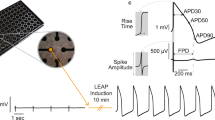
Abstract
Induced pluripotent stem cell (iPSC)-derived cardiomyocytes (iPSC-CMs) have been shown to have great potential to play a key role in investigating cardiac diseases in vitro. Multielectrode array (MEA) system is sometimes preferable to patch-clamp in electrophysiological experiments in terms of several advantages. Here we show our protocol of electrophysiological examinations using MEA.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bellin M, Casini S, Davis RP et al (2013) Isogenic human pluripotent stem cell pairs reveal the role of a KCNH2 mutation in long-QT syndrome. EMBO J 32:3161–3175
Moretti A, Bellin M, Welling A, Jung CB, Lam JT, Bott-Flügel L, Dorn T, Goedel A, Höhnke C, Hofmann F, Seyfarth M (2010) Patient-specific induced pluripotent stem-cell models for long-QT syndrome. N Engl J Med 363(15):1397–1409
Yazawa M, Hsueh B, Jia X et al (2011) Using induced pluripotent stem cells to investigate cardiac phenotypes in Timothy syndrome. Nature 471:230–236
Limpitikul WB, Dick IE, Tester DJ et al (2017) A precision medicine approach to the Rescue of Function on malignant Calmodulinopathic long-QT syndrome. Circ Res 120:39–48
Gibson JK, Yue Y, Bronson J et al (2014) Human stem cell-derived cardiomyocytes detect drug-mediated changes in action potentials and ion currents. J Pharmacol Toxicol Methods 70:255–267
Peng S, Lacerda AE, Kirsch GE et al (2010) The action potential and comparative pharmacology of stem cell-derived human cardiomyocytes. J Pharmacol Toxicol Methods 61:277–286
Yamazaki K, Hihara T, Taniguchi T et al (2012) A novel method of selecting human embryonic stem cell-derived cardiomyocyte clusters for assessment of potential to influence QT interval. Toxicol In Vitro 26:335–342
Liang P, Sallam K, Wu H et al (2016) Patient-specific and genome-edited induced pluripotent stem cell–derived cardiomyocytes elucidate single-cell phenotype of Brugada syndrome. J Am Coll Cardiol 68:2086–2096
Jung CB, Moretti A, Mederos y Schnitzler M et al (2012) Dantrolene rescues arrhythmogenic RYR2 defect in a patient-specific stem cell model of catecholaminergic polymorphic ventricular tachycardia. EMBO Mol Med 4:180–191
Novak A, Barad L, Zeevi-Levin N et al (2012) Cardiomyocytes generated from CPVT D307H patients are arrhythmogenic in response to β-adrenergic stimulation. J Cell Mol Med 16:468–482
Bellin M, Marchetto MC, Gage FH et al (2012) Induced pluripotent stem cells: the new patient? Nat Rev Mol Cell Biol 13:713–726
Sinnecker D, Laugwitz KL, Moretti A (2014) Induced pluripotent stem cell-derived cardiomyocytes for drug development and toxicity testing. Pharmacol Ther 143:246–252
Terrenoire C, Wang K, Chan Tung KW et al (2013) Induced pluripotent stem cells used to reveal drug actions in a long QT syndrome family with complex genetics. J Gen Physiol 141:61–72
Yoshinaga D, Baba S, Makiyama T et al (2019) Phenotype-based high-throughput classification of long QT syndrome subtypes using human induced pluripotent stem cells. Stem Cell Rep 13:394–404
Wuriyanghai Y, Makiyama T, Sasaki K et al (2018) Complex aberrant splicing in the induced pluripotent stem cell–derived cardiomyocytes from a patient with long QT syndrome carrying KCNQ1-A344Aspl mutation. Hear Rhythm 15:1566–1574
Abi-Gerges N, Pointon A, Oldman KL et al (2017) Assessment of extracellular field potential and Ca2+ transient signals for early QT/pro-arrhythmia detection using human induced pluripotent stem cell-derived cardiomyocytes. J Pharmacol Toxicol Methods 83:1–15
del Álamo JC, Lemons D, Serrano R et al (2016) High throughput physiological screening of iPSC-derived cardiomyocytes for drug development. Biochim Biophys Acta, Mol Cell Res 1863:1717–1727
Blinova K, Stohlman J, Vicente J et al (2017) Comprehensive translational assessment of human-induced pluripotent stem cell derived cardiomyocytes for evaluating drug-induced arrhythmias. Toxicol Sci 155:234–247
Meyer T, Boven KH, Günther E et al (2004) Micro-electrode arrays in cardiac safety pharmacology: a novel tool to study QT interval prolongation. Drug Saf 27(11):763–772
Halbach MD, Egert U, Hescheler J et al (2003) Estimation of action potential changes from field potential recordings in multicellular mouse cardiac myocyte cultures. Cell Physiol Biochem 13:271–284
Asakura K, Hayashi S, Ojima A et al (2015) Improvement of acquisition and analysis methods in multi-electrode array experiments with {iPS} cell-derived cardiomyocytes. J Pharmacol Toxicol 75:17–26
Kawatou M, Masumoto H, Fukushima H et al (2017) Modelling torsade de pointes arrhythmias in vitro in 3D human iPS cell-engineered heart tissue. Nat Commun 8:1–11
Izumi-Nakaseko H, Kanda Y, Nakamura Y et al (2017) Development of correction formula for field potential duration of human induced pluripotent stem cell-derived cardiomyocytes sheets. J Pharmacol Sci 135:44–50
Acknowledgment
This work was supported by JSPS KAKENHI (Grant Number JP20K17147 to D.Y., JP19K17595 to Y.W., and JP16K09499, JP19K08538 to T.M.).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Yoshinaga, D., Wuriyanghai, Y., Makiyama, T. (2021). Multielectrode Array Assays Using Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes. In: Yoshida, Y. (eds) Pluripotent Stem-Cell Derived Cardiomyocytes. Methods in Molecular Biology, vol 2320. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1484-6_12
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1484-6_12
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1483-9
Online ISBN: 978-1-0716-1484-6
eBook Packages: Springer Protocols