Abstract
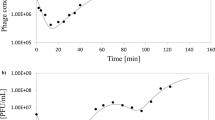
Computational models can be used to optimize the production of bacteriophages. Here a model is described for production in a two-stage self-cycling process. Theoretical and practical considerations for modeling bacteriophage production are first introduced. The key experimental protocols required to estimate key kinetic parameters for the model, including determining variable infection rates as a function of substrate concentration, are described. ppSim is an open-source R-script that can simulate bacteriophage production to optimize productivity or minimize costs. The steps included to run the simulation using the experimentally determined infection parameters are described. An example is also presented, where a level sensor and cycle time are optimized to maximize bacteriophage productivity in two sequential 1-L bioreactors, resulting in a production rate of 4.46 × 1010 bacteriophage particles/hour. The protocols and programs described here will allow users to potentially optimize production of their own bacteriophage–bacteria pairing by effectively applying bacteriophage modeling.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Matsuzaki S et al (2005) Bacteriophage therapy: a revitalized therapy against bacterial infectious diseases. J Infect Chemother 11(5):211–219
Monk AB et al (2010) Bacteriophage applications: where are we now?: Bacteriophage applications. Lett Appl Microbiol 51(4):363–369
Summer, N.S., et al. (2008) Phage remediation of microbe induced corrosion. In 17th international corrosion congress
Withey S et al (2005) Bacteriophages—potential for application in wastewater treatment processes. Sci Total Environ 339(1–3):1–18
Krysiak-Baltyn K et al (2016) Computational models of populations of bacteria and lytic phage. Crit Rev Microbiol 42(6):942–968
Levin BR, Stewart FM, Chao L (1977) Resource-limited growth, competition, and predation: a model and experimental studies with bacteria and bacteriophage. Am Nat 111(977):3–24
Campbell A (1961) Conditions for the existence of bacteriophage. Evolution 15:153–165
Sauvageau D, Cooper DG (2010) Two-stage, self-cycling process for the production of bacteriophages. Microb Cell Factories 9(1):1
Lenski RE, Levin BR (1985) Constraints on the coevolution of bacteria and virulent phage: a model, some experiments, and predictions for natural communities. Am Nat 125:585–602
Sauvageau D (2010) Two-stage, self-cycling process for the production of bacteriophages. McGill University, Montreal, QC
Krysiak-Baltyn K et al (2016) Simulation of phage dynamics in multi-reactor models of complex wastewater treatment systems. Biochem Eng J 122:91–102
Jensen MA et al (2006) Modeling the role of bacteriophage in the control of cholera outbreaks. PNAS 103:4652–4657
Bull JJ et al (2014) Phenotypic resistance and the dynamics of bacterial escape from phage control. PLoS One 9(4):e94690
Bohannan BJM, Lenski RE (1997) Effect of resource enrichment on a chemostat community of bacteria and bacteriophage. Ecology 78(8):2303–2315
Bohannan BJM, Lenski RE (2000) The relative importance of competition and predation varies with productivity in a model community. Am Nat 156(4):329–340
Hadas H et al (1997) Bacteriophage T4 development depends on the physiology of its host Escherichia coli. Microbiology 143(1):179–185
You L, Suthers PF, Yin J (2002) Effects of Escherichia coli physiology on growth of phage T7 in vivo and in silico. J Bacteriol 184(7):1888–1894
Schrag SJ, Mittler JE (1996) Host-parasite coexistence: the role of spatial refuges in stabilizing bacteria-phage interactions. Am Nat 148:348–377
Hyman P, Abedon ST (2009) Practical methods for determining phage growth parameters. In: Clokie MRJ, Kropinski AM (eds) Bacteriophages: methods and protocols, volume 1: isolation, characterization, and interactions. Humana Press, Totowa, NJ, pp 175–202
Kropinski AM (2009) Measurement of the rate of attachment of bacteriophage to cells. In: Clokie MRJ, Kropinski AM (eds) Bacteriophages: methods and protocols, volume 1: isolation, characterization, and interactions. Humana Press, Totowa, NJ, pp 151–155
Khawaldeh A et al (2011) Bacteriophage therapy for refractory Pseudomonas aeruginosa urinary tract infection. J Med Microbiol 60(11):1697–1700
Brussow H (2005) Phage therapy: the Escherichia coli experience. Microbiology 151(7):2133–2140
Acknowledgement
This work was funded by an Australian Research Council Linkage Grant (LP120100304) with input from Melbourne Water, South Australia Water and Water Corporation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Krysiak-Baltyn, K., Martin, G.J.O., Gras, S.L. (2018). Computational Modeling of Bacteriophage Production for Process Optimization. In: Azeredo, J., Sillankorva, S. (eds) Bacteriophage Therapy. Methods in Molecular Biology, vol 1693. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7395-8_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7395-8_16
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7394-1
Online ISBN: 978-1-4939-7395-8
eBook Packages: Springer Protocols