Abstract
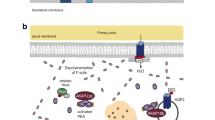
Pharmacological chaperones recently opened new possibilities in G protein-coupled receptor drug discovery. Even more interestingly, some unique ligands combine pharmacological chaperoning and biased agonism properties, boosting their therapeutic interest in many human diseases resulting from G protein-coupled receptor mutation and misfolding. These compounds displaying dual characteristics would constitute a perfect treatment for congenital Nephrogenic Diabetes Insipidus, a typical conformational disease. This X-linked genetic pathology is mostly associated with inactivating mutations of the renal arginine-vasopressin V2 receptor leading to misfolding and intracellular retention of the receptor, causing the inability of patients to concentrate their urine in response to the antidiuretic hormone. Cell-permeable pharmacological chaperones have been successfully challenged to restore plasma membrane localization of many V2 receptor mutants. In addition, different classes of specific ligands such as antagonists, agonists as well as biased agonists of the V2 receptor have proven their usefulness in rescuing mutant receptor function. This is particularly relevant for small-molecule biased agonists which only trigger Gs protein activation and cyclic adenosine monophosphate production, the V2-induced signaling pathway responsible for water reabsorption. In parallel, high-throughput screening assays based on receptor trafficking rescue approaches have been developed to discover novel V2 pharmacological chaperone molecules from different chemical libraries. These new hit compounds, which still need to be pharmacologically characterized and functionally tested in vivo, represent promising candidates for the treatment of congenital Nephrogenic Diabetes Insipidus.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- AQP2:
-
Aquaporin-2
- AVP:
-
Arginine-vasopressin
- cAMP:
-
Cyclic adenosine monophosphate
- cNDI:
-
Congenital nephrogenic diabetes insipidus
- ER:
-
Endoplasmic reticulum
- FDA:
-
US food and drug administration
- GnRHR:
-
Gonadotropin-releasing hormone receptor
- GPCR:
-
G protein-coupled receptor
- Gs:
-
G protein subunit αs
- LSD:
-
Lysosomal storage disorder
- NMR:
-
Nuclear magnetic resonance
- OT:
-
Oxytocin
- PC:
-
Pharmacological chaperone, pharmacochaperone, pharmacoperone
- PCT:
-
Pharmacological chaperone therapy
- 3D:
-
Three-dimensional
- TM:
-
Transmembrane
- V2R:
-
Vasopressin type 2 receptor
References
Ala Y, Morin D, Mouillac B et al (1998) Functional studies of twelve mutant V2 vasopressin receptors related to nephrogenic diabetes insipidus: molecular basis of a mild clinical phenotype. J Am Soc Nephrol 9(10):1861–1872
Aronson D, Verbalis JG, Mueller M et al (2011) Short- and long-term treatment of dilutional hyponatraemia with satavaptan, a selective arginine-vasopressin V2 receptor antagonist: the DILIPO study. Eur J Heart Fail 13(3):327–336
Auzan RJ, Ventura MA, Clauser E (2005) Mechanisms of cell-surface rerouting of an endoplasmic reticulum-retained mutant of the vasopressin V1b/V3 receptor by a pharmacological chaperone. J Biol Chem 280(51):42198–42206
Bernier V, Bichet DG, Bouvier M (2004a) Pharmacological chaperone action on G protein-coupled receptors. Curr Opin Pharmacol 4(5):528–533
Bernier V, Lagacé M, Lonergan M et al (2004b) Functional rescue of the constitutively internalized V2 vasopressin receptor mutant R137H by the pharmacological chaperone action of SR49059. Mol Endocrinol 18(8):2074–2084
Bernier V, Morello JP, Zarruk A et al (2006) Pharmacologic chaperones as a potential treatment for X-linked nephrogenic diabetes insipidus. J Am Soc Nephrol 17(1):233–243
Bichet DG, Birnbaumer M, Lonergan M et al (1994) Nature and recurrence of AVPR2 mutations in X-linked nephrogenic diabetes insipidus. Am J Hum Genet 55(2):278–286
Birnbaumer M, Seibold A, Gilbert S et al (1992) Molecular cloning of the receptor for human antidiuretic hormone. Nature 357(6376):333–335
Bockenhauer D, Carpentier E, Rochdi D et al (2010) Vasopressin type 2 receptor V88M mutation: molecular basis of partial and complete nephrogenic diabetes insipidus. Nephron Physiol 114(1):1–10
Bockenhauer D, Bichet DG (2014) Urinary concentration: different ways to open and close the tap. Pediatr Nephrol 29(8):1297–1303
Brady RO (2006) Enzyme replacement for lysosomal diseases. Annu Rev Med 57:283–296
Carter AA, Hill SJ (2005) Characterization of isoprenaline- and salmeterol-stimulated interactions between beta2-adrenoceptors and beta-arrestin 2 using beta-galactosidase complementation in C2C12 cells. J Pharmacol Exp Ther 315(2):839–848
Chaudhuri TK, Paul S (2006) Protein-misfolding diseases and chaperone-based therapeutic approaches. FEBS J 273(7):1331–1349
Chen X, Sassano MF, Zheng L et al (2012) Structure-functional selectivity relationship studies of β-arrestin-biased dopamine D2 receptor agonists. J Med Chem 55(16):7141–7153
Cohen FE, Kelly LW (2003) Therapeutic approaches to protein-misfolding diseases. Nature 426(6968):905–909
Conn PM, Ulloa-Aguire A, Ito J et al (2007) G protein-coupled receptor trafficking in health and disease: lessons learned to prepare for therapeutic mutant rescue in vivo. Pharmacol Rev 59(3):225–250
Conn PM, Ulloa-Aguirre A (2010) Trafficking of G protein-coupled receptors to the plasma membrane: insights from pharmacoperone drugs. Trends Endocrinol Metab 21(3):190–197
Conn PM, Ulloa-Aguirre A (2011) Pharmacological chaperones for misfolded gonadotropin-releasing hormone receptors. Adv Pharmacol 62:109–141
Conn PM, Smith E, Hodder PS et al (2013) High throughput screen for pharmacoperones of the vasopressin type 2 receptor. J Biomol Screen 18(8):930–937
Conn PM, Smithson DC, Hodder PS et al (2014) Transitioning pharmacoperones to therapeutic use: in vivo proof-of-principle and design of high throughput screens. Pharmacol Res 83:38–51
Conn PM, Spicer TP, Scampavia L et al (2015) Assays strategies for identification of therapeutic leads that target protein trafficking. Trends Pharmacol Sci 36(8):498–505
Feinstein TN, Yui N, Webber MJ et al (2013) Noncanonical control of vasopressin receptor type 2 signaling by retromer and arrestin. J Biol Chem 288(39):27849–27860
Ferguson SS (2001) Evolving concepts in G protein-coupled receptor endocytosis: the role in receptor desensitization and signaling. Pharmacol Rev 53(1):1–24
Galandrin S, Oligny-Longpré G, Bouvier M (2007) The evasine nature of drug efficacy: implications for drug discovery. Trends Pharmacol Sci 28(8):423–430
Germain DP, Giugliani R, Hughes DA et al (2012) Safety and pharmacodynamic effects of a pharmacological chaperone on α-galactosidase A activity and globotriaosylceramide clearance in Fabry disease: report from two phase 2 clinical studies. Orphanet J Rare Dis 7:91
Germain DP, Hughes DA, Nicholls K et al (2016) Treatment of Fabry’s disease with the pharmacologic chaperone migalastat. N Engl J Med 375(6):545–555
Groer CE, Tidgewell K, Moyer RA et al (2007) An opioid agonist that does not induce mu-opioid receptor-arrestin interactions or receptor internalization. Mol Pharmacol 71(2):549–557
Hawtin SR (2006) Pharmacological chaperone activity of SR49059 to functionally recover misfolded mutations of the vasopressin V1a receptor. J Biol Chem 281(21):14604–14614
Holloway AC, Qian H, Pipolo L et al (2002) Side-chain substitutions within angiotensin II reveal different requirements for signaling, internalization, and phosphorylation of type 1A angiotensin receptors. Mol Pharmacol 61(4):768–777
Janovick JA, Maya-Nunez G, Conn PM (2002) Rescue of hypogonadotropic hypogonadism-causing and manufactured GnRH receptor mutants by a specific protein-folding template: misrouted proteins as a novel disease etiology and therapeutic target. J Clin Endocrinol Metab 87(7):3255–3262
Janovick JA, Maya-Nunez G, Ulloa-Aguire A et al (2009) Increased plasma membrane expression of human follicle-stimulating hormone receptor by a small molecule thienopyr(im)idine. Mol Cell Endocrinol 298(1–2):84–88
Janovick JA, Park BS, Conn PM (2011) Therapeutic rescue of misfolded mutants: validation of primary high throughput screens for identification of pharmacoperone drugs. PLoS One 6(7):e22784
Janovick JA, Stewart MD, Jacob D et al (2013) Restoration of testis function in hypogonadotropic hypogonadal mice harboring a misfolded GnRHR mutant by pharmacoperone drug therapy. Proc Natl Acad Sci U S A 110(52):21030–21035
Janovick JA, Spicer TP, Smith E et al (2016) Receptor antagonism/agonism can be uncoupled from pharmacoperone activity. Mol Cell Endocrinol 434:176–185
Janovick JA, Spicer TP, Bannister TD et al (2017) Pharmacoperone rescue of vasopressin 2 receptor mutants reveals unexpected constitutive activity and coupling bias. PLoS One 12(8):e0181830
Jean-Alphonse F, Perkovska S, Frantz MC et al (2009) Biased agonist pharmacochaperones of the AVP V2 receptor may treat congenital nephrogenic diabetes insipidus. J Am Soc Nephrol 20(10):2190–2203
Karageorgos LE, Isaac EL, Brooks DA et al (1997) Lysosomal biogenesis in lysosomal storage disorders. Exp Cell Res 234(1):85–97
Laugwitz KL, Allgeier A, Offermanns S et al (1996) The human thyrotropin receptor: a heptahelical receptor capable of stimulating members of all four G protein families. Proc Natl Acad Sci U S A 93(1):116–120
Leidenheimer NJ, Ryder KG (2014) Pharmacological chaperoning: a primer on mechanism and pharmacology. Pharmacol Res 83:10–19
Lolait SJ, Carroll AM, McBride OW et al (1992) Cloning and characterization of a vasopressin V2 receptor and possible link to nephrogenic diabetes insipidus. Nature 357(6376):526–529
Loo TW, Clarke DM (1997) Correction of defective protein kinesis of human P-glycoprotein mutants by substrates and modulators. J Biol Chem 272(2):709–712
Los EL, Deen PMT, Robben JH (2010) Potential of nonpeptide (ant)agonists to rescue vasopressin V2 receptor mutants for the treatment of X-linked nephrogenic diabetes insipidus. J Neuroendocrinol 22(5):393–399
Luttrell LM, Gesty-Palmer D (2010) Beyond desensitization: physiological relevance of arrestin-dependent signaling. Pharmacol Rev 62(2):305–330
Lutrell LM (2014) More than just a hammer: ligand “bias” and pharmaceutical discovery. Mol Endocrinol 28(3):281–294
Luttrell LM, Maudsley S, Bohn LM (2015) Fulfilling the promise of “biased” G protein-coupled receptor agonism. Mol Pharmacol 88(3):579–588
Mendre C, Mouillac B (2010) Pharmacological chaperones: a potential therapeutic treatment for conformational diseases. Med Sci (Paris) 26(6–7):627–635
Moeller HB, Rittig S, Fenton RA (2013) Nephrogenic diabetes insipidus: essential insights into the molecular background and potential therapies for treatment. Endocr Rev 34(2):278–301
Morello JP, Salahpour A, Laperrière A et al (2000a) Pharmacological chaperones rescue cell-surface expression and function of misfolded V2 vasopressin receptor mutants. J Clin Invest 105(7):887–895
Morello JP, Petäjä-Repo UE, Bichet DG et al (2000b) Pharmacological chaperones: a new twist on receptor folding. Trends Pharmacol Sci 21(12):466–469
Morello JP, Bichet DG (2001) Nephrogenic diabetes insipidus. Annu Rev Physiol 63:607–630
Mouillac B, Mendre C (2014) Vasopressin receptors and pharmacological chaperones: from functional rescue to promising therapeutic strategies. Pharmacol Res 83:74–78
Newton CL, Whay AM, McArdle CA et al (2011) Rescue of expression and signaling of human luteinizing hormone G protein-coupled receptor mutants with an allosterically binding small-molecule agonist. Proc Natl Acad Sci U S A 108(17):7172–7176
Offermanns S, Wieland T, Homann D et al (1994) Transfected muscarinic acetylcholine receptors selectively couple to Gi-type G proteins and Gq/11. Mol Pharmacol 45(5):890–898
Parenti G, Andria G, Valenzano KJ (2015) Pharmacological chaperone therapy: preclinical development, clinical translation, and prospects for the treatment of lysosomal storage disorders. Mol Ther 23(7):1138–1148
Parkinson-Lawrence EJ, Shandala T, Prodoehl M et al (2010) Lysosomal storage disease: revealing lysosomal function and physiology. Physiology (Bethesda) 25(2):102–115
Platt FM, Jeyakumar M (2008) Substrate reduction therapy. Acta Paediatr 97(457):88–93
Rahmeh R, Damian M, Cottet M et al (2012) Structural insights into biased G protein-coupled receptor signaling revealed by fluorescence spectroscopy. Proc Natl Acad Sci U S A 109(17):6733–6738
Rajagopal S, Rajagopal K, Lefkowitz RJ (2010) Teaching old receptors new tricks: biasing seven-transmembrane receptors. Nat Rev Drug Discov 9(5):373–386
Reiter E, Ahn S, Shukla AK et al (2012) Molecular mechanism of β-arrestin-biased agonism at seven-transmembrane receptors. Annu Rev Pharmacol Toxicol 52:179–197
Robben JH, Sze M, Knoers NV et al (2006) Rescue of vasopressin V2 receptor mutants by chemical chaperones: specificity and mechanism. Mol Biol Cell 17(1):379–386
Robben JH, Sze M, Knoers NV et al (2007) Functional rescue of vasopressin V2 receptor mutants in MDCK cells by pharmacochaperones: relevance to therapy of nephrogenic diabetes insipidus. Am J Physiol Renal Physiol 292(1):F253–F260
Robben JH, Kortenoeven MLA, Sze M et al (2009) Intracellular activation of vasopressin V2 receptor mutants in nephrogenic diabetes insipidus by nonpeptide agonists. Proc Natl Acad Sci U S A 106(29):12195–12200
Rosenthal W, Seibold A, Antaramian A et al (1992) Molecular identification of the gene responsible for congenital nephrogenic diabetes insipidus. Nature 359(6392):233–235
Sagan S, Chassaing G, Pradier L et al (1996) Tachykinin peptides affect differently the second messenger pathways after binding to CHO-expressed human NK-1 receptors. J Pharmacol Exp Ther 276(3):1039–1048
Sato S, Ward CL, Krouse ME et al (1996) Glycerol reverses the misfolding phenotype of the most common cystic fibrosis mutation. J Biol Chem 271(2):635–638
Schrier RW, Gross P, Gheorghiade M (2006) Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. New Engl J Med 355(20):2099–2112
Semple G, Skinner PJ, Gharbaoui T et al (2008) 3-(1H-tetrazol-5-yl)-1,4,5,6-tetrahydro-cyclopentapyrazole (MK-0354): a partial agonist of the nicotinic acid receptor, G-protein coupled receptor 109a, with antilipolytic but no vasodilatory activity in mice. J Med Chem 51(16):5101–5108
Serradeil-Le Gal C, Lacour C, Valette G et al (1996) Characterization of SR 121463A, a highly potent and selective, orally active vasopressin V2 receptor antagonist. J Clin Invest 98(12):2729–2738
Shenoy S, Lefkowitz RJ (2011) β-Arrestin-mediated receptor trafficking and signal transduction. Trends Pharmacol Sci 32(9): 521-533
Smith E, Janovick JA, Bannister TD et al (2016) Identification of potential pharmacoperones capable of rescuing the functionality of misfolded vasopressin 2 receptor involved in nephrogenic diabetes insipidus. J Biomol Screen 21(8):824–831
Smithson DC, Janovick JA, Conn PM (2013) Therapeutic rescue of misfolded/mistrafficked mutants: automation-friendly high-throughput assays for identification of pharmacoperone drugs of GPCRs. Methods Enzymol 521:3–16
Takasu H, Gardella TJ, Luck MD et al (1999) Amino-terminal modifications of human parathyroid hormone (PTH) selectively alter phospholipase C signaling via the type 1 PTH receptor: implications for design of signal-specific PTH ligands. Biochemistry 38(41):13453–13460
Tamarappoo BK, Verkman AS (1998) Defective aquaporin-2 trafficking in nephrogenic diabetes insipidus and correction by chemical chaperones. J Clin Invest 101(10):2257–2267
Tan CM, Nickols HH, Limbird LE (2003) Appropriate polarization following pharmacological rescue of V2 vasopressin receptors encoded by X-linked nephrogenic diabetes insipidus alleles involves a conformation of the receptor that also attains mature glycosylation. J Biol Chem 278(37):35678–35686
Thurmond RL, Desai PJ, Dunford PJ et al (2004) A potent and selective histamine H4 receptor antagonist with anti-inflammatory properties. J Pharmacol Exp Ther 309(1):404–413
Treschan TA, Peters J (2006) The vasopressin system. Anesthesiology 105(3):599–612
Tsukagushi H, Matsubara H, Taketani S et al (1995) Binding, intracellular transport and biosynthesis-defective mutants of vasopressin type 2 receptor in patients with X-linked nephrogenic diabetes insipidus. J Clin Invest 96(4):2043–2050
Violin JD, DeWire SM, Yamashita D et al (2010) Selectively engaging β-arrestins at the angiotensin II type 1 receptor reduces blood pressure and increases cardiac performance. J Pharmacol Exp Ther 335(3):572–579
Wesche D, Deen PMT, Knoers NV (2012) Congenital nephrogenic diabetes insipidus: the current state of affairs. Pediatr Nephrol 27(12):2183–2204
White E, McKenna J, Cavanaugh A et al (2009) Pharmacochaperone-mediated rescue of calcium-sensing receptor loss-of-function mutants. Mol Endocrinol 23(7):1115–1123
Wisler JW, DeWire SM, Whalen EJ et al (2007) A unique mechanism of beta-blocker action: carvedilol stimulates beta-arrestin signaling. Proc Natl Acad Sci U S A 104(42):16657–16662
Wüller S, Wiesner B, Loffler A et al (2004) Pharmacochaperones post-translationally enhance cell surface expression by increasing conformational stability of wild-type and mutant vasopressin V2 receptors. J Biol Chem 279(45):47254–47263
Zimran A, Altarescu G, Elstein D (2013) Pilot study using ambroxol as a pharmacological chaperone in type 1 Gaucher disease. Blood Cells Mol Dis 50(2):134–137
Acknowledgments
We would like to dedicate this chapter in memory of P. Michael Conn.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Mouillac, B., Mendre, C. (2017). Pharmacological Chaperones as Potential Therapeutic Strategies for Misfolded Mutant Vasopressin Receptors. In: Ulloa-Aguirre, A., Tao, YX. (eds) Targeting Trafficking in Drug Development. Handbook of Experimental Pharmacology, vol 245. Springer, Cham. https://doi.org/10.1007/164_2017_50
Download citation
DOI: https://doi.org/10.1007/164_2017_50
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-74163-5
Online ISBN: 978-3-319-74164-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)