16(3):R77–R80. https://doi.org/10.1016/j.cub.2006.01.043
Petrides M (1982) Motor conditional associative-learning after selective prefrontal lesions in the monkey. Behav Brain Res 5(4):407–413. https://doi.org/10.1016/0166-4328(82)90044-4
Ramakrishnan A, Ifft PJ, Pais-Vieira M, Byun YW, Zhuang KZ, Lebedev MA, Nicolelis MAL (2015) Computing arm movements with a monkey Brainet. Sci Rep 5(1):10767. https://doi.org/10.1038/srep10767
Reyes-Puerta V, Kim S, Sun J-J, Imbrosci B, Kilb W, Luhmann HJ (2015) High stimulus-related information in barrel cortex inhibitory interneurons. PLoS Comput Biol 11(6):e1004121. https://doi.org/10.1371/journal.pcbi.1004121
Ruyter Van Steveninck RR d, Bialek W, Potters M, Carlson RH (1994) Statistical adaptation and optimal estimation in movement computation by the blowfly visual system. In: Proceedings of IEEE International Conference on systems, man and cybernetics, vol 1. IEEE, Piscataway, NJ. Accessed 6 Dec 2019, pp 302–307. https://doi.org/10.1109/ICSMC.1994.399855
Sahin M, Pikov V (2011) Wireless microstimulators for neural prosthetics. Crit Rev Biomed Eng 39(1):63–77. https://doi.org/10.1615/critrevbiomedeng.v39.i1.50
Salzman CD, Britten KH, Newsome WT (1990) Cortical microstimulation influences perceptual judgements of motion direction. Nature 346(6280):174–177. https://doi.org/10.1038/346174a0
Sotgiu ML, Biella G, Riva L (1995) Poststimulus afterdischarges of spinal WDR and NS units in rats with chronic nerve constriction. Neuroreport 9(6):1021–1024
Stoet G, Snyder LH (2004) Single neurons in posterior parietal cortex of monkeys encode cognitive set. Neuron 42(6):1003–1012. https://doi.org/10.1016/J.NEURON.2004.06.003
Talakoub O, Marquez-Chin C, Popovic MR, Navarro J, Fonoff ET, Hamani C, Wong W (2017) Reconstruction of reaching movement trajectories using Electrocorticographic signals in humans. PLoS One 12(9):e0182542. https://doi.org/10.1371/journal.pone.0182542
Tao L, Porto D, Li Z, Fechner S, Lee SA, Goodman MB, Shawn Xu XZ, Lu H, Shen K (2019) Parallel processing of two mechanosensory modalities by a single neuron in C. elegans. Dev Cell 51(5):617–631.e3. https://doi.org/10.1016/J.DEVCEL.2019.10.008
Toni I, Passingham RE (1999) Prefrontal-basal ganglia pathways are involved in the learning of arbitrary visuomotor associations: a PET study. Exp Brain Res 127(1):19–32. https://doi.org/10.1007/s002210050770
van Dijk L, van der Sluis CK, van Dijk HW, Bongers RM (2016) Task-oriented gaming for transfer to prosthesis use. IEEE Trans Neural Syst Rehabil Eng 24(12):1384–1394. https://doi.org/10.1109/TNSRE.2015.2502424
Wagshul ME, Eide PK, Madsen JR (2011) The pulsating brain: a review of experimental and clinical studies of intracranial pulsatility. Fluids Barriers CNS 8(1):5. https://doi.org/10.1186/2045-8118-8-5
Wallis JD, Anderson KC, Miller EK (2001) Single neurons in prefrontal cortex encode abstract rules. Nature 411(6840):953–956. https://doi.org/10.1038/35082081
White IM, Wise SP (1999) Rule-dependent neuronal activity in the prefrontal cortex. Exp Brain Res 126(3):315–335. https://doi.org/10.1007/s002210050740
Wittenburg P (2010) Archiving and accessing language resources. Concurr Comput Pract Exp 22(17):2354–2368. https://doi.org/10.1002/cpe.1605
Zohary E, Shadlen MN, Newsome WT (1994) Correlated neuronal discharge rate and its implications for psychophysical performance. Nature 370(6485):140–143. https://doi.org/10.1038/370140a0
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Zippo, A.G., Biella, G.E.M. (2021). Brain Machine Interfaces Within a Critical Perspective. In: Opris, I., A. Lebedev, M., F. Casanova, M. (eds) Modern Approaches to Augmentation of Brain Function. Contemporary Clinical Neuroscience. Springer, Cham. https://doi.org/10.1007/978-3-030-54564-2_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-54564-2_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-54563-5
Online ISBN: 978-3-030-54564-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)