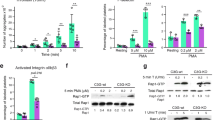
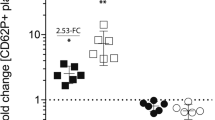
Abstract
Platelets are small blood cells derived from cytoplasmic fragments of megakaryocytes and play an essential role in thrombosis and hemostasis. Platelet activation depends on the rapid phosphorylation and dephosphorylation of key signaling molecules, and a number of kinases and phosphatases have been identified as major regulators of platelet function. However, the investigation of novel signaling proteins has suffered from technical limitations due to the anucleate nature of platelets and their very limited levels of mRNA and de novo protein synthesis. In the past, experimental methods were restricted to the generation of genetically modified mice and the development of specific antibodies. More recently, novel (phospho)proteomic technologies and pharmacological approaches using specific small-molecule inhibitors have added additional capabilities to investigate specific platelet proteins.
In this chapter, we report methods for using genetic and pharmacological approaches to investigate the function of platelet signaling proteins. While the described experiments focus on the role of the dual-specificity phosphatase 3 (DUSP3) in platelet signaling, the presented methods are applicable to any signaling enzyme. Specifically, we describe a testing strategy that includes (1) aggregation and secretion experiments with mouse and human platelets, (2) immunoprecipitation and immunoblot assays to study platelet signaling events, (3) detailed protocols to use selected animal models in order to investigate thrombosis and hemostasis in vivo, and (4) strategies for utilizing pharmacological inhibitors on human platelets.
*These authors are contributed equaly to this work.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Italiano JE, Patel-Hett S, Hartwig JH (2007) Mechanics of proplatelet elaboration. J Thromb Haemost 5:18–23
Junt T, Schulze H, Chen Z, Massberg S, Goerge T, Krueger A, Wagner DD, Graf T, Italiano JE, Shivdasani RA, von Andrian UH (2007) Dynamic visualization of thrombopoiesis within bone marrow. Science 317:1767–1770
Varga-Szabo D, Pleines I, Nieswandt B (2008) Cell adhesion mechanisms in platelets. Arterioscler Thromb Vasc Biol 28:403–413
Offermanns S (2006) Activation of platelet function through G protein-coupled receptors. Circ Res 99:1293–1304
Furie B, Furie BC (2007) In vivo thrombus formation. J Thromb Haemost 5:12–17
Watson SP, Auger JM, McCarty OJT, Pearce AC (2005) GPVI and integrin alphaIIb beta3 signaling in platelets. J Thromb Haemost 3:1752–1762
Karisch R, Fernandez M, Taylor P, Virtanen C, St-Germain JR, Jin LL, Harris IS, Mori J, Mak TW, Senis YA, Östman A, Moran MF, Neel BG (2011) Global proteomic assessment of the classical protein-tyrosine phosphatome and “redoxome”. Cell 146:826–840
Tautz L, Senis YA, Oury C, Rahmouni S (2015) Perspective: Tyrosine phosphatases as novel targets for antiplatelet therapy. Bioorg Med Chem 15:2786–2797
Ming Z, Hu Y, Xiang J, Polewski P, Newman PJ, Newman DK (2011) Lyn and PECAM-1 function as interdependent inhibitors of platelet aggregation. Blood 117:3903–3906
Musumeci L, Kuijpers MJ, Gilio K, Hego A, Théâtre E, Maurissen L, Vandereyken M, Diogo CV, Lecut C, Guilmain W, Bobkova EV, Eble JA, Dahl R, Drion P, Rascon J, Mostofi Y, Yuan H, Sergienko E, Chung TD, Thiry M, Senis Y, Moutschen M, Mustelin T, Lancellott RS (2015) Dual-specificity phosphatase 3 deficiency or inhibition limits platelet activation and arterial thrombosis. Circulation 131:656–668
Gao C, Boylan B, Fang J, Wilcox DA, Newman DK, Newman PJ (2011) Heparin promotes platelet responsiveness by potentiating αIIbβ3-mediated outside-in signaling. Blood 117:4946–4952
Cattaneo M, Cerletti C, Harrison P, Hayward CPM, Kenny D, Nugent D, Nurden P, Rao AK, Schmaier AH, Watson SP, Lussana F, Pugliano MT, Michelson AD (2013) Recommendations for the standardization of light transmission aggregometry: a consensus of the working party from the platelet physiology subcommittee of SSC/ISTH. J Thromb Haemost 11:1183–1189
Born GV, Cross MJ (1963) The aggregation of blood platelets. J Physiol 168:178–195
Born G (1962) Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature 194:927–929
Zhou L, Schmaier AH (2005) Platelet aggregation testing in platelet-rich plasma: description of procedures with the aim to develop standards in the field. Am J Clin Pathol 123:172–183
McEver RP, Bennett EM, Martin MN (1983) Identification of two structurally and functionally distinct sites on human platelet membrane glycoprotein IIb-IIIa using monoclonal antibodies. J Biol Chem 258:5269–5275
Nieuwenhuis HK, van Oosterhout JJ, Rozemuller E, van Iwaarden F, Sixma JJ (1987) Studies with a monoclonal antibody against activated platelets: evidence that a secreted 53,000-molecular weight lysosome-like granule protein is exposed on the surface of activated platelets in the circulation. Blood 70:838–845
Shattil SJ, Hoxie JA, Cunningham M, Brass LF (1985) Changes in the platelet membrane glycoprotein IIb.IIIa complex during platelet activation. J Biol Chem 260:11107–11114
Rahmouni S, Delacroix L, Liu WH, Tautz L (2013) Evaluating effects of tyrosine phosphatase inhibitors on T cell receptor signaling. Methods Mol Biol 1053:241–270
Furie B, Furie BC (2005) Thrombus formation in vivo. J Clin Invest 115:3355–3362
Kurz KD, Main BW, Sandusky GE (1990) Rat model of arterial thrombosis induced by ferric chloride. Thromb Res 60:269–280
DiMinno G, Silver MJ (1983) Mouse antithrombotic assay: a simple method for the evaluation of antithrombotic agents in vivo. Potentiation of antithrombotic activity by ethyl alcohol. J Pharmacol Exp Ther 225:57–60
Tautz L, Critton DA, Grotegut S (2013) Protein tyrosine phosphatases: structure, function, and implication in human disease. Methods Mol Biol 1053:179–221
Chiang TM (1992) Okadaic acid and vanadate inhibit collagen-induced platelet aggregation; the functional relation of phosphatases on platelet aggregation. Thromb Res 67:345–354
Posner BI, Faure R, Burgess JW, Bevan AP, Lachance D, Zhang-Sun G, Fantus IG, Ng JB, Hall DA, Lum BS, Shaver A (1994) Peroxovanadium compounds. A new class of potent phosphotyrosine phosphatase inhibitors which are insulin mimetics. J Biol Chem 269:4596–4604
Wu S, Vossius S, Rahmouni S, Miletic AV, Vang T, Vazquez-Rodriguez J, Cerignoli F, Arimura Y, Williams S, Hayes T, Moutschen M, Vasile S, Pellecchia M, Mustelin T, Tautz L (2009) Multidentate small-molecule inhibitors of Vaccinia H1-related (VHR) phosphatase decrease proliferation of cervix cancer cells. J Med Chem 52:6716–6723
Tautz L, Sergienko EA (2013) High-throughput screening for protein tyrosine phosphatase activity modulators. Methods Mol Biol 1053:223–240
Tautz L, Mustelin T (2007) Strategies for developing protein tyrosine phosphatase inhibitors. Methods 42:250–260
Acknowledgement
This work was supported by the Belgian National Fund for Scientific Research (F.R.S.-FNRS: PDR N° T.0105.13), the University of Liège (Fonds Spéciaux pour la Recherche) to (CO and SR), the National Institutes of Health (Grants 1R21CA132121 and 1R03MH084230 to LT), and the American Heart Association (Innovative Research Grant 14IRG18980075 to LT).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this protocol
Cite this protocol
Rahmouni, S. et al. (2016). Functional Analysis of Protein Tyrosine Phosphatases in Thrombosis and Hemostasis. In: Pulido, R. (eds) Protein Tyrosine Phosphatases. Methods in Molecular Biology, vol 1447. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-3746-2_17
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3746-2_17
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-3744-8
Online ISBN: 978-1-4939-3746-2
eBook Packages: Springer Protocols