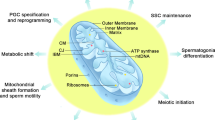
Abstract
Spermatozoa are motile cells specifically designed to transfer the paternal genetic material across a challenging spatial route to an ovum. The importance of sperm motility to fertility has been firmly established. In this chapter, we address the central role that sperm mitochondria play in providing the energy necessary for the generation of motility by summarizing the metabolic pathways for both anaerobic (glycolytic) and aerobic (mitochondrial) generation of ATP. We stress the importance of the glycolytic-metabolic end products being essential metabolic substrates for the mitochondria oxidative phosphorylation, where ATP production is dramatically escalated. The anomaly and duality of mitochondrial and nuclear DNA, and their interplay in ensuring functional oxidative phosphorylation, are discussed by illustrating how single-point or serial mutations have the ability to infer male infertility. In this regard, the balance of redox reactions is therefore crucial in preventing oxidative stress.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Gray MW. Mitochondrial evolution. Cold Spring Harb Perspect Biol. 2012;4:a011403.
Croteau DL, Stierum RH, Bohr VA. Mitochondrial DNA repair pathways. Mutat Res. 1999;434:137–48.
Folgero T, Bertheussen K, Lindal S, Torbergsen T, Oian P. Mitochondrial disease and reduced sperm motility. Hum Reprod. 1993;8:1863–8.
Lodish H, Berk A, Matsudaira P, Kaiser CA, Krieger M, Scott MP, Zipursky L, Darnell J. Molecular cell biology. 6th ed. New York: W.H. Freeman, ©; 2008.
Piomboni P, Focarelli R, Stendardi A, Ferramosca A, Zara V. The role of mitochondria in energy production for human sperm motility. Int J Androl. 2012;35:109–24.
Davila MP, Muñoz PM, Bolaños JMG, Stout TAE, Gadella BM, Tapia JA, da Silva CB, Ferrusola CO, Peña FJ. Mitochondrial ATP is required for the maintenance of membrane integrity in stallion spermatozoa, whereas motility requires both glycolysis and oxidative phosphorylation. Reproduction. 2016;152:683–94.
St John JC, Jokhi RP, Barratt CL. The impact of mitochondrial genetics on male infertility. Int J Androl. 2005;28(2):65–73.
Koppers AJ, De Iuliis GN, Finnie JM, McLaughlin EA, Aitken RJ. Significance of mitochondrial reactive oxygen species in the generation of oxidative stress in spermatozoa. J Clin Endocrinol Metab. 2008;93:3199–207.
Kim KH, Son JM, Benayoun BA, Lee C. The mitochondrial-encoded peptide MOTS-c translocates to the nucleus to regulate nuclear gene expression in response to metabolic stress. Cell Metab. 2018;28(3):516–24.
Sharpley MS, Marciniak C, Eckel-Mahan K, McManus M, Crimi M, Waymire K, Lin CS, Masubuchi S, Friend N, Koike M, et al. Heteroplasmy of mouse mtDNA is genetically unstable and results in altered behavior and cognition. Cell. 2012;151:333–43.
Luo S, Valencia CA, Zhang J, Lee NC, Slone J, Gui B, Wang X, Li Z, Dell S, Brown J, Chen SM, Chien YH, Hwu WL, Fan PC, Wong LI, Atwal PS, Huang T. Biparental inheritance of mitochondrial DNA in humans. Proc Natl Acad Sci U S A. 2018;115(51):13039–44.
Cummings JM, Jequier AM, Kan R. Molecular biology of human male infertility: links with aging, mitochondrial genetics, and oxidative stress? Mol Reprod Dev. 1994;37:345–62.
Lestienne P, Reynier P, Chretien MF, Penisson-Besnier I, Malthiery Y, Rohmer V. Oligoasthenospermia associated with multiple mitochondrial DNA rearrangements. Mol Hum Reprod. 1997;3:811–4.
Rajender S, Rahul P, Mahdi AA. Mitochondria, spermatogenesis and male infertility. Mitochondrion. 2010;10:419–28.
Ferramosca A, Focarelli R, Piomboni P, Coppola L, Zara V. Oxygen uptake by mitochondria in demembranated human sperma- tozoa: a reliable tool for the evaluation of sperm respiratory efficiency. Int J Androl. 2008;31:337–45.
Green DR, Reed JC. Mitochondria and apoptosis. Science. 1998;281:1309–12.
Desagher S, Martinou JC. Mitochondria as the central control point of apoptosis. Trends Cell Biol. 2000;10:369–77.
Leist M, Nicotera P. The shape of cell death. Biochem Biophys Res Commun. 1997;236:1–9.
Kasai T, Ogawa K, Mizuno K, Nagai S, Uchida Y, Ohta S, Fujie M, Suzuki K, Hirata S, Hoshi K. Relationship between sperm mitochondrial membrane potential, sperm motility, and fertility potential. Asian J Androl. 2002;4:97–103.
Gallon F, Marchetti C, Jouy N, Marchetti P. The functionality of mitochondria differentiates human spermatozoa with high and low fertilizing capability. Fertil Steril. 2006;86:1526–30.
Espinoza JA, Schulz MA, Sanchez R, Villegas JV. Integrity of mitochondrial membrane potential reflects human sperm quality. Andrologia. 2009;41:51–4.
Paoli D, Gallo M, Rizzo F, Baldi E, Francavilla S, Lenzi A, Lombardo F, Gandini L. Mitochondrial membrane potential profile and its correlation with increasing sperm motility. Fertil Steril. 2011;95:2315–9.
Marchetti P, Ballot C, Jouy N, Thomas P, Marchetti C. Influence of mitochondrial membrane potential of spermatozoa on in vitro fertilisation outcome. Andrologia. 2012;44:136–41.
Sharbatoghli M, Valojerdi MR, Amanlou M, Khosravi F, Jafar-abadi MA. Relationship of sperm DNA fragmentation, apoptosis and dysfunction of mitochondrial membrane potential with semen parameters and ART outcome after intracytoplasmic sperm injection. Arch Gynecol Obstet. 2012;286:1315–22.
Agnihotri SK, Agrawal AK, Hakim BA, Vishwakarma AL, Narender T, Sachan R, Sachdev M. Mitochondrial membrane potential (MMP) regulates sperm motility. In Vitro Cell Dev Biol Anim. 2016;52:953–60.
Henkel R. ROS and DNA integrity—implications of male accessory gland infections. In: Björndahl L, Giwercman A, Tournaye H, Weidner W, editors. Clinical andrology. London: Informa Healthcare (incorporating Parthenon Publishing, Marcel Dekker, and Taylor & Francis Medical; 2010. p. 324–8.
Zhang WD, Zhang Z, Jia LT, Zhang LL, Fu T, Li YS, Wang P, Sun L, Shi Y, Zhang HZ. Oxygen free radicals and mitochondrial signaling in oligospermia and asthenospermia. Mol Med Rep. 2014;10:1875–80.
Treulen F, Uribe P, Boguen R, Villegas JV. Mitochondrial permeability transition increases reactive oxygen species production and induces DNA fragmentation in human spermatozoa. Hum Reprod. 2015;30:767–76.
Zhang G, Yang W, Zou P, Jiang F, Zeng Y, Chen Q, Sun L, Yang H, Zhou N, Wang X, Liu J, Cao J, Zhou Z, Ao L. Mitochondrial functionality modifies human sperm acrosin activity, acrosome reaction capability and chromatin integrity. Hum Reprod. 2019;34:3–11.
Marchetti C, Obert G, Deffosez A, Formstecher P, Marchetti P. Study of mitochondrial membrane potential, reactive oxygen species, DNA fragmentation and cell viability by flow cytometry in human sperm. Hum Reprod. 2002;17:1257–65.
Reed JC. Mechanisms of apoptosis. Am J Pathol. 2000;157:1415–30.
Green DR. Overview: apoptotic signaling pathways in the immune system. Immunol Rev. 2003;193:5–9.
Koppers AJ, Mitchell LA, Wang P, Lin M, Aitken RJ. Phosphoinositide-3-kinase signaling pathway involvement in a truncated apoptotic cascade associated with motility loss and oxidative DNA damage in human spermatozoa. Biochem J. 2011;436:687–98.
Aitken RJ, Gibb Z, Baker MA, Drevet J, Gharagozloo P. Causes and consequences of oxidative stress in spermatozoa. Reprod Fertil Dev. 2016;28:1–10.
Ohno M, Sakumi K, Fukumura R, Furuichi M, Iwasaki Y, Hokama M, Ikemura T, Tsuzuki T, Gondo Y, Nakabeppu Y. 8-oxoguanine causes spontaneous de novo germline mutations in mice. Sci Rep. 2014;4:4689.
Sotolongo B, Huang TT, Isenberger E, Ward WS. An endogenous nuclease in hamster, mouse, and human spermatozoa cleaves DNA into loop-sized fragments. J Androl. 2005;26:272–80.
Thomson LK, Fleming SD, Aitken RJ, De Iuliis GN, Zieschang JA, Clark AM. Cryopreservation-induced human sperm DNA damage is predominantly mediated by oxidative stress rather than apoptosis. Hum Reprod. 2009;24:2061–70.
Hezavehei M, Sharafi M, Kouchesfahani HM, Henkel R, Agarwal A, Esmaeili V, Shahverdi A. Sperm cryopreservation: a review on current molecular cryobiology and advanced approaches. Reprod Biomed Online. 2018;37:327–39.
Aitken RJ, Curry BJ. Redox regulation of human sperm function: from the physiological control of sperm capacitation to the etiology of infertility and DNA damage in the germ line. Antioxid Redox Signal. 2011;14:367–81.
Agarwal A, Majzoub A. Role of antioxidants in male infertility. BJUI Knowledge. 2016:1–9.
Lavi R, Shainberg A, Shneyvays V, Hochauser E, Isaac A, Zinman T, Friedmann H, Lubart R. Detailed analysis of reactive oxygen species induced by visible light in various cell types. Lasers Surg Med. 2010;42:473–80.
Kesari KK, Agarwal A, Henkel R. Radiations and male fertility. Reprod Biol Endocrinol. 2018;16:118.
Henkel R, Kierspel E, Stalf T, Mehnert C, Menkveld R, Tinneberg HR, Schill WB, Kruger TF. Effect of reactive oxygen species produced by spermatozoa and leukocytes on sperm functions in non-leukocytospermic patients. Fertil Steril. 2005;83:635–42.
Zorn B, Ihan A, Kopitar AN, Kolbezen M, Sesek-Briski A, Meden-Vrtovec H. Changes in sperm apoptotic markers as related to seminal leukocytes and elastase. Reprod Biomed Online. 2010;21:84–92.
Shi TY, Chen G, Huang X, Yuan Y, Wu X, Wu B, Li Z, Shun F, Chen H, Shi H. Effects of reactive oxygen species from activated leucocytes on human sperm motility, viability and morphology. Andrologia. 2012;44(Suppl. 1):696–703.
Aitken RJ, Whiting S, De Iuliis GN, McClymont S, Mitchell LA, Baker MA. Electrophilic aldehydes generated by sperm metabolism activate mitochondrial reactive oxygen species generation and apoptosis by targeting succinate dehydrogenase. J Biol Chem. 2012;287:33048–60.
Platt N, da Silva RP, Gordon S. Recognizing death: the phagocytosis of apoptotic cells. Trends Cell Biol. 1998;8:365–72.
Fawzy F, Hussein A, Eid MM, El Kashash AM, Salem HK. Cryptorchidism and fertility. Clin Med Insights Reprod Health. 2015;9:39–43.
Bedford JM. Human spermatozoa and temperature: the elephant in the room. Biol Reprod. 2015;93(4):97, 1–5.
Rao M, Xia W, Yang J, Hu LX, Hu SF, Lei H, Wu YQ, Zhu CH. Transient scrotal hyperthermia affects human sperm DNA integrity, sperm apoptosis, and sperm protein expression. Andrology. 2016;4(6):1054–63.
Gong Y, Guo H, Zhang Z, Zhou H, Zhao R, He B. Heat stress reduces sperm motility via activation of glycogen synthase kinase-3α and inhibition of mitochondrial protein import. Front Physiol. 2017;8:718, 1–10.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Fisher, D., Henkel, R. (2020). Mitochondrial Function and Male Infertility. In: Arafa, M., Elbardisi, H., Majzoub, A., Agarwal, A. (eds) Genetics of Male Infertility. Springer, Cham. https://doi.org/10.1007/978-3-030-37972-8_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-37972-8_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-37971-1
Online ISBN: 978-3-030-37972-8
eBook Packages: MedicineMedicine (R0)