Abstract
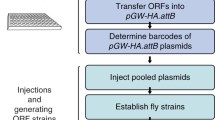
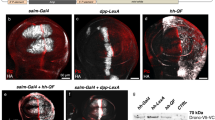
The Drosophila Gal4/UAS system allows the expression of any gene of interest in restricted domains. We devised a genetic strategy, based on the P-element replacement and UAS-y + techniques, to generate Gal4 lines inserted in Hox genes of Drosophila that are, at the same time, mutant for the resident genes. This makes possible to express different wild-type or mutant Hox proteins in the precise domains of Hox gene expression, and thus to test the functional value of these proteins in mutant rescue experiments.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Maeda RK, Karch F (2006) The ABC of the BX-C: the bithorax complex explained. Development 133:1413–1422
Kaufman TC, Seeger MA, Olsen G (1990) Molecular and genetic organization of the antennapedia gene complex of Drosophila melanogaster. Adv Genet 27:309–362
Alexander T, Nolte C, Krumlauf R (2009) Hox genes and segmentation of the hindbrain and axial skeleton. Annu Rev Cell Dev Biol 25:431–456
Wellik DM, Capecchi MR (2003) Hox10 and Hox11 genes are required to globally pattern the mammalian skeleton. Science 301:363–367
Greer JM, Puetz J, Thomas KR et al (2000) Maintenance of functional equivalence during paralogous Hox gene evolution. Nature 403:661–665
Zhao Y, Potter SS (2001) Functional specificity of the Hoxa13 homeobox. Development 128:3197–3207
Zhao Y, Potter SS (2002) Functional comparison of the Hoxa4, Hoxa10 and Hoxa11 homeoboxes. Dev Biol 244:21–36
Greig S, Akam M (1995) The role of homeotic genes in the specification of the Drosophila gonad. Curr Biol 5:1057–1062
Casares F, Calleja M, Sánchez-Herrero E (1996) Functional similarity in appendage specification by the Ultrabithorax and abdominal-A Drosophila Hox genes. EMBO J 15:3934–3942
Hirth F, Loop T, Egger B et al (2001) Functional equivalence of Hox gene products in the specification of the tritocerebrum during embryonic brain development of Drosophila. Development 128:4781–4788
Gehring WJ, Affolter M, Bürglin T (1994) Homeodomain proteins. Annu Rev Biochem 63:487–526
Merabet S, Hudry B, Saadaoui M et al (2009) Classification of sequence signatures: a guide to Hox protein function. Bioessays 31:500–511
Rong YS, Golic KG (2000) Gene targeting by homologous recombination in Drosophila. Science 288:2013–2018
Hittinger CT, Stern DL, Carroll SB (2005) Pleiotropic functions of a conserved insect-specific Hox peptide motif. Development 132:5261–5270
O’Keefe DD, Thor S, Thomas JB (1998) Function and specificity of LIM domains in Drosophila nervous system and wing development. Development 125:3915–3923
Rincón-Limas DE, Lu CH, Canal I et al (2000) The level of DLDB/CHIP controls the activity of the LIM homeodomain protein Apterous: evidence for a functional tetramer complex in vivo. EMBO J 19:2602–2614
Engels WR (1996) P-elements in Drosophila. In: Saedler H, Gierl A (eds) Transposable elements. Springer, Berlin, pp 103–123
Sepp KJ, Auld VJ (1999) Conversion of lacZ enhancer trap lines to GAL4 lines using targeted transposition in Drosophila melanogaster. Genetics 151:1093–1101
Calleja M, Moreno E, Pelaz S et al (1996) Visualization of gene expression in living adult Drosophila. Science 274:252–255
Engström Y, Schneuwly S, Gehring WJ (1992) Spatial and temporal expression of an Antennapedia/lacZ gene construct integrated into the endogenous Antennapedia gene of Drosophila melanogaster. Roux’s Arch Dev Biol 201:65–80
Galloni M, Gyurkovics H, Schedl P et al (1993) The bluetail transposon: evidence for independent cis-regulatory domains and domain boundaries in the bithorax complex. EMBO J 12:1087–1097
McCall K, O’Connor MB, Bender W (1994) Enhancer traps in the Drosophila bithorax complex mark parasegmental domains. Genetics 138:389–399
Casares F, Bender W, Merriam J et al (1997) Interactions of Drosophila Ultrabithorax regulatory regions with native and foreign promoters. Genetics 145:123–137
Zhou J, Levine M (1999) A novel cis-regulatory element, the PTS, mediates an anti-insulator activity in the Drosophila embryo. Cell 99:567–575
Barges S, Mihaly J, Galloni M et al (2000) The Fab-8 boundary defines the distal limit of the bithorax complex iab-7 domain and insulates iab-7 from initiation elements and a PRE in the adjacent iab-8 domain. Development 127:779–790
Bender W, Hudson A (2000) P element homing to the Drosophila bithorax complex. Development 127:3981–3992
Fitzgerald DP, Bender W (2001) Polycomb group repression reduces DNA accessibility. Mol Cell Biol 21:6585–6597
Estrada B, Casares F, Busturia A et al (2002) Genetic and molecular characterization of a novel iab-8 regulatory domain in the Abdominal-B gene of Drosophila melanogaster. Development 129:5195–5204
de Navas LF, Foronda D, Suzanne M et al (2006) A simple and efficient method to identify replacements of P-lacZ by P-Gal4 lines allows obtaining Gal4 insertions in the bithorax complex of Drosophila. Mech Dev 123:860–867
Hudry B, Viala S, Graba Y et al (2011) Visualization of protein interactions in living Drosophila embryos by the bimolecular fluorescence complementation assay. BMC Dev Biol 9:5
Robertson HM, Preston CR, Phillis RW et al (1988) A stable genomic source of P element transposase in Drosophila melanogaster. Genetics 118:461–470
Reuter G, Hoffmann G, Dorn R et al (1993) Construction and characterization of a TM3 balancer carrying P[(ry+) ∆2-3] as a stable transposase source. Dros Info Serv 72:78–79
Engels WR, Johnson-Schlitz DM, Eggleston WB et al (1990) High-frequency P element loss in Drosophila is homolog dependent. Cell 62:515–525
Gloor GB, Nassif NA, Johnson-Schlitz DM et al (1991) Targeted gene replacement in Drosophila via P element-induced gap repair. Science 253:1110–1117
Gohl DM, Silies MA, Gao XJ et al (2011) A versatile in vivo system for directed dissection of gene expression patterns. Nat Methods 8:231–237
Potter CJ, Tasic B, Russler EV et al (2010) The Q system: a repressible binary system for transgene expression, lineage tracing, and mosaic analysis. Cell 141:536–548
Yagi R, Mayer F, Basler K (2010) Refined LexA transactivators and their use in combination with the Drosophila Gal4 system. Proc Natl Acad Sci U S A 107:6166–61671
Nassif N, Penney J, Pal S et al (1994) Efficient copying of nonhomologous sequences from ectopic sites via P-element-induced gap repair. Mol Cell Biol 14:1613–1625
Acknowledgements
Work in the laboratory is being supported by a grant from the Spanish Ministerio de Economía y Competitividad (BFU2011-26075) and an institutional grant from the Fundación Ramón Areces. Delia del Saz is being supported by an FPI fellowship from the Spanish Ministerio de Economía y Competitividad.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
de Navas, L., Foronda, D., del Saz, D., Sánchez-Herrero, E. (2014). A Genetic Strategy to Obtain P-Gal4 Elements in the Drosophila Hox Genes. In: Graba, Y., Rezsohazy, R. (eds) Hox Genes. Methods in Molecular Biology, vol 1196. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1242-1_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1242-1_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1241-4
Online ISBN: 978-1-4939-1242-1
eBook Packages: Springer Protocols