Abstract
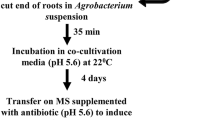
Chickpea is a major protein source in low socio-economic classes and cultivated in marginal soil without fertilizer or irrigation. As a result of its root nodule formation capacity chickpea can directly use atmospheric nitrogen. Chickpea is recalcitrant to stable transformation, particularly root regeneration efficiency of chickpea is low. The composite plant-based system with a non-transformed shoot and transformed root is particularly important for root biologist and this approach has already been used successfully for root nodule symbiosis, arbuscular mycorrhizal symbiosis, and other root-related studies. Use of fluorescent marker-based approach can accurately identify the transformed root from its non-transgenic counterpart. RNAi-based gene knockout, overexpression of genes, promoter GUS analysis to understand tissue specific expression and localization of protein can be achieved using the hairy root-based system. We have already published a hairy root-based transformation and composite plant regeneration protocol of chickpea. Here we are describing the recent modification that we have made to increase the transformation frequency and nodule morphology. Further, we have developed a pouch based artificial system, large number of plants can be scored for its nodule developmental phenotype, by using this system.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jain M, Misra G, Patel RK, Priya P, Jhanwar S, Khan AW et al (2013) A draft genome sequence of the pulse crop chickpea (Cicer arietinum L.). Plant J 74:715–729
Mandal D, Sinharoy S (2019) A toolbox for nodule development studies in chickpea: a hairy-root transformation protocol and an efficient laboratory strain of Mesorhizobium sp. Mol Plant-Microbe Interact 32:367–378
Montiel J, Downie JA, Farkas A, Bihari P, Herczeg R, Balint B et al (2017) Morphotype of bacteroids in different legumes correlates with the number and type of symbiotic NCR peptides. Proc Natl Acad Sci U S A 114:5041–5046
Varshney RK, Thudi M, Roorkiwal M, He W, Upadhyaya HD, Yang W et al (2019) Resequencing of 429 chickpea accessions from 45 countries provides insights into genome diversity, domestication and agronomic traits. Nat Genet 51:857–864
Pandey A, Chakraborty S, Chakraborty N (2018) Nuclear proteome: isolation of intact nuclei, extraction of nuclear proteins, and 2-DE analysis. Methods Mol Biol 1696:41–55
Ramalingam A, Kudapa H, Pazhamala LT, Weckwerth W, Varshney RK (2015) Proteomics and metabolomics: two emerging areas for legume improvement. Front Plant Sci 6:1116
Varshney RK, Song C, Saxena RK, Azam S, Yu S, Sharpe AG et al (2013) Draft genome sequence of chickpea (Cicer arietinum) provides a resource for trait improvement. Nat Biotechnol 31:240–246
Georgiev MI, Agostini E, Ludwig-Muller J, Xu J (2012) Genetically transformed roots: from plant disease to biotechnological resource. Trends Biotechnol 30:528–537
Stougaard J, Petersen TE, Marcker KA (1987) Expression of a complete soybean leghemoglobin gene in root nodules of transgenic Lotus corniculatus. Proc Natl Acad Sci U S A 84:5754–5757
Limpens E, Ramos J, Franken C, Raz V, Compaan B, Franssen H et al (2004) RNA interference in agrobacterium rhizogenes-transformed roots of Arabidopsis and Medicago truncatula. J Exp Bot 55:983–992
Sinharoy S, Pislariu CI, Udvardi MK (2015) A high-throughput RNA interference (RNAi)-based approach using hairy roots for the study of plant-rhizobia interactions. Methods Mol Biol 1287:159–178
Kumagai H, Kouchi H (2003) Gene silencing by expression of hairpin RNA in Lotus japonicus roots and root nodules. Mol Plant-Microbe Interact 16:663–668
Kereszt A, Li D, Indrasumunar A, Nguyen CD, Nontachaiyapoom S, Kinkema M et al (2007) Agrobacterium rhizogenes-mediated transformation of soybean to study root biology. Nat Protoc 2:948–952
Boisson-Dernier A, Chabaud M, Garcia F, Becard G, Rosenberg C, Barker DG (2001) Agrobacterium rhizogenes-transformed roots of Medicago truncatula for the study of nitrogen-fixing and endomycorrhizal symbiotic associations. Mol Plant-Microbe Interact 14:695–700
Diaz CL, Spaink HP, Kijne JW (2000) Heterologous rhizobial lipochitin oligosaccharides and chitin oligomers induce cortical cell divisions in red clover roots, transformed with the pea lectin gene. Mol Plant-Microbe Interact 13:268–276
Stiller J, Martirani L, Tuppale S, Chian R-J, Chiurazzi M, Gresshoff PM (1997) High frequency transformation and regeneration of transgenic plants in the model legume Lotus japonicus. J Exp Bot 48:1357–1365
Diouf D, Gherbi H, Prin Y, Franche C, Duhoux E, Bogusz D (1995) Hairy root nodulation of Casuarina glauca: a system for the study of symbiotic gene expression in an actinorhizal tree. Mol Plant-Microbe Interact 8:532–537
Quandt HJ, Puhler A, Broer I (1993) Transgenic root nodules of Vicia hirsuta. A fast and efficient system for the study of gene expression in indeterminate-type nodules. Mol Plant-Microbe Interact 6:699–703
Lee NG, Stein B, Suzuki H, Verma DP (1993) Expression of antisense nodulin-35 RNA in Vigna aconitifolia transgenic root nodules retards peroxisome development and affects nitrogen availability to the plant. Plant J 3:599–606
Cheon CI, Lee NG, Siddique ABM, Bal AK, Verma DPS (1993) Roles of plant homologs of Rabi p and Rab7p in the biogenesis of the peribacteroid membrane, a subcellular compartment formed de novo during root nodule symbiosis. EMBO J 12:4125–4135
Díaz C, Melchers LS, Hooykaas PJJ, Lugtenberg BJJ, Kijne JW (1989) Root lectin as a determinant of host-plant specificity in the rhizobium-legume symbiosis. Nature 338:579–558
Mellor KE, Hoffman AM, Timko MP (2012) Use of ex vitro composite plants to study the interaction of cowpea (Vigna unguiculata L.) with the root parasitic angiosperm Striga gesnerioides. Plant Methods 8:22
Imanishi L, Vayssieres A, Franche C, Bogusz D, Wall L, Svistoonoff S (2011) Transformed hairy roots of Discaria trinervis: a valuable tool for studying actinorhizal symbiosis in the context of intercellular infection. Mol Plant-Microbe Interact 24:1317–1324
Clemow SR, Clairmont L, Madsen LH, Guinel FC (2011) Reproducible hairy root transformation and spot-inoculation methods to study root symbioses of pea. Plant Methods 7:46
Bonaldi K, Gherbi H, Franche C, Bastien G, Fardoux J, Barker D et al (2010) The nod factor-independent symbiotic signaling pathway: development of agrobacterium rhizogenes-mediated transformation for the legume Aeschynomene indica. Mol Plant-Microbe Interact 23:1537–1544
Sinharoy S, Saha S, Chaudhury SR, Dasgupta M (2009) Transformed hairy roots of Arachis hypogea: a tool for studying root nodule symbiosis in a non-infection thread legume of the Aeschynomeneae tribe. Mol Plant-Microbe Interact 22:132–142
Estrada-Navarrete G, Alvarado-Affantranger X, Olivares JE, Diaz-Camino C, Santana O, Murillo E et al (2006) Agrobacterium rhizogenes transformation of the Phaseolus spp.: a tool for functional genomics. Mol Plant-Microbe Interact 19:1385–1393
Van-de-Velde W, Mergeay J, Holsters M, Goormachtig S (2003) Agrobacterium rhizogenes-mediated transformation of Sesbania rostrata. Plant Sci 165:1281–1288
Ron M, Kajala K, Pauluzzi G, Wang D, Reynoso MA, Zumstein K et al (2014) Hairy root transformation using agrobacterium rhizogenes as a tool for exploring cell type-specific gene expression and function using tomato as a model. Plant Physiol 166:455–469
Hofgen R, Willmitzer L (1988) Storage of competent cells for agrobacterium transformation. Nucleic Acids Res 16:9877
Auriac MC, Timmers AC (2007) Nodulation studies in the model legume Medicago truncatula: advantages of using the constitutive EF1alpha promoter and limitations in detecting fluorescent reporter proteins in nodule tissues. Mol Plant-Microbe Interact 20:1040–1047
Karimi M, Inze D, Depicker A (2002) GATEWAY vectors for agrobacterium-mediated plant transformation. Trends Plant Sci 7:193–195
Ivanov S, Harrison MJ (2014) A set of fluorescent protein-based markers expressed from constitutive and arbuscular mycorrhiza-inducible promoters to label organelles, membranes and cytoskeletal elements in Medicago truncatula. Plant J 80:1151–1163
Acknowledgments
We thank J. Terpolilli, Murdoch University, Australia for providing M. ciceri CC1192; Senthil-Kumar Muthappa, NIPGR, New Delhi for providing chickpea seeds (BGD 112, PUSA 372, GNG 1958, GNG 1488, GNG 1581, RSG 931); Michael Udvardi, Nobel Research Institute, Oklahoma, USA for A. rhizogenes ARqua1 strain; Maria J. Harrison, Boyce Thompson Institute for Plant Research Ithaca, USA for providing pAtUb driven subcellular marker construct; M. DasGupta, Department of Biochemistry, University of Calcutta, for providing pCAMBIA2301 vector; NIPGR for their confocal facilities; and CIF-NIPGR; NIPGR-DELCON for their support. This work is supported by core research grant from National Institute of Plant Genome Research, Ramalingwaswami Re-entry grant, DBT (BT/RLF/Re-entry/41/2013).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Mandal, D., Srivastava, D., Sinharoy, S. (2020). Optimization of Hairy Root Transformation for the Functional Genomics in Chickpea: A Platform for Nodule Developmental Studies. In: Jain, M., Garg, R. (eds) Legume Genomics. Methods in Molecular Biology, vol 2107. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0235-5_18
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0235-5_18
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0234-8
Online ISBN: 978-1-0716-0235-5
eBook Packages: Springer Protocols