Abstract
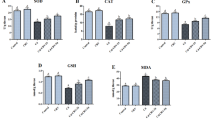
Although copper is an essential micronutrient involved in a variety of biological processes indispensable for sustaining life, it can be toxic when administered in excess. Licorice (Glycyrrhizaglabra) has been used in Chinese folk medicine for the treatment of various disorders. Licorice has the biological capabilities of detoxication, antioxidation, and antiinfection. Here, we test the hypothesis that licorice could ameliorate copper-induced neurotoxic and genotoxic effects in adult male albino rats. For this purpose, 48 adult male albino rats were randomized into five groups: group I (8 rats), untreated control; group II (16 rats), subdivided into; vehicle control IIa (8 rats) which received 1 mL saline twice weekly intraperitoneally for 8 weeks and vehicle control IIb (8 rats) received 0.5 mL distilled water/day orally gavaged for 8 weeks; group III (8 rats), treated with licorice dissolved in 0.5 mL of distilled water, 50 mg/kg b.w./day orally gavaged for 8 weeks; group IV (8 rats), copper chloride (CuCl2) dissolved in 0.5 mL saline, 7 mg/kg b.w. twice weekly intraperitoneal for 8 weeks; and group V (8 rats), CuCl2 + licorice (the same previously mentioned doses) licorice extract were orally given for 10 days before treatment was initiated then followed by CuCl2 intraperitoneally for 8 weeks. We found that CuCl2 exposure significantly increased brain oxidative stress as manifested by elevated malondialdehyde levels, decreased reduced glutathione content, and depressed antioxidant enzyme activities in brain tissues when compared with control groups. This was accompanied by histopathological changes in the form of increased cellularity and swelling of astrocytes that showed dense eosinophilic cytoplasm, pyknotic nuclei, and multiple apoptotic bodies that associated with degenerated neurons with deep eosinophilic cytoplasm. Also, strong Bax immunoreactions in the brain were detected. Furthermore, comet assay results confirmed CuCl2-related oxidative DNA damage. Notably, all these changes were partially ameliorated in rats treated concomitantly with licorice and CuCl2. Our results showed that licorice exerts protective effects against CuCl2-induced neuro- and genotoxicities. These effects may be attributed to the antioxidative property of licorice.



Similar content being viewed by others
References
Al-Qarawi AA, Abdel-Rahman HA, Ali BH, El Mougy SA (2002) Liquorice (Glycyrrhiza glabra) and the adrenal-kidney-pituitary axis in rats. Food Chem Toxicol 40(10):1525–1527
Arnal N, de Alaniz M, Marra C (2010) Alterations in copper homeostasis and oxidative stress biomarkers in women using the intrauterine device TCu380A. Toxicol Lett 192:373–378
Arnal N, de Alaniz M, Marra C (2011) Carnosine and neocuproine as neutralizing agents for copper overload induced damages in cultured human cells. Chem Biol Interact 192:257–263
Arnal N, de Alaniz M, Marra C (2012) Cytotoxic effects of copper overload on human-derived lung and liver cells in culture. Biochim Biophys Acta 1820:931–939
Arnal N, Dominic L, de Tacconi M (2014) Copper-induced alterations in rat brain depends on route of overload and basal copper levels. Nutrition 30(1):96–106
Asl MN, Hosseinzadeh H (2008) Review of pharmacological effects of Glycyrrhiza sp. and its bioactive compounds. Phytother Res 22(6):709–724
Bancroft JD, Gamble M (2007) Connective tissue stains. In: Bancroft JD, Gamble M (eds) Theory and practice of histological techniques, 6th edn. Churchill Livingstone, London New York Philadelphia, p 150
Chen ZA, Wang JL, Liu RT, Ren JP, Wen LQ, Chen XJ, Bian GX (2009) Liquiritin potentiate neurite outgrowth induced by nerve growth factor in PC12 cells. Cytotechnology 60(1–3):125–132
Choi B, Zheng W (2009) Copper transport to the brain by the blood–brain barrier and blood–CSF barrier. Brain Res 1248:14–21
Ellman G, Lysko H (1967) Disulfide and sulfhydryl compounds in TCA extracts of human blood and plasma. J Lab Clin Med 70:518
Er E, Oliver L, Cartron PF, Juin P, Manon S, Vallette FM (2006) Mitochondria as the target of the pro-apoptotic protein Bax. Biochim Biophys Acta (BBA)-Bioenergetics 1757(9):1301–1311
Fraga C (2005) Relevance, essentiality and toxicity of trace elements in human health. Mol Asp Med 26:235–244
Frem J, Sarson Y, Sternberg T, Cole CR (2010) Copper supplementation in parenteral nutrition of cholestatic infants. J Pediatr Gastroenterol Nutr 50(6):650
Gouider-Khouja N (2009) Wilson’s disease. Parkinsonism Relat Disord 15(Suppl 3):S126–S129
Guecheva T, Henriques J, Erdtmann B (2001) Genotoxic effects of copper sulphate in freshwater planarian in vivo, studied with the single-cell gel test (comet assay). Mutat Res 497(1–2):19–27
Gutteridge JM, Halliwell B (2000) Free radicals and antioxidants in the year 2000. A historical look to the future. Ann N Y Acad Sci 899:136–147
Halliwell B, Aeschbach R, Loliger J, Aruoma OI (1995) The characterization of antioxidants. Food Chem Toxicol 33:601–617
Hazra B, Biswas S, Mandal N (2008) Antioxidant and free radical scavenging activity of Spondias pinnata. BMC Complement Altern Med 8:63
Heidelberger C, Freeman AE, Pienta RJ, Sivak A, Bertram JS, Casto BC, Dunkel VC, Francis MW, Kakunaga T, Little JB, Schechtman LM (1983) Cell transformation by chemical agents—a review and analysis of the literature: a report of the US Environmental Protection Agency Gene-Tox Program. Mutat Res/Rev Genet Toxicol 114(3):283–385
Heimbrook DC, Oliff A (1998) Therapeutic intervention and signaling. Current opinion in cell biology.10(2):284–288
Institute of Laboratory Animal Resources, Commission on Life Sciences and National Research Council (1996) Guide for the care and use of laboratory animals. National Academy Press, Washington DC, pp 21–55
Kao TC, Shyu MH, Yen GC (2009) Neuroprotective effects of glycyrrhizic acid and 27 18beta-glycyrrhetinic acid in PC12 cells via modulation of the PI3K/Akt pathway. J Agric Food Chem 57:754–761
Kao TC, Wu CH, Yen GC (2014) Bioactivity and potential health benefits of licorice. J Agric Food Chem 62(3):542–553
Kawakami M, Inagawa R, Hosokawa T, Saito T, Kurasaki M (2008) Mechanism of apoptosis induced by copper in PC12 cells. Food Chem Toxicol 46(6):2157–2164
Kozlowski H, Janicka-Klos A, Brasun J, Gaggelli E, Valensin D, Valensin G (2009) Copper, iron, and zinc ions homeostasis and their role in neurodegenerative disorders (metal uptake, transport, distribution and regulation). Coord Chem Rev 253(21):2665–2685
Linder C (2001) Copper and genomic stability in mammals. Mutat Res 475(1–2):141–152
Linder C (2012) The relationship of copper to DNA damage and damage prevention in humans. Mutat Res 733:83–91
Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4:118–126
Lu J, Zheng YL, Wu DM, Sun DX, Shan Q, Fan SH (2006) Trace amounts of copper induce neurotoxicity in the cholesterol-fed mice through apoptosis. FEBS Lett 580(28–29):6730–6740
MacGibbon GA, Lawlor PA, Sirimanne ES, Walton MR, Connor B, Young D, Williams C, Gluckman P, Faull RL, Hughes P, Dragunow M (1997) Bax expression in mammalian neurons undergoing apoptosis, and in Alzheimer’s disease hippocampus. Brain Res 750(1):223–234
Marklund SL (1985) Superoxide dismutase isoenzymes in tissues and plasma from New Zealand black mice, nude mice and normal BALB/c mice. Mutat Res Fundam Mol Mech Mutagen 148:129–134
Mitra S, Keswani T, Dey M, Bhattacharya S, Sarkar S, Goswami S, Ghosh N, Dutta A, Bhattacharyya A (2012) Copper-induced immunotoxicity involves cell cycle arrest and cell death in the spleen and thymus. Toxicology 293(1):78–88
Mitra S, Keswani T, Ghosh N, Goswami S, Datta A, Das S, Maity S, Bhattacharyya A (2013) Copper induced immunotoxicity promote differential apoptotic pathways in spleen and thymus. Toxicology 306:74–84
Monnot A, Behl M, Ho S (2011) Regulation of brain copper homeostasis by the brain barrier systems: effects of Fe overload and Fe-deficiency. Toxicol Appl Pharmacol 256(3):249–257
Montgomery HAC , Dymock JF (1961) The determination of nitrite in water. Analyst. 86(102):414–416
Ozturk P, Kurutas EB, Ataseven A (2013) Copper/zinc and copper/selenium ratios, and oxidative stress as biochemical markers in recurrent aphthous stomatitis. J Trace Elem Med Biol 27(4):312–316
Paget GE , Barnes JM (1964) Toxicity tests. In: Lawrence D R, Bacharach A L (eds) Evaluation of drug activities: pharmacokinetics, Vol 1. Academic Press, London, pp 135-165
Pal A (2014) Copper toxicity induced hepatocerebral and neurodegenerative diseases: an urgent need for prognostic biomarkers. Neurotoxicology 40:97–101
Pal A, Prasad R (2014) Recent discoveries on the functions of astrocytes in the copper homeostasis of the brain: a brief update. Neurotox Res 26(1):78–84
Pal A, Badyal RK, Vasishta RK, Attri SV, Thapa BR, Prasad R (2013) Biochemical, histological, and memory impairment effects of chronic copper toxicity: a model for non-Wilsonian brain copper toxicosis in Wistar rat. Biol Trace Elem Res 153(1–3):257–268
Pal A, Siotto M, Prasad R, Squitti R (2015) Towards a unified vision of copper involvement in Alzheimer’s disease: a review connecting basic, experimental, and clinical research. J Alzheimers Dis 44(2):343–354
Pan HZ, Zhang H, Chang D, Li H, Sui H (2008) The change of oxidative stress products in diabetes mellitus and diabetic retinopathy. Br J Ophthalmol 92(4):548–551
Pena LB, Azpilicueta CE, Gallego SM (2011) Sunflower cotyledons cope with copper stress by inducing catalase subunits less sensitive to oxidation. J Trace Elem Med Biol 25(3):125–129
Pietrangelo A, Panduro A, Chowdhury JR, Shafritz DA (1992) Albumin gene expression is down-regulated by albumin or macromolecule infusion in the rat. J Clin Investig 89(6):1755
Prá D, Franke SI, Giulian R, Yoneama ML, Dias JF, Erdtmann B, Henriques JA (2008) Genotoxicity and mutagenicity of iron and copper in mice. Biometals 21(3):289–297
Ramos-Vara JA, Kiupel M, Baszler T, Bliven L, Brodersen B, Chelack B, Czub S, Del Piero F, Dial S, Ehrhart EJ, Graham T (2008) American association of veterinary laboratory diagnosticians subcommittee on standardization of immunohistochemistry suggested guidelines for immunohistochemical techniques in veterinary diagnostic laboratories. J Vet Diagn Investig 20:393–413
Rivera-Mancía S, Pérez-Neri I, Ríos C, Tristán-López L, Rivera-Espinosa L, Montes S (2010) The transition metals copper and iron in neurodegenerative diseases. Chem Biol Interact 186(2):184–199
Roy DN, Mandal S, Sen G, Biswas T (2009) Superoxide anion mediated mitochondrial dysfunction leads to hepatocyte apoptosis preferentially in the periportal region during copper toxicity in rats. Chem Biol Interact 182(2):136–147
Sakr SA, ElKenawy A, El-Sahara D (2009) Protective effect of licorice on metiram fungicide induced liver injury in mice. Can J Pure Appl Sci 3(2):787–793
Schuler M, Green DR (2001) Mechanisms of p53-dependent apoptosis. Biochem Soc Trans 29(6):684–688
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175(1):184–191
Tiffany-Castiglioni E, Hong S, Qian Y (2011) Copper handling by astrocytes: insights into neurodegenerative diseases. Int J Dev Neurosci 29(8):811–818
Uchiyama M, Mihara M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Valko M, Izakovic M, Mazur M, Rhodes CJ, Telser J (2004) Role of oxygen radicals in DNA damage and cancer incidence. Mol Cell Biochem 266(1–2):37–56
Xu CL, Wang QZ, Sun LM, Li XM, Deng JM, Li LF, Zhang J, Xu R, Ma SP (2012) Asiaticoside: attenuation of neurotoxicity induced by MPTP in a rat model of Parkinsonism via maintaining redox balance and up-regulating the ratio of Bcl-2/Bax. Pharmacol Biochem Behav 100(3):413–418
Zeiger E (2010) Genetic toxicology testing. Elsevier Ltd, Chapel Hill, pp 139–158
Zhai Q, Ji H, Zheng Z, Yu X, Sun L, Liu X (2000) Copper induces apoptosis in BA/F3β cells: Bax, reactive oxygen species, and NFκB are involved. J Cell Physiol 184(2):161–170
Zhao Z, Wang W, Guo H, Zhou D (2008) Antidepressant-like effect of liquiritin from Glycyrrhiza uralensis in chronic variable stress induced depression model rats. Behav Brain Res 194(1):108–113
Acknowledgements
Sincere appreciation is expressed to all members in forensic Medicine and Clinical Toxicology Department for their encouragement and special gratefulness for Prof. Dr. Mei Samir for her valuable guidance and support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiment was carried out in compliance with the guidelines of Institute of Laboratory Animal Resources, Commission on Life Sciences and National Research Council (1996) and the institutional guidelines for the care and use of experimental animals approved by the Medical Research Ethics Committee of Zagazig University, Egypt.
Conflicts of interest
The authors declare that they have no conflict of interest.
Funding source
This study was not funded by any source.
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
ESM 1
(DOCX 31 kb).
Rights and permissions
About this article
Cite this article
Mostafa, H.ES., Alaa-Eldin, E.A., El-Shafei, D.A. et al. Alleviative effect of licorice on copper chloride-induced oxidative stress in the brain: biochemical, histopathological, immunohistochemical, and genotoxic study. Environ Sci Pollut Res 24, 18585–18595 (2017). https://doi.org/10.1007/s11356-017-9503-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9503-6