Abstract
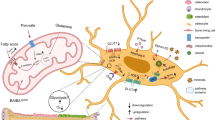
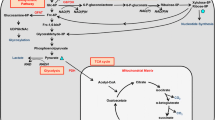
Measuring cellular metabolism accurately is necessary to understand bioenergetic pathways in cells. The major ATP generating pathways in cells are oxidative phosphorylation and glycolysis. We have recently analyzed and published bioenergetic pathways active in osteoblasts undergoing differentiation in response to various substrates. Based on those studies, here we provide step-by-step procedures to isolate, culture, plate and run a seahorse assay for measuring cellular metabolism. Furthermore, we provide an example of oxygen consumption and extracellular acidification rate traces obtained from MC3T3E1-C4 cells using the XFe96 seahorse analyzer. One of the limitations of studying bioenergetics in bone cells is the current lack of techniques to analyze bioenergetics in vivo in live animals. There are currently techniques that have been developed using third harmonic generation to study osteocytes using three-photon microscopy along with metabolic changes using endogenous two-photon excited fluorescence. However, these sophisticated techniques are not widely available. The relative ease with which one can obtain data pertaining to metabolic parameters using the XF technology makes it a very attractive technique to utilize on a monolayer of adherent cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Karsenty G, Oury F (2012) Biology without walls: the novel endocrinology of bone. Annu Rev Physiol 74(1):87–105. https://doi.org/10.1146/annurev-physiol-020911-153233
Lee WC, Guntur AR, Long F, Rosen CJ (2017) Energy metabolism of the osteoblast: implications for osteoporosis. Endocr Rev 38(3):255–266. https://doi.org/10.1210/er.2017-00064
Zhang J, Zhang Q (2019) Using seahorse machine to measure OCR and ECAR in cancer cells. Methods Mol Biol (Clifton, NJ) 1928:353–363. Epub 2019/02/07. https://doi.org/10.1007/978-1-4939-9027-6_18
Divakaruni AS, Paradyse A, Ferrick DA, Murphy AN, Jastroch M (2014) Analysis and interpretation of microplate-based oxygen consumption and pH data. Methods Enzymol 547:309–354. Epub 2014/11/25. https://doi.org/10.1016/b978-0-12-801415-8.00016-3
Mookerjee SA, Goncalves RLS, Gerencser AA, Nicholls DG, Brand MD (2015) The contributions of respiration and glycolysis to extracellular acid production. Biochim Biophys Acta 1847(2):171–181. Epub 2014/12/03. https://doi.org/10.1016/j.bbabio.2014.10.005
Guntur AR, Gerencser AA, Le PT, DeMambro VE, Bornstein SA, Mookerjee SA, Maridas DE, Clemmons DE, Brand MD, Rosen CJ (2018) Osteoblast-like MC3T3-E1 cells prefer glycolysis for ATP production but adipocyte-like 3T3-L1 cells prefer oxidative phosphorylation. J Bone Miner Res 33(6):1052–1065. https://doi.org/10.1002/jbmr.3390
Wang C, Silverman RM, Shen J, O'Keefe RJ (2018) Distinct metabolic programs induced by TGF-beta1 and BMP2 in human articular chondrocytes with osteoarthritis. J Orthop Translat 12:66–73. https://doi.org/10.1016/j.jot.2017.12.004
Guntur AR, Le PT, Farber CR, Rosen CJ (2014) Bioenergetics during calvarial osteoblast differentiation reflect strain differences in bone mass. Endocrinology 155(5):1589–1595. https://doi.org/10.1210/en.2013-1974
Shum LC, White NS, Mills BN, Bentley KL, Eliseev RA (2016) Energy metabolism in mesenchymal stem cells during osteogenic differentiation. Stem Cells Dev 25(2):114–122. https://doi.org/10.1089/scd.2015.0193
Liu Z, Solesio ME, Schaffler MB, Frikha-Benayed D, Rosen CJ, Werner H, Kopchick JJ, Pavlov EV, Abramov AY, Yakar S (2019) Mitochondrial function is compromised in cortical bone osteocytes of Long-lived growth hormone receptor null mice. J Bone Miner Res Off J Am Soc Bone Miner Res 34(1):106–122. Epub 2018/09/15. https://doi.org/10.1002/jbmr.3573
Sun N, Uda Y, Azab E, Kochen A, Santos R, Shi C, Kobayashi T, Wein MN, Divieti PP (2019) Effects of histone deacetylase inhibitor Scriptaid and parathyroid hormone on osteocyte functions and metabolism. J Biol Chem 294(25):9722–9733. https://doi.org/10.1074/jbc.RA118.007312
Arnett TR, Orriss IR (2018) Metabolic properties of the osteoclast. Bone 115:25–30. Epub 2017/12/25. https://doi.org/10.1016/j.bone.2017.12.021
Maridas DE, Rendina-Ruedy E, Le PT, Rosen CJ (2018) Isolation, culture, and differentiation of bone marrow stromal cells and osteoclast progenitors from mice. J Vis Exp (131):56750. https://doi.org/10.3791/56750
Acknowledgments
This work was funded by NIGMS to A.R.G. through P20GM121301, Phase I: Mesenchymal and Neural Regulation of Metabolic Networks, Lucy Liaw, PhD, Program Director.
Disclosure Statement: The authors have nothing to disclose
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Tian, L., Rosen, C.J., Guntur, A.R. (2021). Mitochondrial Function and Metabolism of Cultured Skeletal Cells. In: Hilton, M.J. (eds) Skeletal Development and Repair. Methods in Molecular Biology, vol 2230. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1028-2_27
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1028-2_27
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1027-5
Online ISBN: 978-1-0716-1028-2
eBook Packages: Springer Protocols