Abstract
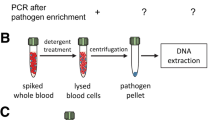
A critical point of molecular diagnosis of systemic infections is the method employed for the extraction of microbial DNA from blood. A DNA isolation method has to be able to fulfill several fundamental requirements for optimal performance of diagnostic assays. First of all, low- and high-molecular-weight substances of the blood inhibitory to downstream analytical reactions like PCR amplification have to be removed. This includes human DNA which is a known source of false-positive results and factor decreasing the analytical sensitivity of PCR assays by unspecific primer binding. At the same time, even extremely low amounts of microbial DNA need to be supplied to molecular diagnostic assays in order to detect low pathogen loads in the blood. Further, considering the variety of microbial etiologies of sepsis, a method should be capable of lysing Gram-positive, Gram-negative, and fungal organisms. Last, extraction buffers, reagents, and consumables have to be free of microbial DNA which leads to false-positive results. Here, we describe manual methods which allow the extraction of microbial DNA from small- and large-volume blood samples for the direct molecular analysis of pathogen.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Handschur M, Karlic H, Hertel C et al (2010) Preanalytic removal of human DNA eliminates false signals in general 16S rDNA PCR monitoring of bacterial pathogens in blood. Comp Immunol Microbiol Infect Dis 32:207–219
Disqué C (2007) Einfluss der DNA-Extraktion auf die PCR-Detektion von Sepsiserregern (in German: Influence of DNA extraction on the detection of sepsis pathogens by PCR). BIOspektrum 06:627–629
Horz HP, Scheer S, Huenger F et al (2008) Selective isolation of bacterial DNA from human clinical specimens. J Microbiol Methods 72:98–102
Wellinghausen N, Siegel D, Winter J, Gebert S (2009) Rapid diagnosis of candidaemia by real-time PCR detection of Candida DNA in blood samples. J Med Microbiol 58:1106–1111
Wellinghausen N, Siegel D, Gebert S, Winter J (2009) Rapid detection of Staphylococcus aureus bacteremia and methicillin resistance by real-time PCR in whole blood samples. Eur J Clin Microbiol Infect Dis 28:1001–1005
Wellinghausen N, Kochem AJ, Disqué C et al (2009) Diagnosis of bacteremia in whole-blood samples by use of a commercial universal 16S rRNA gene-based PCR and sequence analysis. J Clin Microbiol 47:2759–2765
Hansen WLJ, Bruggeman CA, Wolffs PFG (2009) Evaluation of new preanalysis sample treatment tools and DNA isolation protocols to improve bacterial pathogen detection in whole blood. J Clin Microbiol 47:2629–2631
Kühn C, Disqué C, Mühl H et al (2011) Evaluation of commercial universal rRNA gene PCR plus sequencing tests for identification of bacteria and fungi associated with infectious endocarditis. J Clin Microbiol 49:2919–2923
Esteban J, Alonso-Rodriguez N, del-Prado G et al (2012) PCR-hybridization after sonication improves diagnosis of implant-related infection. Acta Orthop 3:299–304
Xu Y, Børsholt RV, Simonsen O et al (2012) Bacterial diversity in suspected prosthetic joint infections: an exploratory study using 16S rRNA gene analysis. FEMS Immunol Med Microbiol 65:291–304
Benítez-Páez A, Álvarez M, Belda-Ferre P et al (2013) Detection of transient bacteraemia following dental extractions by 16S rDNA pyrosequencing: a pilot study. PLoS One 8:e57782. doi:10.1371/journal.pone.0057782
Haag H, Locher F, Nolte O (2013) Molecular diagnosis of microbial aetiologies using SepsiTest™ in the daily routine of a diagnostic laboratory. Diagn Microbiol Infect Dis 76: 413–418
Harrison E, Stahlberger T, Whelan R et al (2010) Aspergillus DNA contamination in blood collection tubes. Diagn Microbiol Infect Dis 67:392–394
Loeffler J, Hebart H, Bialek R et al (1999) Contaminations occurring in fungal PCR assays. J Clin Microbiol 37:1200–1202
Mohammadi T, Reesink HW, Vandenbroucke-Grauls CMJE, Savelkoul PHM (2005) Removal of contaminating DNA from commercial nucleic acid extraction kit reagents. J Microbiol Methods 61:285–288
Evans GE, Murdoch DR, Anderson TP et al (2003) Contamination of Qiagen DNA extraction kits with Legionella DNA. J Clin Microbiol 41:3452–3453
van der Zee A, Peeters M, de Jong C et al (2002) Qiagen DNA extraction kits for sample preparation for Legionella PCR are not suitable for diagnostic purposes. J Clin Microbiol 40: 1126
Fredricks DN, Smith CS, Meier A (2005) Comparison of six DNA extraction methods for recovery of fungal DNA as assessed by quantitative PCR. J Clin Microbiol 43: 5122–5128
Queipo-Ortuño MI, Tena F, Colmenero JD, Morata P (2008) Comparison of seven commercial DNA extraction kits for the recovery of Brucella DNA from spiked human serum samples using real-time PCR. Eur J Clin Microbiol Infect Dis 27:109–114
Orszag P, Disqué C, Keim S et al (2013) Monitoring of patients supported by extracorporeal membrane oxygenation for systemic infections by broad-range rRNA gene PCR amplification and sequence analysis. J Clin Microbiol 52:307–311. doi:10.1128/JCM.02493-13
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Lorenz, M.G., Mühl, H., Disqué, C. (2015). Bacterial and Fungal DNA Extraction from Blood Samples: Manual Protocols. In: Mancini, N. (eds) Sepsis. Methods in Molecular Biology, vol 1237. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1776-1_11
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1776-1_11
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1775-4
Online ISBN: 978-1-4939-1776-1
eBook Packages: Springer Protocols