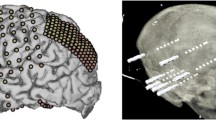
Abstract
The neurophysiological mechanisms that enable cognitive functions are typically studied noninvasively in humans using scalp magnetic resonance imaging (MRI) and magneto- or electroencephalography (M/EEG) or invasively in rodents or nonhuman primates. Intracranial EEG as recorded in pharmacoresistant epilepsy patients who undergo evaluation for resective epilepsy surgery provides a unique approach to bridge the gap between noninvasive in humans and invasive studies in rodents and nonhuman primates. In recent years, iEEG has provided important insights into the functional architecture of human cognitive systems. Here, the principles of successfully conducting intracranial experiments in humans are discussed, with a particular focus on implementing recording setups and analyzing the data. The high spatiotemporal resolution of iEEG provides a number of opportunities and challenges, which are outlined alongside potential solutions. Collectively, intracranial cognitive neurophysiology is a rapidly progressing field providing key insights into the organization of human cognitive systems.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
D’Esposito M (2010) Why methods matter in the study of the biological basis of the mind: a behavioral neurologist’s perspective. In: Reuter-Lorenz P, Baynes K, Mangun GR, Phelps EA (eds) The Cognitive neuroscience of mind: a tribute to Michael Gazzaniga. MIT Press, Cambridge, London, pp 203–221
Szczepanski SM, Knight RT (2014) Insights into human behavior from lesions to the prefrontal cortex. Neuron 83:1002–1018
Parvizi J, Kastner S (2018) Promises and limitations of human intracranial electroencephalography. Nat Neurosci 21:474–483
Corkin S (2002) What’s new with the amnesic patient H.M.? Nat Rev Neurosci 3:153
Squire LR (2009) The legacy of patient H.M. for neuroscience. Neuron 61:6–9
Vaidya AR, Pujara MS, Petrides M et al (2019) Lesion studies in contemporary neuroscience. Trends Cogn Sci (Regul Ed) 23:653–671
Helfrich RF, Knight RT (2016) Oscillatory dynamics of prefrontal cognitive control. Trends Cogn Sci (Regul Ed) 20:916–930
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24:167–202
Engel AK, Fries P, Singer W (2001) Dynamic predictions: oscillations and synchrony in top-down processing. Nat Rev Neurosci 2:704–716
Singer W, Gray CM (1995) Visual feature integration and the temporal correlation hypothesis. Annu Rev Neurosci 18:555–586
Buzsaki G (2006) Rhythms of the brain. Oxford University Press
Varela F, Lachaux JP, Rodriguez E et al (2001) The brainweb: phase synchronization and large-scale integration. Nat Rev Neurosci 2:229–239
Buzsáki G, Anastassiou CA, Koch C (2012) The origin of extracellular fields and currents--EEG, ECoG, LFP and spikes. Nat Rev Neurosci 13:407–420
Pesaran B, Vinck M, Einevoll GT et al (2018) Investigating large-scale brain dynamics using field potential recordings: analysis and interpretation. Nat Neurosci 21:903–919
Engel AK, Moll CKE, Fried I et al (2005) Invasive recordings from the human brain: clinical insights and beyond. Nat Rev Neurosci 6:35–47
Bronstein JM, Tagliati M, Alterman RL et al (2011) Deep brain stimulation for Parkinson disease: an expert consensus and review of key issues. Arch Neurol 68:165–165
Weiss D, Klotz R, Govindan RB et al (2015) Subthalamic stimulation modulates cortical motor network activity and synchronization in Parkinson’s disease. Brain 138:679–693
Zavala BA, Jang AI, Zaghloul KA (2017) Human subthalamic nucleus activity during non-motor decision making. eLife 6. https://elifesciences.org/articles/31007
Helfrich RF, Fiebelkorn IC, Szczepanski SM et al (2018) Neural mechanisms of sustained attention are rhythmic. Neuron 99:854–865.e5
Helfrich RF, Lendner JD, Mander BA et al (2019) Bidirectional prefrontal-hippocampal dynamics organize information transfer during sleep in humans. Nat Commun 10:3572
Mayberg HS, Lozano AM, Voon V et al (2005) Deep brain stimulation for treatment-resistant depression. Neuron 45:651–660
van Westen M, Rietveld E, Figee M et al (2015) Clinical outcome and mechanisms of deep brain stimulation for obsessive-compulsive disorder. Curr Behav Neurosci Rep 2:41–48
Crone NE, Miglioretti DL, Gordon B et al (1998) Functional mapping of human sensorimotor cortex with electrocorticographic spectral analysis. II. Event-related synchronization in the gamma band. Brain 121(Pt 12):2301–2315
Edwards E, Soltani M, Deouell LY et al (2005) High gamma activity in response to deviant auditory stimuli recorded directly from human cortex. J Neurophysiol 94:4269–4280
Ray S, Maunsell JHR (2011) Different origins of gamma rhythm and high-gamma activity in macaque visual cortex. PLoS Biol 9:e1000610
Flinker A, Korzeniewska A, Shestyuk AY et al (2015) Redefining the role of Broca’s area in speech. Proc Natl Acad Sci U S A 112:2871–2875
Dubey A, Ray S (2019) Cortical electrocorticogram (ECoG) is a local signal. J Neurosci 39:4299–4311
Leszczyński M, Barczak A, Kajikawa Y et al (2020) Dissociation of broadband high-frequency activity and neuronal firing in the neocortex. Sci Adv 6:eabb0977
Holdgraf CR, de Heer W, Pasley B et al (2016) Rapid tuning shifts in human auditory cortex enhance speech intelligibility. Nat Commun 7:13654
Li G, Jiang S, Paraskevopoulou SE et al (2018) Optimal referencing for stereo-electroencephalographic (SEEG) recordings. NeuroImage 183:327–335
Kanth ST, Ray S (2020) Electrocorticogram (ECoG) is highly informative in primate visual cortex. J Neurosci 40:2430–2444
Johnson EL, Knight RT (2015) Intracranial recordings and human memory. Curr Opin Neurobiol 31:18–25
Eichenbaum H (2017) Prefrontal-hippocampal interactions in episodic memory. Nat Rev Neurosci 18:547–558
Zheng J, Anderson KL, Leal SL et al (2017) Amygdala-hippocampal dynamics during salient information processing. Nat Commun 8:14413
Zhang H, Fell J, Axmacher N (2018) Electrophysiological mechanisms of human memory consolidation. Nat Commun 9:4103
Vaz AP, Inati SK, Brunel N et al (2019) Coupled ripple oscillations between the medial temporal lobe and neocortex retrieve human memory. Science 363:975–978
Rutishauser U (2019) Testing models of human declarative memory at the single-neuron level. Trend Cog Sci 23:510–524
Fried I, Rutishauser U, Cerf M et al (2014) Single neuron studies of the human brain: probing cognition. MIT Press
Cash SS, Hochberg LR (2015) The emergence of single neurons in clinical neurology. Neuron 86:79–91
Groves DA, Brown VJ (2005) Vagal nerve stimulation: a review of its applications and potential mechanisms that mediate its clinical effects. Neurosci Biobehav Rev 29:493–500
Boëx C, Seeck M, Vulliémoz S et al (2011) Chronic deep brain stimulation in mesial temporal lobe epilepsy. Seizure 20:485–490
Miatton M, Van Roost D, Thiery E et al (2011) The cognitive effects of amygdalohippocampal deep brain stimulation in patients with temporal lobe epilepsy. Epilepsy Behav 22:759–764
Carrette S, Boon P, Sprengers M et al (2015) Responsive neurostimulation in epilepsy. Expert Rev Neurother 15:1445–1454
Noachtar S, Borggraefe I (2009) Epilepsy surgery: a critical review. Epilepsy Behav 15:66–72
Devinsky O, Vezzani A, O’Brien TJ et al (2018) Epilepsy 4:1–24
Rao VR, Lowenstein DH (2015) Epilepsy. Curr Biol 25:R742–R746
Gelinas JN, Khodagholy D, Thesen T et al (2016) Interictal epileptiform discharges induce hippocampal-cortical coupling in temporal lobe epilepsy. Nat Med 22:641–648
Meisel C (2020) Antiepileptic drugs induce subcritical dynamics in human cortical networks. Proc Natl Acad Sci U S A 117:11118–11125
Talairach J, Bancaud J, Bonis A et al (1962) Functional stereotaxic exploration of epilepsy. Confin Neruol 22:328–331
Tandon N, Tong BA, Friedman ER et al (2019) Analysis of morbidity and outcomes associated with use of subdural grids vs stereoelectroencephalography in patients with intractable epilepsy. JAMA Neurol 76:672–681
Schalk G, McFarland DJ, Hinterberger T et al (2004) BCI2000: a general-purpose brain-computer interface (BCI) system. IEEE Trans Biomed Eng 51:1034–1043
Kam JWY, Szczepanski SM, Canolty RT, et al (2016) Differential sources for 2 neural signatures of target detection: an electrocorticography study. Cereb Cortex 28(1):9–20
Gray CM, König P, Engel AK et al (1989) Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature 338:334–337
Miller KJ, Sorensen LB, Ojemann JG et al (2009) Power-law scaling in the brain surface electric potential. PLoS Comput Biol 5:e1000609
Miller KJ, Zanos S, Fetz EE et al (2009) Decoupling the cortical power spectrum reveals real-time representation of individual finger movements in humans. J Neurosci 29:3132–3137
Canolty RT, Edwards E, Dalal SS et al (2006) High gamma power is phase-locked to theta oscillations in human neocortex. Science 313:1626–1628
Brunet N, Vinck M, Bosman CA et al (2014) Gamma or no gamma, that is the question. Trends Cogn Sci (Regul Ed) 18:507–509
Hermes D, Miller KJ, Wandell BA et al (2015) Gamma oscillations in visual cortex: the stimulus matters. Trends Cogn Sci (Regul Ed) 19:57–58
Ray S, Maunsell JHR (2015) Do gamma oscillations play a role in cerebral cortex? Trends Cogn Sci (Regul Ed) 19:78–85
Bruns A (2004) Fourier-, Hilbert- and wavelet-based signal analysis: are they really different approaches? J Neurosci Methods 137:321–332
Foster BL, Rangarajan V, Shirer WR et al (2015) Intrinsic and task-dependent coupling of neuronal population activity in human parietal cortex. Neuron 86:578–590
Haller M, Case J, Crone NE et al (2018) Persistent neuronal activity in human prefrontal cortex links perception and action. Nat Human Behav 2:80–91
Voytek B, Kayser AS, Badre D et al (2015) Oscillatory dynamics coordinating human frontal networks in support of goal maintenance. Nat Neurosci 18:1318–1324
Donoghue T, Haller M, Peterson EJ et al (2020) Parameterizing neural power spectra into periodic and aperiodic components. Nat Neurosci 23:1655–1665
Bastos AM, Schoffelen J-M (2015) A tutorial review of functional connectivity analysis methods and their interpretational pitfalls. Front Syst Neurosci 9:175
Aru J, Aru J, Priesemann V et al (2015) Untangling cross-frequency coupling in neuroscience. Curr Opin Neurobiol 31:51–61
Canolty RT, Knight RT (2010) The functional role of cross-frequency coupling. Trends Cogn Sci (Regul Ed) 14:506–515
Gerber EM, Sadeh B, Ward A et al (2016) Non-sinusoidal activity can produce cross-frequency coupling in cortical signals in the absence of functional interaction between neural sources. PLoS One 11:e0167351
Tort ABL, Kramer MA, Thorn C et al (2008) Dynamic cross-frequency couplings of local field potential oscillations in rat striatum and hippocampus during performance of a T-maze task. Proc Natl Acad Sci U S A 105:20517–20522
Quian Quiroga R, Panzeri S (2009) Extracting information from neuronal populations: information theory and decoding approaches. Nat Rev Neurosci 10:173–185
Vyas S, Golub MD, Sussillo D et al (2020) Computation through neural population dynamics. Annu Rev Neurosci 43:249–275
Stokes MG, Kusunoki M, Sigala N et al (2013) Dynamic coding for cognitive control in prefrontal cortex. Neuron 78:364–375
Stokes MG (2015) “Activity-silent” working memory in prefrontal cortex: a dynamic coding framework. Trends Cogn Sci (Regul Ed) 19:394–405
Cunningham JP, Byron MY (2014) Dimensionality reduction for large-scale neural recordings. Nat Neurosci 17:1500–1509
Kobak D, Brendel W, Constantinidis C et al (2016) Demixed principal component analysis of neural population data. eLife 5:e10989
Kriegeskorte N, Mur M, Bandettini P (2008) Representational similarity analysis - connecting the branches of systems neuroscience. Front Syst Neurosci 2:4
Timme NM, Lapish C (2018) A tutorial for information theory in neuroscience. eNeuro 11:5(3):ENEURO.0052-18.2018. doi: 10.1523/ENEURO.0052-18.2018. PMID: 30211307; PMCID: PMC6131830
Borchers S, Himmelbach M, Logothetis N et al (2012) Direct electrical stimulation of human cortex—the gold standard for mapping brain functions? Nat Rev Neurosci 13:63
Foster BL, Parvizi J (2017) Direct cortical stimulation of human posteromedial cortex. Neurology 88:685–691
Fox KCR, Shi L, Baek S et al (2020) Intrinsic network architecture predicts the effects elicited by intracranial electrical stimulation of the human brain. Nat Hum Behav 4:1039–1052
Suthana N, Haneef Z, Stern J et al (2012) Memory enhancement and deep-brain stimulation of the entorhinal area. N Engl J Med 366:502–510
Titiz AS, Hill MRH, Mankin EA et al (2017) Theta-burst microstimulation in the human entorhinal area improves memory specificity. eLife 6:e29515
Hermes D, Miller KJ, Noordmans HJ et al (2010) Automated electrocorticographic electrode localization on individually rendered brain surfaces. J Neurosci Methods 185:293–298
Groppe DM, Bickel S, Dykstra AR et al (2017) iELVis: an open source MATLAB toolbox for localizing and visualizing human intracranial electrode data. J Neurosci Methods 281:40–48
Blenkmann AO, Phillips HN, Princich JP et al (2017) iElectrodes: a comprehensive open-source toolbox for depth and subdural grid electrode localization. Front Neuroinform 11:14
Dykstra AR, Chan AM, Quinn BT et al (2012) Individualized localization and cortical surface-based registration of intracranial electrodes. NeuroImage 59:3563–3570
Stolk A, Griffin S, van der Meij R et al (2018) Integrated analysis of anatomical and electrophysiological human intracranial data. Nat Protoc 13(7):1699–1723
Oostenveld R, Fries P, Maris E et al (2011) FieldTrip: open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intell Neurosci 2011:156869
Holdgraf C, Appelhoff S, Bickel S et al (2019) iEEG-BIDS, extending the Brain Imaging Data Structure specification to human intracranial electrophysiology. Sci Data 6:102
Faraut MC, Carlson AA, Sullivan S et al (2018) Dataset of human medial temporal lobe single neuron activity during declarative memory encoding and recognition. Sci Data 5:180010
Boran E, Fedele T, Steiner A et al (2020) Dataset of human medial temporal lobe neurons, scalp and intracranial EEG during a verbal working memory task. Sci Data 7:1–7
Johnson EL, Helfrich RF (2016) How brain cells make memories. Front Young Minds 4
Ram B, Helfrich RF Waves of percetion. Front Young Minds 5
Chiong W, Leonard MK, Chang EF (2018) Neurosurgical patients as human research subjects: ethical considerations in intracranial electrophysiology research. Neurosurgery 83:29–37
Ammanuel SG, Kleen JK, Leonard MK et al (2020) Interictal epileptiform discharges and the quality of human intracranial neurophysiology data. Front Hum Neurosci 14:44
Buzsáki G (2015) Hippocampal sharp wave-ripple: a cognitive biomarker for episodic memory and planning. Hippocampus 25:1073–1188
Szczepanski SM, Crone NE, Kuperman RA et al (2014) Dynamic changes in phase-amplitude coupling facilitate spatial attention control in fronto-parietal cortex. PLoS Biol 12:e1001936
Saxe R, Brett M, Kanwisher N (2006) Divide and conquer: a defense of functional localizers. NeuroImage 30:1088–1096
Anumanchipalli GK, Chartier J, Chang EF (2019) Speech synthesis from neural decoding of spoken sentences. Nature 568:493–498
Pasley BN, David SV, Mesgarani N et al (2012) Reconstructing speech from human auditory cortex. PLoS Biol 10:e1001251
Oganian Y, Chang EF (2018) A speech envelope landmark for syllable encoding in human superior temporal gyrus. Sci Adv 5:388280
Fiebelkorn IC, Pinsk MA, Kastner S (2018) A dynamic interplay within the Frontoparietal network underlies rhythmic spatial attention. Neuron 99:842–853.e8
Acknowledgments
This work was supported by the German Research Foundation (DFG, HE 8329/2-1), the Hertie Foundation (Hertie Network of Excellence in Clinical Neuroscience), and the Baden-Württemberg Foundation.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Helfrich, R.F. (2022). Human Intracranial Cognitive Neurophysiology. In: Vertes, R.P., Allen, T. (eds) Electrophysiological Recording Techniques. Neuromethods, vol 192. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2631-3_10
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2631-3_10
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2630-6
Online ISBN: 978-1-0716-2631-3
eBook Packages: Springer Protocols