Abstract
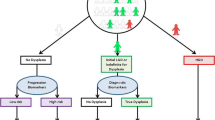
Risk stratification of patients with Barrett’s esophagus (BE) presently relies on the histopathologic grade of dysplasia found in esophageal biopsies, which is limited by sampling error and inter-pathologist variability. p53 immunostaining of BE biopsies has shown promise as an adjunct tool but is not recommended by American gastroenterology societies, who cite insufficient evidence of its prognostic value. We have conducted a systematic review and meta-analyses to clarify this value. We searched for studies that: (1) used immunohistochemistry to assess p53 expression in esophageal biopsies of BE patients and (2) reported subsequent neoplastic progression. We performed separate meta-analyses of case-control studies and cohort studies. We identified 14 relevant reports describing 8 case-control studies comprising 1435 patients and 7 cohort studies comprising 582 patients. In the case-control study meta-analysis of the risk of neoplasia with aberrant p53 expression, the fixed- and random-effect estimates of average effect size with aberrant p53 expression were OR 3.84, p < .001 (95% CI 2.79–5.27) and OR 5.95, p < .001 (95% CI 2.68–13.22), respectively. In the cohort study meta-analysis, the fixed- and random-effect estimates of average effect size were RR = 17.31, p < .001 (95% CI 9.35–32.08) and RR = 14.25, p < .001 (95% CI 6.76–30.02), respectively. Separate meta-analyses of case-control and cohort studies of BE patients who had baseline biopsies with p53 immunostaining revealed consistent, strong, and significant associations between aberrant p53 immunostaining and progression to high-grade dysplasia or esophageal adenocarcinoma. These findings support the use of p53 immunostaining as an adjunct to routine clinical diagnosis for dysplasia in BE patients.


Similar content being viewed by others
References
Pohl H, Sirovich B, Welch HG. Esophageal adenocarcinoma incidence: are we reaching the peak? Cancer Epidemiol Biomarkers Prev. 2010;19:1468–1470.
Hvid-Jensen F, Pedersen L, Drewes AM, Sorensen HT, Funch-Jensen P. Incidence of adenocarcinoma among patients with Barrett’s esophagus. N Engl J Med. 2011;365:1375–1383.
Naini BV, Souza RF, Odze RD. Barrett’s esophagus: a comprehensive and contemporary review for pathologists. Am J Surg Pathol. 2016;40:e45–e66.
Shaheen NJ, Sharma P, Overholt BF, et al. Radiofrequency ablation in Barrett’s esophagus with dysplasia. N Engl J Med. 2009;360:2277–2288.
Desai M, Saligram S, Gupta N, et al. Efficacy and safety outcomes of multimodal endoscopic eradication therapy in Barrett’s esophagus-related neoplasia: a systematic review and pooled analysis. Gastrointest Endosc. 2017;85:482–495.
Fitzgerald RC, di Pietro M, Ragunath K, et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett’s oesophagus. Gut. 2014;63:7–42.
Spechler SJ, Sharma P, Souza RF, Inadomi JM, Shaheen NJ. American Gastroenterological Association medical position statement on the management of Barrett’s esophagus. Gastroenterology. 2011;140:1084–1091.
Wani S, Rubenstein JH, Vieth M, Bergman J. Diagnosis and management of low-grade dysplasia in Barrett’s esophagus: expert review from the clinical practice updates committee of the American Gastroenterological Association. Gastroenterology. 2016;151:822–835.
Shaheen NJ, Falk GW, Iyer PG, Gerson LB. Diagnosis and management of Barrett’s esophagus. Am J Gastroenterol. 2016;111:30–50.
Evans JA, Early DS, Fukami N, et al. The role of endoscopy in Barrett’s esophagus and other premalignant conditions of the esophagus. Gastrointest Endosc. 2012;76:1087–1094.
Sharma P. Low-grade dysplasia in Barrett’s esophagus. Gastroenterology. 2004;127:1233–1238.
Curvers WL, ten Kate FJ, Krishnadath KK, et al. Low-grade dysplasia in Barrett’s esophagus: overdiagnosed and underestimated. Am J Gastroenterol. 2010;105:1523–1530.
Reid BJ, Weinstein WM, Lewin KJ, et al. Endoscopic biopsy can detect high-grade dysplasia or early adenocarcinoma in Barrett’s esophagus without grossly recognizable neoplastic lesions. Gastroenterology. 1988;94:81–90.
Cameron AJ, Carpenter HA. Barrett’s esophagus, high-grade dysplasia, and early adenocarcinoma: a pathological study. Am J Gastroenterol. 1997;92:586–591.
Lopes CV, Pereira-Lima J, Hartmann AA. P53 immunohistochemical expression in Barrett’s esophagus before and after endoscopic ablation by argon plasma coagulation. Scand J Gastroenterol. 2005;40:259–263.
Konda VJ, Cherkezyan L, Subramanian H, et al. Nanoscale markers of esophageal field carcinogenesis: potential implications for esophageal cancer screening. Endoscopy. 2013;45:983–988.
Kerkhof M, Steyerberg EW, Kusters JG, et al. Aneuploidy and high expression of p53 and Ki67 is associated with neoplastic progression in Barrett esophagus. Cancer Biomark. 2008;4:1–10.
Schulmann K, Sterian A, Berki A, et al. Inactivation of p16, RUNX3, and HPP1 occurs early in Barrett’s-associated neoplastic progression and predicts progression risk. Oncogene. 2005;24:4138–4148.
Jin Z, Cheng Y, Gu W, et al. A multicenter, double-blinded validation study of methylation biomarkers for progression prediction in Barrett’s esophagus. Cancer Res. 2009;69:4112–4115.
Choi W-T, Tsai J-H, Rabinovitch PS, et al. Diagnosis and risk stratification of Barrett’s dysplasia by flow cytometric DNA analysis of paraffin-embedded tissue. Gut. 2018;67:1229–1238.
Stachler MD, Camarda ND, Deitrick C, et al. Detection of mutations in Barrett’s esophagus before progression to high-grade dysplasia or adenocarcinoma. Gastroenterology. 2018;155:156–167.
Khan S, Do KA, Kuhnert P, et al. Diagnostic value of p53 immunohistochemistry in Barrett’s esophagus: an endoscopic study. Pathology. 1998;30:136–140.
Kaye PV, Haider SA, James PD, et al. Novel staining pattern of p53 in Barrett’s dysplasia—the absent pattern. Histopathology. 2010;57:933–935.
Kastelein F, Biermann K, Steyerberg EW, et al. Aberrant p53 protein expression is associated with an increased risk of neoplastic progression in patients with Barrett’s oesophagus. Gut. 2013;62:1676–1683.
Younes M, Brown K, Lauwers GY, et al. P53 protein accumulation predicts malignant progression in Barrett’s metaplasia: a prospective study of 275 patients. Histopathology. 2017;71:27–33.
Wells G, Shea B, O’Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa: Ottawa Hospital Research Institute. Available at: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp; 2012. Accessed 30.11.2017.
Bani-Hani K, Martin IG, Hardie LJ, et al. Prospective study of cyclin D1 overexpression in Barrett’s esophagus: association with increased risk of adenocarcinoma. J Natl Cancer Inst. 2000;92:1316–1321.
Skacel M, Petras RE, Rybicki LA, et al. p53 expression in low grade dysplasia in Barrett’s esophagus: correlation with interobserver agreement and disease progression. Am J Gastroenterol. 2002;97:2508–2513.
Murray L, Sedo A, Scott M, et al. TP53 and progression from Barrett’s metaplasia to oesophageal adenocarcinoma in a UK population cohort. Gut. 2006;55:1390–1397.
Bird-Lieberman EL, Dunn JM, Coleman HG, et al. Population-based study reveals new risk-stratification biomarker panel for Barrett’s esophagus. Gastroenterology. 2012;143:927–935.
Horvath B, Singh P, Xie H, Thota PN, Sun X, Liu X. Expression of p53 predicts risk of prevalent and incident advanced neoplasia in patients with Barrett’s esophagus and epithelial changes indefinite for dysplasia. Gastroenterol Rep. 2016;4:304–309.
Younes M, Lebovitz RM, Lechago LV, Lechago J. P53 protein accumulation in Barrett’s metaplasia, dysplasia, and carcinoma: a follow-up study. Gastroenterology. 1993;105:1637–1642.
Kim R, Clarke MR, Melhem MF, et al. Expression of p53, PCNA, and C-erbB-2 in Barrett’s metaplasia and adenocarcinoma. Dig Dis Sci. 1997;42:2453–2462. https://doi.org/10.1023/A:1018891923998.
Younes M, Ertan A, Lechago LV, Somoano JR, Lechago J. p53 protein accumulation is a specific marker of malignant potential in Barrett’s metaplasia. Dig Dis Sci. 1997;42:697–701. https://doi.org/10.1023/A:1018828207371.
Gimenez A, de Haro LM, Parrilla P, Bermejo J, Perez-Guillermo M, Ortiz MA. Immunohistochemical detection of p53 protein could improve the management of some patients with Barrett esophagus and mild histologic alterations. Arch Pathol Lab Med. 1999;123:1260–1263.
Weston AP, Banerjee SK, Sharma P, Tran TM, Richards R, Cherian R. p53 protein overexpression in low grade dysplasia (LGD) in Barrett’s esophagus: immunohistochemical marker predictive of progression. Am J Gastroenterol. 2001;96:1355–1362.
Davelaar AL, Calpe S, Lau L, et al. Aberrant TP53 detected by combining immunohistochemistry and DNA-FISH improves Barrett’s esophagus progression prediction: a prospective follow-up study. Genes Chromosomes Cancer. 2015;54:82–90.
Reid BJ, Prevo LJ, Galipeau PC, et al. Predictors of progression in Barrett’s esophagus II: baseline 17p (p53) loss of heterozygosity identifies a patient subset at increased risk for neoplastic progression. Am J Gastroenterol. 2001;96:2839–2848.
Timmer MR, Martinez P, Lau CT, et al. Derivation of genetic biomarkers for cancer risk stratification in Barrett’s oesophagus: a prospective cohort study. Gut. 2016;65:1602–1610.
Casson AG, Manolopoulos B, Troster M, et al. Clinical implications of p53 gene mutation in the progression of Barrett’s epithelium to invasive esophageal cancer. Am J Surg. 1994;167:52–57.
Maley CC, Galipeau PC, Li X, et al. The combination of genetic instability and clonal expansion predicts progression to esophageal adenocarcinoma. Cancer Res. 2004;64:7629–7633.
Altaf K, Xiong JJ, la Iglesia D, Hickey L, Kaul A. Meta-analysis of biomarkers predicting risk of malignant progression in Barrett’s oesophagus. Br J Surg. 2017;104:493–502.
Janmaat VT, van Olphen SH, Biermann KE, Looijenga LHJ, Bruno MB, Spaander MCW. Use of immunohistochemical biomarkers as independent predictor of neoplastic progression in Barrett’s oesophagus surveillance: a systematic review and meta-analysis. PLoS ONE. 2017;12:1–16.
Author information
Authors and Affiliations
Contributions
PS collected, analyzed, and interpreted the data, and drafted the manuscript. KD and RFS analyzed and interpreted the data, and critically revised the manuscript for important intellectual content. SJS conceived the topic, analyzed and interpreted the data, and critically revised the manuscript for important intellectual content. DJC evaluated the performance of statistical analyses and critically revised the manuscript for important intellectual content. VJAK conceived the topic, analyzed and interpreted the data, critically revised the manuscript for important intellectual content, and was responsible for the final approval.
Corresponding author
Ethics declarations
Conflict of interest
Rhonda F. Souza has served as a consultant and receives research support from Ironwood Pharmaceuticals. Stuart Jon Spechler has served as a consultant for Ironwood Pharmaceuticals. Vani J.A. Konda has received grant support from Pentax Medical and Ironwood Pharmaceuticals. Patrick Snyder, Kerry Dunbar, and Daisha J. Cipher have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Snyder, P., Dunbar, K., Cipher, D.J. et al. Aberrant p53 Immunostaining in Barrett’s Esophagus Predicts Neoplastic Progression: Systematic Review and Meta-Analyses. Dig Dis Sci 64, 1089–1097 (2019). https://doi.org/10.1007/s10620-019-05586-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05586-7