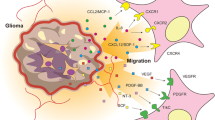
Abstract
Malignant brain tumors are nearly untreatable due to their highly infiltrative nature and resistance to existing therapies. The main reason for recurrent tumor growth is believed to be the presence of tumor cells that migrate great distances into the brain tissue. In addition, poor delivery of therapeutics to the tumors due to blood–brain barrier limits the clinical success of currently available systemically delivered antitumor therapies. Recently, a different mode of therapeutic delivery, whereby therapeutic biomolecules are expressed by tumor-tropic neural stem cells (NSCs), has gained considerable attention. Exploiting the intrinsic tumor-homing ability of NSCs, the past decade has witnessed significant advances in the discovery and development of NSC-based therapies for malignant brain tumors. Prodrug converting enzymes, immunomodulatory cytokines, pro-apoptotic (tumouricidal) agents, growth-inhibiting factors, anti-angiogenic agents, and viral particles have been among the most commonly studied antitherapeutic molecules produced by NSCs. While the mechanisms of tumor-directed NSC migration and fate of NSCs after engrafting are still not truly understood, the results from current preclinical tumor models have demonstrated promising utility for NSCs as “armed vehicles” in treatment of aggressive brain tumors. Indeed, the first clinical trial with NSC-delivered antitumor agents is now in progress for recurrent gliomas.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- GBM:
-
Glioblastoma multiforme
- TMZ:
-
Temozolomide
- BBB:
-
Blood–brain barrier
- NSC:
-
Neural stem cell
- CNS:
-
Central nervous system
- ES:
-
Embryonic stem
- SVZ:
-
Subventricular zone
- Fluc:
-
Firefly luciferase
- Rluc:
-
Renilla luciferase
- MRI:
-
Magnetic resonance imaging
- BLI:
-
Bioluminescence imaging
- FE-Pro:
-
Ferumoxide protamine sulfate complex
- SF/HGF:
-
Scatter factor/hepatocyte growth factor
- SDF-1:
-
Stromal-derived factor
- CXCR4:
-
CXC chemokine receptor 4
- VEGF:
-
Vascular endothelial growth factor
- PI3K:
-
Phosphoinositide 3 kinase
- TMEM18:
-
Transmembrane protein 18
- ECM:
-
Extracellular matrix
- PCE:
-
Prodrug converting enzyme
- HSV-TK:
-
Herpes simplex virus-thymidine kinase
- CD:
-
Cytosine deaminase
- CE:
-
Carboxylesterase
- GCV:
-
Ganciclovir
- 5-FC:
-
5-Fluorocytosine
- 5-FU:
-
5-Fluorouracil
- CPT-11:
-
Camptothecin-11
- CPA:
-
Cyclophosphamide
- CYP2B6:
-
CPA-activating enzyme cytochrome p450 2B6
- TSP:
-
Thrombospondin
- OV:
-
Oncolytic virus
- CRAd:
-
Conditionally replicating adenovirus
- MMP:
-
Matrix metalloproteinase
- EGFR:
-
Epidermal growth factor receptor
- ENb:
-
EGFR targeting nanobody
- IL:
-
Interleukin
- IFN:
-
Interferon
- BM-NSC:
-
Bone marrow-derived NSC
- TRAIL:
-
Tumor necrosis factor-related apoptosis-inducing ligand
- GMP:
-
Good manufacturing practice
- iPSc:
-
Induced pluripotent stem cell
References
Huse JT, Holland EC (2010) Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat Rev Cancer 10(5):319–331
Berens ME, Giese A (1999) “…those left behind.” Biology and oncology of invasive glioma cells. Neoplasia 1(3):208–219
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996
Bagci T, Wu JK, Pfannl R, Ilag LL, Jay DG (2009) Autocrine semaphorin 3A signaling promotes glioblastoma dispersal. Oncogene 28(40):3537–3550
Jain RK, di Tomaso E, Duda DG, Loeffler JS, Sorensen AG, Batchelor TT (2007) Angiogenesis in brain tumours. Nat Rev Neurosci 8(8):610–622
Zhou J, Atsina KB, Himes BT, Strohbehn GW, Saltzman WM (2012) Novel delivery strategies for glioblastoma. Cancer J 18(1):89–99
Conti L, Cattaneo E (2010) Neural stem cell systems: physiological players or in vitro entities? Nat Rev Neurosci 11(3):176–187
Conti L, Reitano E, Cattaneo E (2006) Neural stem cell systems: diversities and properties after transplantation in animal models of diseases. Brain Pathol 16(2):143–154
Rossi F, Cattaneo E (2002) Opinion: neural stem cell therapy for neurological diseases: dreams and reality. Nat Rev Neurosci 3(5):401–409
Tsuji O, Miura K, Fujiyoshi K, Momoshima S, Nakamura M, Okano H (2011) Cell therapy for spinal cord injury by neural stem/progenitor cells derived from iPS/ES cells. Neurotherapeutics 8(4):668–676
Aboody K, Capela A, Niazi N, Stern JH, Temple S (2011) Translating stem cell studies to the clinic for CNS repair: current state of the art and the need for a Rosetta stone. Neuron 70(4):597–613
Okabe S, Forsberg-Nilsson K, Spiro AC, Segal M, McKay RD (1996) Development of neuronal precursor cells and functional postmitotic neurons from embryonic stem cells in vitro. Mech Dev 59(1):89–102
Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, Studer L (2009) Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol 27(3):275–280
Ming GL, Song H (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70(4):687–702
Muller FJ, Snyder EY, Loring JF (2006) Gene therapy: can neural stem cells deliver? Nat Rev Neurosci 7(1):75–84
Ahmed AU, Alexiades NG, Lesniak MS (2010) The use of neural stem cells in cancer gene therapy: predicting the path to the clinic. Curr Opin Mol Ther 12(5):546–552
Kim SU (2011) Neural stem cell-based gene therapy for brain tumors. Stem Cell Rev 7(1):130–140
Aboody KS, Najbauer J, Danks MK (2008) Stem and progenitor cell-mediated tumor selective gene therapy. Gene Ther 15(10):739–752
Aboody KS, Brown A, Rainov NG, Bower KA, Liu S, Yang W, Small JE, Herrlinger U, Ourednik V, Black PM, Breakefield XO et al (2000) Neural stem cells display extensive tropism for pathology in adult brain: evidence from intracranial gliomas. Proc Natl Acad Sci USA 97(23):12846–12851
Kim SU (2007) Genetically engineered human neural stem cells for brain repair in neurological diseases. Brain Dev 29(4):193–201
Kim JH, Lee JE, Kim SU, Cho KG (2010) Stereological analysis on migration of human neural stem cells in the brain of rats bearing glioma. Neurosurgery 66(2):333–342, discussion 342
Lin D, Najbauer J, Salvaterra PM, Mamelak AN, Barish ME, Garcia E, Metz MZ, Kendall SE, Bowers M, Kateb B, Kim SU et al (2007) Novel method for visualizing and modeling the spatial distribution of neural stem cells within intracranial glioma. Neuroimage 37(Suppl 1): S18–S26
Jeon JY, An JH, Kim SU, Park HG, Lee MA (2008) Migration of human neural stem cells toward an intracranial glioma. Exp Mol Med 40(1):84–91
Tang Y, Shah K, Messerli SM, Snyder E, Breakefield X, Weissleder R (2003) In vivo tracking of neural progenitor cell migration to glioblastomas. Hum Gene Ther 14(13):1247–1254
Shah K, Bureau E, Kim DE, Yang K, Tang Y, Weissleder R, Breakefield XO (2005) Glioma therapy and real-time imaging of neural precursor cell migration and tumor regression. Ann Neurol 57(1):34–41
Shah K, Hingtgen S, Kasmieh R, Figueiredo JL, Garcia-Garcia E, Martinez-Serrano A, Breakefield X, Weissleder R (2008) Bimodal viral vectors and in vivo imaging reveal the fate of human neural stem cells in experimental glioma model. J Neurosci 28(17):4406–4413
Shah K (2009) Imaging neural stem cell fate in mouse model of glioma. Curr Protoc Stem Cell Biol Chapter 5: unit 5A 1
Shah K (2011) Imaging fate of stem cells at a cellular resolution in the brains of mice. Methods Mol Biol 680:91–101
Ashwal S, Obenaus A, Snyder EY (2009) Neuroimaging as a basis for rational stem cell therapy. Pediatr Neurol 40(3):227–236
Thu MS, Najbauer J, Kendall SE, Harutyunyan I, Sangalang N, Gutova M, Metz MZ, Garcia E, Frank RT, Kim SU, Moats RA et al (2009) Iron labeling and pre-clinical MRI visualization of therapeutic human neural stem cells in a murine glioma model. PLoS One 4(9):e7218
Heese O, Disko A, Zirkel D, Westphal M, Lamszus K (2005) Neural stem cell migration toward gliomas in vitro. Neuro Oncol 7(4):476–484
Imitola J, Raddassi K, Park KI, Mueller FJ, Nieto M, Teng YD, Frenkel D, Li J, Sidman RL, Walsh CA, Snyder EY et al (2004) Directed migration of neural stem cells to sites of CNS injury by the stromal cell-derived factor 1alpha/CXC chemokine receptor 4 pathway. Proc Natl Acad Sci USA 101(52):18117–18122
Ehtesham M, Yuan X, Kabos P, Chung NH, Liu G, Akasaki Y, Black KL, Yu JS (2004) Glioma tropic neural stem cells consist of astrocytic precursors and their migratory capacity is mediated by CXCR4. Neoplasia 6(3):287–293
Harris AL (2002) Hypoxia – a key regulatory factor in tumour growth. Nat Rev Cancer 2(1):38–47
Schmidt NO, Przylecki W, Yang W, Ziu M, Teng Y, Kim SU, Black PM, Aboody KS, Carroll RS (2005) Brain tumor tropism of transplanted human neural stem cells is induced by vascular endothelial growth factor. Neoplasia 7(6):623–629
Zhao D, Najbauer J, Garcia E, Metz MZ, Gutova M, Glackin CA, Kim SU, Aboody KS (2008) Neural stem cell tropism to glioma: critical role of tumor hypoxia. Mol Cancer Res 6(12):1819–1829
Kendall SE, Najbauer J, Johnston HF, Metz MZ, Li S, Bowers M, Garcia E, Kim SU, Barish ME, Aboody KS, Glackin CA (2008) Neural stem cell targeting of glioma is dependent on phosphoinositide 3-kinase signaling. Stem Cells 26(6):1575–1586
Jurvansuu J, Zhao Y, Leung DS, Boulaire J, Yu YH, Ahmed S, Wang S (2008) Transmembrane protein 18 enhances the tropism of neural stem cells for glioma cells. Cancer Res 68(12):4614–4622
Ziu M, Schmidt NO, Cargioli TG, Aboody KS, Black PM, Carroll RS (2006) Glioma-produced extracellular matrix influences brain tumor tropism of human neural stem cells. J Neurooncol 79(2):125–133
An JH, Lee SY, Jeon JY, Cho KG, Kim SU, Lee MA (2009) Identification of gliotropic factors that induce human stem cell migration to malignant tumor. J Proteome Res 8(6): 2873–2881
Yip S, Shah K (2008) Stem-cell based therapies for brain tumors. Curr Opin Mol Ther 10(4):334–342
Yip S, Aboody KS, Burns M, Imitola J, Boockvar JA, Allport J, Park KI, Teng YD, Lachyankar M, McIntosh T, O’Rourke DM et al (2003) Neural stem cell biology may be well suited for improving brain tumor therapies. Cancer J 9(3):189–204
Binello E, Germano IM (2012) Stem cells as therapeutic vehicles for the treatment of high-grade gliomas. Neuro Oncol 14(3):256–265
Brown AB, Yang W, Schmidt NO, Carroll R, Leishear KK, Rainov NG, Black PM, Breakefield XO, Aboody KS (2003) Intravascular delivery of neural stem cell lines to target intracranial and extracranial tumors of neural and non-neural origin. Hum Gene Ther 14(18):1777–1785
Kim SK, Kim SU, Park IH, Bang JH, Aboody KS, Wang KC, Cho BK, Kim M, Menon LG, Black PM, Carroll RS (2006) Human neural stem cells target experimental intracranial medulloblastoma and deliver a therapeutic gene leading to tumor regression. Clin Cancer Res 12(18):5550–5556
Shimato S, Natsume A, Takeuchi H, Wakabayashi T, Fujii M, Ito M, Ito S, Park IH, Bang JH, Kim SU, Yoshida J (2007) Human neural stem cells target and deliver therapeutic gene to experimental leptomeningeal medulloblastoma. Gene Ther 14(15):1132–1142
Lee DH, Ahn Y, Kim SU, Wang KC, Cho BK, Phi JH, Park IH, Black PM, Carroll RS, Lee J, Kim SK (2009) Targeting rat brainstem glioma using human neural stem cells and human mesenchymal stem cells. Clin Cancer Res 15(15):4925–4934
Gutova M, Najbauer J, Chen MY, Potter PM, Kim SU, Aboody KS (2010) Therapeutic targeting of melanoma cells using neural stem cells expressing carboxylesterase, a CPT-11 activating enzyme. Curr Stem Cell Res Ther 5(3):273–276
Aboody KS, Najbauer J, Schmidt NO, Yang W, Wu JK, Zhuge Y, Przylecki W, Carroll R, Black PM, Perides G (2006) Targeting of melanoma brain metastases using engineered neural stem/progenitor cells. Neuro Oncol 8(2):119–126
Joo KM, Park IH, Shin JY, Jin J, Kang BG, Kim MH, Lee SJ, Jo MY, Kim SU, Nam DH (2009) Human neural stem cells can target and deliver therapeutic genes to breast cancer brain metastases. Mol Ther 17(3):570–575
Lee SJ, Kim Y, Jo MY, Kim HS, Jin Y, Kim SU, Jin J, Joo KM, Nam DH (2011) Combined treatment of tumor-tropic human neural stem cells containing the CD suicide gene effectively targets brain tumors provoking a mild immune response. Oncol Rep 25(1):63–68
Danks MK, Yoon KJ, Bush RA, Remack JS, Wierdl M, Tsurkan L, Kim SU, Garcia E, Metz MZ, Najbauer J, Potter PM et al (2007) Tumor-targeted enzyme/prodrug therapy mediates long-term disease-free survival of mice bearing disseminated neuroblastoma. Cancer Res 67(1):22–25
Lim SH, Choi SA, Lee JY, Wang KC, Phi JH, Lee DH, Song SH, Song JH, Jin X, Kim H, Lee HJ et al (2011) Therapeutic targeting of subdural medulloblastomas using human neural stem cells expressing carboxylesterase. Cancer Gene Ther 18(11):817–824
Gutova M, Shackleford GM, Khankaldyyan V, Herrmann KA, Shi XH, Mittelholtz K, Abramyants Y, Blanchard MS, Kim SU, Annala AJ, Najbauer J et al (2012) Neural stem cell-mediated CE/CPT-11 enzyme/prodrug therapy in transgenic mouse model of intracerebellar medulloblastoma. Gene Ther 20(2):143–150
Seol HJ, Jin J, Seong DH, Joo KM, Kang W, Yang H, Kim J, Shin CS, Kim Y, Kim KH, Kong DS et al (2011) Genetically engineered human neural stem cells with rabbit carboxyl esterase can target brain metastasis from breast cancer. Cancer Lett 311(2):152–159
Zhao D, Najbauer J, Annala AJ, Garcia E, Metz MZ, Gutova M, Polewski MD, Gilchrist M, Glackin CA, Kim SU, Aboody KS (2012) Human neural stem cell tropism to metastatic breast cancer. Stem Cells 30(2):314–325
Li S, Tokuyama T, Yamamoto J, Koide M, Yokota N, Namba H (2005) Bystander effect-mediated gene therapy of gliomas using genetically engineered neural stem cells. Cancer Gene Ther 12(7):600–607
Li S, Tokuyama T, Yamamoto J, Koide M, Yokota N, Namba H (2005) Potent bystander effect in suicide gene therapy using neural stem cells transduced with herpes simplex virus thymidine kinase gene. Oncology 69(6):503–508
Li S, Gao Y, Tokuyama T, Yamamoto J, Yokota N, Yamamoto S, Terakawa S, Kitagawa M, Namba H (2007) Genetically engineered neural stem cells migrate and suppress glioma cell growth at distant intracranial sites. Cancer Lett 251(2):220–227
Uhl M, Weiler M, Wick W, Jacobs AH, Weller M, Herrlinger U (2005) Migratory neural stem cells for improved thymidine kinase-based gene therapy of malignant gliomas. Biochem Biophys Res Commun 328(1):125–129
Zhao Y, Wang S (2010) Human NT2 neural precursor-derived tumor-infiltrating cells as delivery vehicles for treatment of glioblastoma. Hum Gene Ther 21(6):683–694
Mercapide J, Rappa G, Anzanello F, King J, Fodstad O, Lorico A (2010) Primary gene-engineered neural stem/progenitor cells demonstrate tumor-selective migration and antitumor effects in glioma. Int J Cancer 126(5):1206–1215
Wang C, Natsume A, Lee HJ, Motomura K, Nishimira Y, Ohno M, Ito M, Kinjo S, Momota H, Iwami K, Ohka F et al (2012) Neural stem cell-based dual suicide gene delivery for metastatic brain tumors. Cancer Gene Ther 19(11):796–801
van Eekelen M, Sasportas LS, Kasmieh R, Yip S, Figueiredo JL, Louis DN, Weissleder R, Shah K (2010) Human stem cells expressing novel TSP-1 variant have anti-angiogenic effect on brain tumors. Oncogene 29(22):3185–3195
Herrlinger U, Woiciechowski C, Sena-Esteves M, Aboody KS, Jacobs AH, Rainov NG, Snyder EY, Breakefield XO (2000) Neural precursor cells for delivery of replication-conditional HSV-1 vectors to intracerebral gliomas. Mol Ther 1(4):347–357
Tyler MA, Ulasov IV, Sonabend AM, Nandi S, Han Y, Marler S, Roth J, Lesniak MS (2009) Neural stem cells target intracranial glioma to deliver an oncolytic adenovirus in vivo. Gene Ther 16(2):262–278
Ahmed AU, Thaci B, Alexiades NG, Han Y, Qian S, Liu F, Balyasnikova IV, Ulasov IY, Aboody KS, Lesniak MS (2011) Neural stem cell-based cell carriers enhance therapeutic efficacy of an oncolytic adenovirus in an orthotopic mouse model of human glioblastoma. Mol Ther 19(9):1714–1726
Ahmed AU, Tyler MA, Thaci B, Alexiades NG, Han Y, Ulasov IV, Lesniak MS (2010) A comparative study of neural and mesenchymal stem cell-based carriers for oncolytic adenovirus in a model of malignant glioma. Mol Pharm 8(5):1559–1572
Thaci B, Ahmed AU, Ulasov IV, Tobias AL, Han Y, Aboody KS, Lesniak MS (2012) Pharmacokinetic study of neural stem cell-based cell carrier for oncolytic virotherapy: targeted delivery of the therapeutic payload in an orthotopic brain tumor model. Cancer Gene Ther 19(6):431–442
Kim SK, Cargioli TG, Machluf M, Yang W, Sun Y, Al-Hashem R, Kim SU, Black PM, Carroll RS (2005) PEX-producing human neural stem cells inhibit tumor growth in a mouse glioma model. Clin Cancer Res 11(16):5965–5970
Firer MA, Gellerman G (2012) Targeted drug delivery for cancer therapy: the other side of antibodies. J Hematol Oncol 5(1):70
Frank RT, Edmiston M, Kendall SE, Najbauer J, Cheung CW, Kassa T, Metz MZ, Kim SU, Glackin CA, Wu AM, Yazaki PJ et al (2009) Neural stem cells as a novel platform for tumor-specific delivery of therapeutic antibodies. PLoS One 4(12):e8314
Holliger P, Hudson PJ (2005) Engineered antibody fragments and the rise of single domains. Nat Biotechnol 23(9):1126–1136
Muyldermans S (2001) Single domain camel antibodies: current status. J Biotechnol 74(4):277–302
Van de Water JA, Bagci-Onder T, Agarwal AS, Wakimoto H, Roovers RC, Zhu Y, Kasmieh R, Bhere D, Van Bergen En Henegouwen PM, Shah K (2012) Therapeutic stem cells expressing variants of EGFR-specific nanobodies have antitumor effects. Proc Natl Acad Sci USA 109(41):16642–16647
Benedetti S, Pirola B, Pollo B, Magrassi L, Bruzzone MG, Rigamonti D, Galli R, Selleri S, Di Meco F, De Fraja C, Vescovi A et al (2000) Gene therapy of experimental brain tumors using neural progenitor cells. Nat Med 6(4):447–450
Ehtesham M, Kabos P, Kabosova A, Neuman T, Black KL, Yu JS (2002) The use of interleukin 12-secreting neural stem cells for the treatment of intracranial glioma. Cancer Res 62(20):5657–5663
Yuan X, Hu J, Belladonna ML, Black KL, Yu JS (2006) Interleukin-23-expressing bone marrow-derived neural stem-like cells exhibit antitumor activity against intracranial glioma. Cancer Res 66(5):2630–2638
Hingtgen S, Kasmieh R, Elbayly E, Nesterenko I, Figueiredo JL, Dash R, Sarkar D, Hall D, Kozakov D, Vajda S, Fisher PB et al (2012) A first-generation multi-functional cytokine for simultaneous optical tracking and tumor therapy. PLoS One 7(7):e40234
Wang BX, Rahbar R, Fish EN (2011) Interferon: current status and future prospects in cancer therapy. J Interferon Cytokine Res 31(7):545–552
Dickson PV, Hamner JB, Burger RA, Garcia E, Ouma AA, Kim SU, Ng CY, Gray JT, Aboody KS, Danks MK, Davidoff AM (2007) Intravascular administration of tumor tropic neural progenitor cells permits targeted delivery of interferon-beta and restricts tumor growth in a murine model of disseminated neuroblastoma. J Pediatr Surg 42(1):48–53
Sims TL Jr, Hamner JB, Bush RA, Williams RF, Zhou J, Kim SU, Aboody KS, Danks MK, Davidoff AM (2008) Neural progenitor cell-mediated delivery of interferon beta improves neuroblastoma response to cyclophosphamide. Ann Surg Oncol 15(11):3259–3267
Pitti RM, Marsters SA, Ruppert S, Donahue CJ, Moore A, Ashkenazi A (1996) Induction of apoptosis by Apo-2 ligand, a new member of the tumor necrosis factor cytokine family. J Biol Chem 271(22):12687–12690
Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA et al (1995) Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3(6):673–682
Ashkenazi A (2008) Targeting the extrinsic apoptosis pathway in cancer. Cytokine Growth Factor Rev 19(3–4):325–331
Ashkenazi A, Holland P, Eckhardt SG (2008) Ligand-based targeting of apoptosis in cancer: the potential of recombinant human apoptosis ligand 2/Tumor necrosis factor-related apoptosis-inducing ligand (rhApo2L/TRAIL). J Clin Oncol 26(21):3621–3630
Dimberg LY, Anderson CK, Camidge R, Behbakht K, Thorburn A, Ford HL (2012) On the TRAIL to successful cancer therapy? Predicting and counteracting resistance against TRAIL-based therapeutics. Oncogene 32(11):1341–1350
Gonzalvez F, Ashkenazi A (2010) New insights into apoptosis signaling by Apo2L/TRAIL. Oncogene 29(34):4752–4765
Johnstone RW, Frew AJ, Smyth MJ (2008) The TRAIL apoptotic pathway in cancer onset, progression and therapy. Nat Rev Cancer 8(10):782–798
Kelley SK, Harris LA, Xie D, Deforge L, Totpal K, Bussiere J, Fox JA (2001) Preclinical studies to predict the disposition of Apo2L/tumor necrosis factor-related apoptosis-inducing ligand in humans: characterization of in vivo efficacy, pharmacokinetics, and safety. J Pharmacol Exp Ther 299(1):31–38
Wiezorek J, Holland P, Graves J (2010) Death receptor agonists as a targeted therapy for cancer. Clin Cancer Res 16(6):1701–1708
Ashkenazi A (2008) Directing cancer cells to self-destruct with pro-apoptotic receptor agonists. Nat Rev Drug Discov 7(12):1001–1012
Ashkenazi A, Herbst RS (2008) To kill a tumor cell: the potential of proapoptotic receptor agonists. J Clin Invest 118(6):1979–1990
Mahalingam D, Szegezdi E, Keane M, de Jong S, Samali A (2009) TRAIL receptor signalling and modulation: are we on the right TRAIL? Cancer Treat Rev 35(3):280–288
Pennarun B, Meijer A, de Vries EG, Kleibeuker JH, Kruyt F, de Jong S (2010) Playing the DISC: turning on TRAIL death receptor-mediated apoptosis in cancer. Biochim Biophys Acta 1805(2):123–140
Ehtesham M, Kabos P, Gutierrez MA, Chung NH, Griffith TS, Black KL, Yu JS (2002) Induction of glioblastoma apoptosis using neural stem cell-mediated delivery of tumor necrosis factor-related apoptosis-inducing ligand. Cancer Res 62(24):7170–7174
Shah K, Tang Y, Breakefield X, Weissleder R (2003) Real-time imaging of TRAIL-induced apoptosis of glioma tumors in vivo. Oncogene 22(44):6865–6872
Shah K, Tung CH, Breakefield XO, Weissleder R (2005) In vivo imaging of S-TRAIL-mediated tumor regression and apoptosis. Mol Ther 11(6):926–931
Shah K, Tung CH, Yang K, Weissleder R, Breakefield XO (2004) Inducible release of TRAIL fusion proteins from a proapoptotic form for tumor therapy. Cancer Res 64(9):3236–3242
Corsten MF, Miranda R, Kasmieh R, Krichevsky AM, Weissleder R, Shah K (2007) MicroRNA-21 knockdown disrupts glioma growth in vivo and displays synergistic cytotoxicity with neural precursor cell delivered S-TRAIL in human gliomas. Cancer Res 67(19):8994–9000
Kock N, Kasmieh R, Weissleder R, Shah K (2007) Tumor therapy mediated by lentiviral expression of shBcl-2 and S-TRAIL. Neoplasia 9(5):435–442
Hingtgen S, Ren X, Terwilliger E, Classon M, Weissleder R, Shah K (2008) Targeting multiple pathways in gliomas with stem cell and viral delivered S-TRAIL and Temozolomide. Mol Cancer Ther 7(11):3575–3585
Bagci-Onder T, Wakimoto H, Anderegg M, Cameron C, Shah K (2011) A dual PI3K/mTOR inhibitor, PI-103, cooperates with stem cell-delivered TRAIL in experimental glioma models. Cancer Res 71(1):154–163
Bagci-Onder T, Agarwal A, Flusberg D, Wanningen S, Sorger P, Shah K (2012) Real-time imaging of the dynamics of death receptors and therapeutics that overcome TRAIL resistance in tumors. Oncogene. doi:10.1038/onc.2012.304
Balyasnikova IV, Ferguson SD, Han Y, Liu F, Lesniak MS (2011) Therapeutic effect of neural stem cells expressing TRAIL and bortezomib in mice with glioma xenografts. Cancer Lett 310(2):148–159
Hingtgen SD, Kasmieh R, Van de Water J, Weissleder R, Shah K (2010) A novel molecule integrating therapeutic and diagnostic activities reveals multiple aspects of stem cell-based therapy. Stem Cells 28(4):832–841
Ito S, Natsume A, Shimato S, Ohno M, Kato T, Chansakul P, Wakabayashi T, Kim SU (2010) Human neural stem cells transduced with IFN-beta and cytosine deaminase genes intensify bystander effect in experimental glioma. Cancer Gene Ther 17(5):299–306
Kauer TM, Figueiredo JL, Hingtgen S, Shah K (2012) Encapsulated therapeutic stem cells implanted in the tumor resection cavity induce cell death in gliomas. Nat Neurosci 15(2):197–204
Tsuji O, Miura K, Okada Y, Fujiyoshi K, Mukaino M, Nagoshi N, Kitamura K, Kumagai G, Nishino M, Tomisato S, Higashi H et al (2010) Therapeutic potential of appropriately evaluated safe-induced pluripotent stem cells for spinal cord injury. Proc Natl Acad Sci USA 107(28):12704–12709
Han SS, Williams LA, Eggan KC (2011) Constructing and deconstructing stem cell models of neurological disease. Neuron 70(4):626–644
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Bagci-Onder, T. (2013). Neural Stem Cells as Therapeutic Delivery Vehicles for Malignant Brain Tumors. In: Turksen, K. (eds) Stem Cells: Current Challenges and New Directions. Stem Cell Biology and Regenerative Medicine. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-8066-2_12
Download citation
DOI: https://doi.org/10.1007/978-1-4614-8066-2_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-8065-5
Online ISBN: 978-1-4614-8066-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)