Abstract
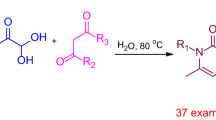
Pyridine N-oxides are a class of mild Lewis bases that can activate certain kinds of Lewis acidic parts of molecules and hence increase reactivity of its nucleophilic part towards various reactions with electrophiles. This review aims to summarize the applications of various non-chiral and chiral pyridine N-oxides in catalysis of a variety of reactions. In addition, the applications of these reactions in syntheses of natural and bioactive compounds are mentioned as well.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
Trichlorosilyl enolates of aldehydes and ketones as the reagents of the first choice were found not reactive enough for addition to ketones.
References
Dalko PI, Moisa L (2001) Angew Chem Int Ed Engl 40:3726
Jha SC, Joshi NN (2002) ARKIVOC:167
Chelucci V, Thummel RP (2002) Chem Rev 102:3129
Malkov AV, Kočovský P (2003) Curr Org Chem 7:1737
Denmark SE, Fu J (2003) Chem Rev 103:2763
Dalko PI, Moisa L (2004) Angew Chem Int Ed Engl 43:5138
Kočovský P, Malkov AV (2004) Russ Chem Bull Int Ed 53:1806
Chelucci V, Murineddu G, Pinna GA (2004) Tetrahedron Asymmetry 15:1373
Rendler S, Oesterreich M (2005) Synlett:1727
Orito Y, Nakajima M (2006) Synlett:1391
Malkov AV, Kočovský P (2007) Eur J Org Chem:29
Liu X, Lin L, Feng X (2013) Acc Chem Res 44:574
Sutar RL, Joshi NN (2013) Tetrahedron Asymmetry 24:1345
Antkowiac WZ, Gessner WP (1979) Tetrahedron Lett 20:1931
Herrmann A, Hedman H, Rosén J, Jansson D, Haraldsson B, Hellenäs K-E (2012) J Nat Prod 75:1690
O’Donnell G, Poeschl R, Zimhony O, Gunaratnam M, Moreira JBO, Neidle S, Evangenopoulos D, Bhakta S, Malkinson JP, Boshoff HI, Lenaerts A, Gibbonds S (2009) J Nat Prod 72:360
Palani A, Shapiro S, Josien H, Bara T, Clader JW, Greenlee WJ, Cox K, Strizki JM, Baroudy BM (2002) J Med Chem 45:3143
Nantermet PG, Burgez CS, Robinson K, Pellicore JM, Newton CL, Deng JZ, Selnick HG, Lewis SD, Lucas BJ, Krueger JA, Miller-Stein C, White RB, Wong B, McMasters DR, Wallace AA, Lynch Jr JJ, Yan Y, Chen Z, Kuo L, Gardell SJ, Shafer JA, Vacca JP, Lyle TA (2005) Bioorg Med Chem Lett 15:2771
Mfuh AM, Larionov OV (2015) Curr Med Chem 22:2819
Balzarini J, Stevens M, De Clercq E, Schols D, Pannecouque C (2005) J Antimicrob Chemother 55:135
Brycki B, Szafran M (1984) J Chem Soc Perkin 2:223
Chmurzyński L (1992) J Solution Chem 21:171
Chmurzyński L, Liwo A, Barcyński P (1996) Anal Chim Acta 335:147
Chmurzyński L, Wawryniak G, Warnke Z (1997) J Heterocycl Chem 34:215
Ducháčková L, Kadlčíková A, Kotora M, Roithová J (2010) J Am Chem Soc 132:12660
Abraham MH, Honcharova L, Rocco SA, Acree Jr WE, De Fina KM (2011) New J Chem 35:930
Bergbreiter DE, Ortiz-Acosta D (2008) Tetrahedron Lett 49:5608
Hagiwara H, Inoguchi H, Fukushima M, Hoshi T, Suzuki T (2005) Synlett:2388
Denmark SE, Fan Y (2005) J Org Chem 70:9667
Murray JI, Woscholski R, Spivey AC (2014) Chem Commun 50:13608
Yoshida K, Takao K (2014) Tetrahedron Lett 55:6861
Yoshida K, Fujino Y, Itatsu Y, Inoue H, Kanoko Y, Takao K (2016) Tetrahedron Lett 57:627
Ando M, Emoto S (1975) Bull Soc Chim Jpn 48:1655
Ryzhkov AV, Rodina LL (2006) Russ J Gen Chem 76:126
Belousova IA, Simanenko YS, Savelova VA (2001) Russ J Org Chem 37:969
Belousova IA, Savelova VA, Simanenko YS, Panchenko BV (2002) Russ J Org Chem 38:111
Shiina I, Ushiyama H, Yamada Y, Kawakita Y, Nakata K (2008) Chem Asian J 3:454
Fujii M, Honda A (1992) J Heterocycl Chem 29:931
Fujii M, Honda A (1992) Chem Express 7:329
Nakajima M, Saito M, Shiro M, Hashimoto S (1998) J Am Chem Soc 120:6419
Shimada T, Kina A, Ikeda S, Hayashi T (2002) Org Lett 4:2799
Kina A, Shimada T, Hayashi T (2004) Adv Synth Catal 346:1169
Kwak J, Ohk J, Jung Y, Chang S (2012) J Am Chem Soc 134:17778
Jiao Z, Feng X, Liu B, Chen F, Zhang G, Jiang Y (2003) Eur J Org Chem:3818
Malkov AV, Dufková L, Farrugia L, Kočovský P (2003) Angew Chem Int Ed Engl 42:3674
Hrdina R, Stará GI, Dufková L, Mitchell S, Císařová I, Kotora M (2006) Tetrahedron 62:96
Hrdina R, Kadlčíková A, Valterová I, Hodačová J, Kotora M (2006) Tetrahedron Asymmetry 17:3185
Hrdina R, Valterová I, Hodačová J, Císařová I, Kotora M (2007) Adv Synth Catal 349:822
Hrdina R, Dračínský M, Valterová I, Hodačová J, Císařová I, Kotora M (2008) Adv Synth Catal 350:1449
Kadlčíková A, Hrdina R, Valterová I, Kotora M (2009) Adv Synth Catal 351:1279
Bai B, Shen L, Ren J, Zhu J-H (2012) Adv Synth Catal 354:354
Deng Y, Pan W, Pei N-Y, Li L-J, Bai B, Zhu J-H (2013) Tetrahedron 69:10431
Pignataro L, Benaglia M, Cinquini M, Cozzi F, Celentano G (2005) Chirality 17:396
Nakajima M, Yokota T, Saito M, Hashimoto S (2004) Tetrahedron Lett 45:61
Gao D-W, Gu Q, You S-L (2016) ACS Catal 4:2741
Malkov AV, Orsini M, Pernazza D, Muir WK, Langer V, Meghani P, Kočovský P (2002) Org Lett 4:1047
Malkov AV, Bell M, Orsini M, Pernazza D, Massa A, Herrmann P, Meghani P, Kočovský P (2003) J Org Chem 68:9659
Malkov AV, Bell M, Vassieu M, Bugatti V, Kočovský P (2003) J Mol Catal A Chem 196:179
Malkov AV, Bell M, Casteluzzo F, Kočovský P (2005) Org Lett 7:3219
Chelucci G, Belmonte N, Benaglia M, Pignataro L (2007) Tetrahedron Lett 48:4037
Denmark SE, Fan Y, Eastgate MD (2005) J Org Chem 70:5235
Diana MB, Marchetti M, Melloni G (1995) Tetrahedron Asymmetry 6:1175
Wong W-L, Lee C-S, Leung H-K, Kwong H-L (2004) Org Biomol Chem 2:1967
Pignataro L, Benaglia M, Annunziata R, Cinquini M, Cozzi F (2006) J Org Chem 71:1458
Derdau V, Laschat S, Hupe E, König WA, Dix I, Jones PG (1999) Eur J Inorg Chem:1001
Denmark SE, Fan Y (2002) J Am Chem Soc 124:4233
Bolm C, Ewald M, Zehnder M, Neuburger MA (1992) Chem Ber 125:453
Bolm C, Ewald M, Felder M, Schlingloff G (1992) Chem Ber 125:1169
Boyd RD, Sharma DN, Sbircea L, Murphy D, Malone FJ, James LS, Allen CRC, Hamilton TGJ (2010) Org Biomol Chem 8:1081
Gnanamani E, Someshwar N, Ramanathan RC (2012) Adv Synth Catal 354:2101
Gnanamani E, Someshwar N, Sanjeevi J, Ramanathan RC (2014) Adv Synth Catal 356:2219
Neniškis A, Stončius S (2015) Eur J Org Chem:6359
Gaecía-Flores F, Flores-Michel LS, Juaristi E (2006) Tetrahedron Lett 47:8235
Ikai T, Moro M, Maeda K, Kanoh S (2001) React Funct Polym 71:1055
Tao B, Lo MM-C, Fu GC (2001) J Am Chem Soc 123:353
Chai Q, Song C, Sun Z, Ma Y, Ma C, Dai Y, Andrus BM (2006) Tetrahedron Lett 47:8611
Fulton JR, Glover JE, Kamara L, Rowlands GJ (2011) Chem Commun 47:433
Takenaka N, Sarangthem RS, Captain B (2008) Angew Chem Int Ed Engl 47:9708
Chen J, Takenaka N (2009) Chem A Eur J 15:7268
Chen J, Captain B, Takenaka N (2011) Org Lett 13:2011
O’Hora SP, Incerti-Pradillos CA, Kabeshov AM, Shipilovskikh AS, Rubtsov EA, Elsegood RJM, Malkov VA (2015) Chem A Eur J 21:4551
Denmark SE, Fan Y (2006) Tetrahedron Asymmetry 17:687
Malkov AV, Ramírez-López P, Biedermannová L, Rulíšek L, Dufková L, Kotora M, Zhu F, Kočovský P (2008) J Am Chem Soc 130:5341
Cadart T, Koukal P, Kotora M (2014) Eur J Org Chem:7556
Hessler F, Betík R, Kadlčíková A, Belle R, Kotora M (2014) Eur J Org Chem:7245
Shimada T, Kina A, Hayashi T (2003) J Org Chem 68:6329
Malkov VA, Barlóg M, Miller-Potucká L, Kabeshov MA, Farrugia LJ, Kočovský P (2012) Chem A Eur J 18:6873
Malkov AV, Ramírez-Lopéz P, Biedermannová L, Rulíšek L, Dufková L, Kotora M, Zhu F, Kočovský P (2008) J Am Chem Soc 130:5431
Bai B, Zhu H-J, Pan W (2012) Tetrahedron 68:6829
Malkov AV, Kabeshov AM, Barlog M, Kočovský P (2009) Chem A Eur J 15:1570
Malkov AV, Stončius S, Bell M, Castelluzo F, Ramírez-Lopéz P, Biedermannová L, Langer V, Rulíšek L, Kočovský P (2013) Chem A Eur J 19:9167
Chelucci G, Baldino S, Pinna GA, Benaglia M, Buffa L, Guizzetti S (2008) Tetrahedron 64:7574
Incerti-Pradillos CA, Kabeshov AM, O’Hora SP, Shipilovskikh AS, Rubtsov EA, Drobkova VA, Baladina SY, Malkov AV (2016) Chem A Eur J 21:14390
Kadlčíková A, Vlašaná K, Ducháčková L, Roithová J, Kotora M (2010) Chem A Eur J 16:9442
Vlašaná K, Hrdina R, Valterová I, Kotora M (2010) Eur J Org Chem:7040
Malkov AV, Barzog M, Jewkes Y, Mikušek J, Kočovský P (2011) J Org Chem 76:4800
Koukal P, Ulč J, Nečas D, Kotora M (2016) Eur J Org Chem:2110
Matoušová E, Koukal P, Formánek B, Kotora M (2016) Org Lett 18:5656
Koukal P, Kotora M (2015) Chem A Eur J 21:7408
Vlašaná K, Betík R, Valterová I, Nečas D, Kotora M (2016) Curr Organocatal 3:301
Hrdina R, Boyd T, Valterová I, Hodačová J, Kotora M (2008) Synlett:3141
Kolská K, Ghavre M, Pour M, Hybelbauerová S, Kotora M (2016) Asian J Org Chem 5:646
Nakajima M, Saito M, Hashimoto S (2002) Tetrahedron Asymmetry 13:2449
Peng Y, Takenaka N (2012) Chem Rec 13:28
Lu T, Zhu R, An Y, Wheeler SE (2012) J Am Chem Soc 134:3095
Sugiura M, Sato N, Sonoda Y, Kotani S, Nakajima M (2010) Chem Asian J 5:478
Kadlčíková A, Vlašaná K, Kotora M (2011) Collect Czech Chem Commun 76:415
Chen F-X, Qin B, Feng X, Zhang G, Jiang Y (2004) Tetrahedron 60:10449
Jiao Z, Feng X, Liu B, Chen F, Zhang G, Jiang Y (2003) Eur J Org Chem:3818
Su Z, Hu C, Feng X (2006) Tetrahedron 62:3818
Sugiura M, Kumahara M, Nakajima M (2009) Chem Commun:3585
Diana MB, Marchetti M, Melloni G (1995) Tetrahedron Asymmetry 6:1175
Matoušová E, Koukal P, Formánek B, Kotora M (2016) Org Lett 18:5656
Hessler F, Korotvička A, Nečas D, Valterová I, Kotora M (2014) Eur J Org Chem:2543
Motloch P, Valterová I, Kotora M (2014) Adv Synth Catal 356:199
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Koukal, P., Ulč, J., Nečas, D., Kotora, M. (2017). Pyridine N-Oxides and Derivatives Thereof in Organocatalysis. In: Larionov, O. (eds) Heterocyclic N-Oxides. Topics in Heterocyclic Chemistry, vol 53. Springer, Cham. https://doi.org/10.1007/7081_2017_3
Download citation
DOI: https://doi.org/10.1007/7081_2017_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-60686-6
Online ISBN: 978-3-319-60687-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)