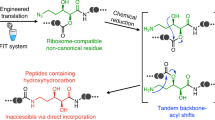
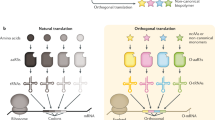
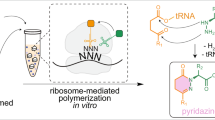
Abstract
Noncanonical peptide backbone structures, such as heterocycles and non-α-amino acids, are characteristic building blocks present in peptidic natural products. To achieve ribosomal synthesis of designer peptides bearing such noncanonical backbone structures, we have devised translation-compatible precursor residues and their chemical posttranslational modification processes. In this chapter, we describe the detailed procedures for the in vitro translation of peptides containing the precursor residues by means of genetic code reprogramming technology and posttranslational generation of objective noncanonical backbone structures.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Walsh CT, O’Brien RV, Khosla C (2013) Nonproteinogenic amino acid building blocks for nonribosomal peptide and hybrid polyketide scaffolds. Angew Chem Int Ed Engl 52:7098–7124
Arnison PG, Bibb MJ, Bierbaum G et al (2013) Ribosomally synthesized and post-translationally modified peptide natural products: overview and recommendations for a universal nomenclature. Nat Prod Rep 30:108–160
Montalban-Lopez M, Scott TA, Ramesh S et al (2020) New developments in RiPP discovery, enzymology and engineering. Nat Prod Rep 38:130–239
Goto Y, Suga H (2021) The RaPID platform for the discovery of pseudo-natural macrocyclic peptides. Acc Chem Res 54:3604–3617
Kofman C, Lee J, Jewett MC (2021) Engineering molecular translation systems. Cell Syst 12:593–607
Hartman MCT (2022) Non-canonical amino acid substrates of E. coli aminoacyl-tRNA synthetases. ChemBioChem 23:e202100299
Hecht SM (2022) Expansion of the genetic code through the use of modified bacterial ribosomes. J Mol Biol 434:167211
Goto Y, Ohta A, Sako Y et al (2008) Reprogramming the translation initiation for the synthesis of physiologically stable cyclic peptides. ACS Chem Biol 3:120–129
Goto Y, Iwasaki K, Torikai K et al (2009) Ribosomal synthesis of dehydrobutyrine- and methyllanthionine-containing peptides. Chem Commun (Camb) 23:3419–3421
Nakajima E, Goto Y, Sako Y et al (2009) Ribosomal synthesis of peptides with C-terminal lactams, thiolactones, and alkylamides. ChemBioChem 10:1186–1192
Kato Y, Kuroda T, Huang Y et al (2020) Chemoenzymatic posttranslational modification reactions for the synthesis of Psi[CH2 NH]-containing peptides. Angew Chem Int Ed Engl 59:684–688
Tsutsumi H, Kuroda T, Kimura H et al (2021) Posttranslational chemical installation of azoles into translated peptides. Nat Commun 12:696
Kuroda T, Huang Y, Nishio S et al (2022) Posttranslational backbone-acyl shift yields natural product-like peptides bearing hydroxyhydrocarbon units. Nat Chem:in press
Murakami H, Ohta A, Ashigai H et al (2006) A highly flexible tRNA acylation method for non-natural polypeptide synthesis. Nat Methods 3:357–359
Goto Y, Katoh T, Suga H (2011) Flexizymes for genetic code reprogramming. Nat Protoc 6:779
Wipf P, Fritch PC, Geib SJ et al (1998) Conformational studies and structure-activity analysis of lissoclinamide 7 and related cyclopeptide alkaloids. J Am Chem Soc 120:4105–4112
Siodlak D, Stas M, Broda MA et al (2014) Conformational properties of oxazole-amino acids: effect of the intramolecular N-H...N hydrogen bond. J Phys Chem B 118:2340–2350
Ahlbach CL, Lexa KW, Bockus AT et al (2015) Beyond cyclosporine A: conformation-dependent passive membrane permeabilities of cyclic peptide natural products. Future Med Chem 7:2121–2130
Marshall CG, Burkart MD, Keating TA et al (2001) Heterocycle formation in vibriobactin biosynthesis: alternative substrate utilization and identification of a condensed intermediate. Biochemistry 40:10655–10663
Schneider TL, Shen B, Walsh CT (2003) Oxidase domains in epothilone and bleomycin biosynthesis: thiazoline to thiazole oxidation during chain elongation. Biochemistry 42:9722–9730
Dunbar KL, Melby JO, Mitchell DA (2012) YcaO domains use ATP to activate amide backbones during peptide cyclodehydrations. Nat Chem Biol 8:569–575
Koehnke J, Bent AF, Zollman D et al (2013) The cyanobactin heterocyclase enzyme: a processive adenylase that operates with a defined order of reaction. Angew Chem Int Ed Engl 52:13991–13996
Melby JO, Li X, Mitchell DA (2014) Orchestration of enzymatic processing by thiazole/oxazole-modified microcin dehydrogenases. Biochemistry 53:413–422
Chowdhury SR, Maini R, Dedkova LM et al (2015) Synthesis of fluorescent dipeptidomimetics and their ribosomal incorporation into green fluorescent protein. Bioorg Med Chem Lett 25:4715–4718
Chowdhury SR, Chauhan PS, Dedkova LM et al (2016) Synthesis and evaluation of a library of fluorescent dipeptidomimetic analogues as substrates for modified bacterial ribosomes. Biochemistry 55:2427–2440
Maini R, Kimura H, Takatsuji R et al (2019) Ribosomal formation of thioamide bonds in polypeptide synthesis. J Am Chem Soc 141:20004–20008
Bott R, Subramanian E, Davies DR (1982) Three-dimensional structure of the complex of the Rhizopus chinensis carboxyl proteinase and pepstatin at 2.5-A resolution. Biochemistry 21:6956–6962
Rich DH, Bernatowicz MS, Agarwal NS et al (1985) Inhibition of aspartic proteases by pepstatin and 3-methylstatine derivatives of pepstatin. Evidence for collected-substrate enzyme inhibition. Biochemistry 24:3165–3173
Umezawa H, Aoyagi T, Morishima H et al (1970) Pepstatin, a new pepsin inhibitor produced by actinomycetes. J Antibiot (Tokyo) 23:259–262
Tumminello FM, Bernacki RJ, Gebbia N et al (1993) Pepstatins: aspartic proteinase inhibitors having potential therapeutic applications. Med Res Rev 13:199–208
Kuranaga T, Matsuda K, Takaoka M et al (2020) Total synthesis and structural revision of kasumigamide, and identification of a new analogue. ChemBioChem 21:3329–3332
Lee J, Schwarz KJ, Kim DS et al (2020) Ribosome-mediated polymerization of long chain carbon and cyclic amino acids into peptides in vitro. Nat Commun 11:4304
Trobro S, Aqvist J (2005) Mechanism of peptide bond synthesis on the ribosome. Proc Natl Acad Sci U S A 102:12395–12400
Voorhees RM, Weixlbaumer A, Loakes D et al (2009) Insights into substrate stabilization from snapshots of the peptidyl transferase center of the intact 70S ribosome. Nat Struct Mol Biol 16:528–533
Shimizu Y, Inoue A, Tomari Y et al (2001) Cell-free translation reconstituted with purified components. Nat Biotechnol 19:751–755
Charlton A, Zachariou M (2008) Immobilized metal ion affinity chromatography of histidine-tagged fusion proteins. Methods Mol Biol 421:137–149
Block H, Maertens B, Spriestersbach A et al (2009) Immobilized-metal affinity chromatography (IMAC): a review. Methods Enzymol 463:439–473
Martinis SA, Schimmel P (1992) Enzymatic aminoacylation of sequence-specific RNA minihelices and hybrid duplexes with methionine. Proc Natl Acad Sci U S A 89:65–69
Putz J, Wientges J, Sissler M et al (1997) Rapid selection of aminoacyl-tRNAs based on biotinylation of alpha-NH2 group of charged amino acids. Nucleic Acids Res 25:1862–1863
Ohta A, Murakami H, Higashimura E et al (2007) Synthesis of polyester by means of genetic code reprogramming. Chem Biol 14:1315–1322
Terasaka N, Hayashi G, Katoh T et al (2014) An orthogonal ribosome-tRNA pair via engineering of the peptidyl transferase center. Nat Chem Biol 10:555–557
Katoh T, Iwane Y, Suga H (2017) Logical engineering of D-arm and T-stem of tRNA that enhances d-amino acid incorporation. Nucleic Acids Res 45:12601–12610
Goto Y, Iseki M, Hitomi A et al (2013) Nonstandard peptide expression under the genetic code consisting of reprogrammed dual sense codons. ACS Chem Biol 8:2630–2634
Olins PO, Devine CS, Rangwala SH et al (1988) The T7 phage gene 10 leader RNA, a ribosome-binding site that dramatically enhances the expression of foreign genes in Escherichia coli. Gene 73:227–235
Sako Y, Goto Y, Murakami H et al (2008) Ribosomal synthesis of peptidase-resistant peptides closed by a nonreducible inter-side-chain bond. ACS Chem Biol 3:241–249
Acknowledgments
We thank H. Tsutsumi and T. Kuroda for their major contributions to the development of the methods presented in this chapter. We also appreciate the financial supports from the Japan Society for the Promotion of Science, KAKENHI (JP16H06444 to H.S. and Y.G.; JP20H05618 to H.S.; JP17H04762, JP18H04382, JP19K22243, JP20H02866 to Y.G.).
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Goto, Y., Suga, H. (2023). Ribosomal Synthesis of Peptides Bearing Noncanonical Backbone Structures via Chemical Posttranslational Modifications. In: Burkart, M., Ishikawa, F. (eds) Non-Ribosomal Peptide Biosynthesis and Engineering. Methods in Molecular Biology, vol 2670. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3214-7_13
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3214-7_13
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3213-0
Online ISBN: 978-1-0716-3214-7
eBook Packages: Springer Protocols