Abstract
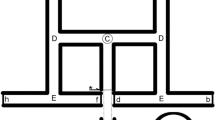
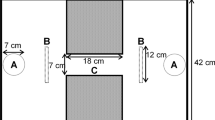
Locomotor performance provides one of the key pieces of information regarding whole-organism function. Experiments encompassing behavioral data commonly endeavor to measure parameters such as burst speed, latency time, distance traveled, and other aspects of locomotion. Behavioral experiments can uncover an immense range of information, from the individual, interspecific, and intraspecific levels up to correlations with ecological factors and parameters from the ecosystem. Here, we explored the locomotor behavior of two lizard species, Lacerta viridis and Lacerta agilis, in an open field test (OFT). The main aim was to reveal changes in locomotion over time. Although we observed no time-related variation in L. agilis, we discovered significant changes in locomotor behavior over the course of the experiment in Lacerta viridis. Measured behavioral traits (resting time, total distance traveled, mean speed) showed significant changes across time in L. viridis, thus indicating the importance of time as a factor when conducting behavioral experiments. Moreover, we observed that in 10-min experimental session, the individuals have undergone different stages from freezing behavior, exploration, to habituation.





Similar content being viewed by others
References
Adolph SC, Pickering T (2008) Estimating maximum performance: effects of intraindividual variation. J Exp Biol 211:1336–1343. https://doi.org/10.1242/jeb.011296
Adolph SC, Porter WP (1993) Temperature, activity, and lizard life histories. Am Nat 142:273–295. https://doi.org/10.1086/285538
Amaral MJ, Carretero MA, Bicho RC, Soares AM, Mann RM (2012b) The use of a lacertid lizard as a model for reptile ecotoxicology studies. Part 1 Field demographics and morphology. Chemosphere 87:757–764. https://doi.org/10.1016/j.chemosphere.2011.12.075
Amaral MJ, Bicho RC, Carretero MA, Sanchez-Hernandez JC, Faustino AM, Soares AM, RM b M (2012a) The use of a lacertid lizard as a model for reptile ecotoxicology studies. Part 2 Biomarkers of exposure and toxicity among pesticide exposed lizards. Chemosphere 87(7):765–774. https://doi.org/10.1016/j.chemosphere.2012.01.048
Amat F, Llorente, GA and Carretero MA (2003) A preliminary study on thermal ecology, activity times and microhabitat use of Lacerta agilis (Squamata: Lacertidae) in the Pyrenees. FOLIA ZOOLOGICAPRAHA 52(4)413–422
Arnold SJ (1983) Morphology, performance and fitness. Am Zool 23:347–361. https://doi.org/10.1093/icb/23.2.347
Arnold SJ, Bennett AF (1988) Behavioural variation in natural populations. V. Morphological correlates of locomotion in the garter snake (Thamnophis radix). Biol J Lin Soc 34:175–190. https://doi.org/10.1111/j.1095-8312.1988.tb01955.x
Avery RA, Mueller CF, Jones SM, Smith JA, Bond DJ (1987) Speeds and movement patterns of European lacertid lizards: a comparative study. J Herpetol 21:324–329. https://doi.org/10.2307/1563974
Bajer K, Horváth G, Molnár O, Török J, Garamszegi LZ, Herczeg G (2015) European green lizard (Lacerta viridis) personalities: linking behavioural types to ecologically relevant traits at different ontogenetic stages. Behav Process 111:67–74. https://doi.org/10.1016/j.beproc.2014.11.020
Bauwens D, Garland T, Castilla AM, Van Damme R (1995) Evolution of sprint speed in lacertid lizards: morphological, physiological, and behavioral covariation. Evolution 49:848–863. https://doi.org/10.2307/2410408
Bulte G, Blouin-Demers G (2006) Cautionary notes on the descriptive analysis of performance curves in reptiles. J Therm Biol 31:287–291. https://doi.org/10.1016/j.jtherbio.2005.11.030
Cavitt JF (2000) Fire and a tallgrass prairie reptile community: effects on relative abundance and seasonal activity. J Herpetol 34:12–20. https://doi.org/10.2307/1565233
Crews D, Gans C (1992) The interaction of hormones, brain, and behavior: an emerging discipline in herpetology. Biology of the Reptilia. Vol 18. Physiology E. Hormones, Brain, and Behavior. University of Chicago Press, Chicago, pp 1–23
Choleris E, Thomas AW, Kavaliers M, Prato FS (2001) A detailed ethological analysis of the mouse open field test: effects of diazepam, chlordiazepoxide and an extremely low frequency pulsed magnetic field. Neurosci Biobehav Rev 25:235–260. https://doi.org/10.1016/S0149-7634(01)00011-2
Ekner-Grzyb A, Sajkowska Z, Dudek K, Gawałek M, Skórka P, Tryjanowski P (2013) Locomotor performance of sand lizards (Lacerta agilis): effects of predatory pressure and parasite load. Acta ethol 16:173–179. https://doi.org/10.1007/s10211-013-0148-2
Garland T Jr, Losos JB (1994) Ecological morphology of locomotor performance in squamate reptiles. In: Wainwright PC, Reilly S (eds) Ecological morphology: integrative organismal biology, 1st edn. University of Chicago Press, Chicago, pp 240–302
Garland T (1984) Physiological correlates of locomotory performance in a lizard: an allometric approach. Am J Physiol Regul Integr Comp Physiol 247:R806–R815
Garrido M, Pérez-Mellado V (2013) Sprint speed is related to blood parasites, but not to ectoparasites, in an insular population of lacertid lizards. Can J Zool 92:67–72. https://doi.org/10.1139/cjz-2013-0092
Garrido M, Pérez-Mellado V, Cooper WE (2015) Complex relationships amongst parasite load and escape behaviour in an insular lizard. Ethology 121:116–124. https://doi.org/10.1111/eth.12322
Gleeson TT, Harrison JM (1988) Muscle composition and its relation to sprint running in the lizard Dipsosaurus dorsalis. Am J Physiol Regul Integr Comp Physiol 255:R470–R477
Greenberg N, Burghardt GM, Crews D, Font E, Jones RE, Vaughan G (1989) Reptile models for biomedical research. Woodhead AD ed. Nonmammalian animal models for biomedical research. Boca Raton: CRC Press, Inc. p 289–308
Huey RB, Pianka ER (1977) Seasonal variation in thermoregulatory behavior and body temperature of diurnal Kalahari lizards. Ecology 58:1066–1075. https://doi.org/10.1655/08-027.1
Huey RB, Stevenson RD (1979) Integrating thermal physiology and ecology of ectotherms: a discussion of approaches. Amer Zool 19:357–366. https://doi.org/10.1093/icb/19.1.357
Husak JF (2006) Does survival depend on how fast you can run or how fast you do run? Funct Ecol 20:1080–1086. https://doi.org/10.1111/j.1365-2435.2006.01195.x
Husak JF, Fox SF (2006) Field use of maximal sprint speed by collared lizards (Crotaphytus collaris): compensation and sexual selection. Evolution 60:1888–1895. https://doi.org/10.1111/j.0014-3820.2006.tb00532.x
Irschick DJ, Garland Jr T (2001) Integrating function and ecology in studies of adaptation: investigations of locomotor capacity as a model system. Annu Rev Ecol Syst 32:367–396. https://doi.org/10.1146/annurev.ecolsys.32.081501.114048
Jayne BC, Bennett AF (1990) Selection on locomotor performance capacity in a natural population of garter snakes. Evolution 44:1204–1229. https://doi.org/10.2307/2409283
Kaliontzopoulou A, Carretero MA, Llorente GA (2010) Intraspecific ecomorphological variation: linear and geometric morphometrics reveal habitat-related patterns within Podarcis bocagei wall lizards. J Evol Biol 23:1234–1244. https://doi.org/10.1111/j.1420-9101.2010.01984.x
Kramer DL, McLaughlin RL (2001) The behavioral ecology of intermittent locomotion. Amer Zool 41:137–153. https://doi.org/10.1093/icb/41.2.137
Le Galliard JF, Clobert J, Ferriere R (2004) Physical performance and Darwinian fitness in lizards. Nature 432:502–505. https://doi.org/10.1038/nature03057
Licht P (1972) Environmental physiology of reptilian breeding cycles: role of temperature. Gen Comp Endocrinol 3:477–488. https://doi.org/10.1016/0016-6480(72)90178-5
López P, Hawlena D, Polo V, Amo L, Martín J (2005) Sources of individual shy–bold variations in antipredator behaviour of male Iberian rock lizards. Anim Behav 69:1–9. https://doi.org/10.1016/j.anbehav.2004.05.010.
Lovern MB, Holmes MM, Wade J (2004) The green anole (Anolis carolinensis): a reptilian model for laboratory studies of reproductive morphology and behavior. ILAR J 45(1):54–64
Miles DB (2004) The race goes to the swift: fitness consequences of variation in sprint performance in juvenile lizards. Evol Ecol Res 6:63–75
Miles DB, Calsbeek R, Sinervo B (2007) Corticosterone, locomotor performance, and metabolism in side-blotched lizards (Uta stansburiana). Horm Behav 51:548–554. https://doi.org/10.1016/j.yhbeh.2007.02.005
Montuelle SJ, Herrel A, Libourel PA, Daillie S, Bels VL (2012) Flexibility in locomotor–feeding integration during prey capture in varanid lizards: effects of prey size and velocity. J Exp Biol 215:3823–3835. https://doi.org/10.1242/jeb.072074
Oppliger A, Celerier ML, Clobert J (1996) Physiological and behaviour changes in common lizards parasitized by haemogregarines. Parasitology 113:433–438. https://doi.org/10.1017/S003118200008149X
Peterson CC, Husak JF (2006) Locomotor performance and sexual selection: individual variation in sprint speed of collared lizards (Crotaphytus collaris). Copeia 2006:216–224. https://doi.org/10.1643/0045-8511(2006)6[216:LPASSI]2.0.CO;2
Rodríguez-Prieto I, Martín J, Fernández-Juricic E (2010) Habituation to low-risk predators improves body condition in lizards. Behav Ecol Sociobiol 64:1937–1945. https://doi.org/10.1007/s00265-010-1004-2
Tatem KS, Quinn JL, Phadke A, Yu Q, Gordish-Dressman H, Nagaraju K (2014) Behavioral and locomotor measurements using an open field activity monitoring system for skeletal muscle diseases. J Vis Exp 29:51785. https://doi.org/10.3791/51785
Van Damme R, Vanhooydonck B (2002) Speed versus manoeuvrability: association between vertebral number and habitat structure in lacertid lizards. J Zool 258:327–334. https://doi.org/10.1017/S0952836902001462
Vanhooydonck B, Van Damme R (2001) Evolutionary trade-offs in locomotor capacities in lacertid lizards: are splendid sprinters clumsy climbers? J Evol Biol 14:46–54. https://doi.org/10.1046/j.1420-9101.2001.00260.x
Vanhooydonck B, Measey J, Edwards S, Makhubo B, Tolley KA, Herrel A (2015) The effects of substratum on locomotor performance in lacertid lizards. Biol J Linn Soc 115(4):869–881
Wainwright PC, Reilly SM (1994) Ecological morphology: integrative organismal biology. University of Chicago Press, Chicago and London
Zamora-Camacho FJ, Reguera S, Moreno-Rueda G, Pleguezuelos JM (2013) Patterns of seasonal activity in a Mediterranean lizard along a 2200m altitudinal gradient. J Therm Biol 38:64–69. https://doi.org/10.1016/j.jtherbio.2012.11.002
Žagar A, Carretero MA, Osojnik N, Sillero N, Vrezec A (2015) A place in the sun: interspecific interference affects thermoregulation in coexisting lizards. Behav Ecol Sociobiol 69:1127–1137. https://doi.org/10.1007/s00265-015-1927-8
Acknowledgements
Natália Pipová, Tiberiu Sahlean, Gabriel Bogdan, and Peter Hiznan were of a great help in locating and capturing lizards. Monika Balogová was an excellent adviser on experiment design. Capturing lizards in protected areas was carried out with official permission no. 5498/2011-2.2 issued by the Ministry of Environment of the Slovak Republic. Capturing lizards in Romania was under supervision of Cluj Napoca University. This study was realized within the frame of project “Centrum of Excellence for Parasitology” no. 26220120022 supported by operating program “Research and Development” funded by European Fund for Regional Development and financially supported also by projects VVGS-PF-2015-483 and VEGA 1/0417/14 (0.6). The work of Anamarija Žagar was supported by the national program “Women in science” from L’Oreal and the Slovenian national commission for UNESCO. The linguistic part of the article was reviewed and edited by David Lee McLean.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Pačuta, A., Žagar, A., Kočíková, B. et al. Time matters. Locomotor behavior of Lacerta viridis and Lacerta agilis in an open field maze. acta ethol 21, 91–99 (2018). https://doi.org/10.1007/s10211-018-0287-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10211-018-0287-6