Abstract
Purpose
To evaluate the effectiveness and safety of image-guided percutaneous sclerotherapy using bleomycin for macrocystic and bevacizumab (Avastin™) for microcystic orbital lymphatic malformations in children.
Materials and Methods
Between October 2015 and July 2018, we prospectively evaluated 10 pediatric patients who presented clinically and radiologically with lymphatic malformations and were treated with percutaneous sclerotherapy. Patients with venous malformations were excluded. Eight females and two males with ages ranging from 3 to 17 years (mean: 8.8, SD: 4.9) were included. Guided with ultrasound and fluoroscopy, macrocysts were treated with bleomycin instillation. For microcystic components in three patients, bevacizumab was injected intralesional. All patients underwent ultrasound and non-contrast MRI to evaluate response to treatment after 6 weeks.
Results
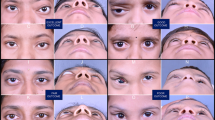
The malformations were macrocystic in seven patients and complex (macro/microcystic) in three. Twenty sclerotherapy sessions were performed, (range: 1–3 sessions, mean: 2, SD: 0.8). Clinically, there was a significant reduction in the proptosis after treatment (P = 0.007) and dystopia (P = 0.018). The local radiological response showed a reduction in the maximum lesions diameters and volumes after treatment (P = 0.005 and 0.005, respectively). Two of the three patients treated with bevacizumab showed a reduction in the lesions volumes by 90.4% and 63.4%, respectively, whereas one patient did not show volume reduction. Transient periorbital edema and ecchymosis occurred following the procedure with no major complications encountered. Follow-up ranged from 9–33 months, mean: 20.3, SD: 7.4.
Conclusion
Bleomycin sclerotherapy is a safe and effective treatment for orbital macrocystic lymphatic malformations. Further use of bevacizumab for microcystic lesions in a larger series is required to outline its efficacy and safety.



Similar content being viewed by others
References
Katz SE, Rootman J, Vangveeravong S, Graeb D. Combined venous lymphatic malformations of the orbit (so-called lymphangiomas): association with noncontiguous intracranial vascular anomalies. Ophthalmology. 1998;105(1):176–84.
Greene AK, Burrows PE, Smith L, et al. Periorbital lymphatic malformation: clinical course and management in 42 patients. Plast Reconstr Surg. 2005;115:22–30.
Mulligan PR, Prajapati HJS, Martin LG, Patel TH. Vascular anomalies: classification, imaging characteristics and implications for interventional radiology treatment approaches. Br J Radiol. 2014;87:1035.
Chung EM, Smirniotopoulos JG, Specht CS, Schroeder JW, Cube R. From the archives of the AFIP: pediatric orbit tumors and tumorlike lesions: non-osseous lesions of the extra ocular orbit. Radiographics. 2007;27:1777–99.
Rootman J, Heran MKS, Graeb DA. Vascular malformations of the orbit: classification and the role of imaging in diagnosis and treatment strategies. Ophthal Plast Reconstr Surg. 2014;30:91–104.
Tunc M, Sadri E, Char DH. Orbital lymphangiomas: an analysis of 26 patients. Br J Ophthalmol. 1999;83:76–80.
Gündüz K, Demirel S, Yagmurlu B, Erden E. Correlation of surgical outcome with neuroimaging findings in periocular lymphangiomas. Ophthalmology (Internet). 2006;113(7):1231–8.
Nassiri N, Rootman J, Rootman DB, Goldberg RA. Orbital lymphaticovenous malformations: current and future treatments. Surv Ophthalmol. 2015;60:383–405.
Güneyli S, Ceylan N, Bayraktaroğlu S, Acar T, Savaş R. Imaging findings of vascular lesions in the head and neck. Diagn Interv Radiol. 2014;20(5):432–7.
Harris GJ, Sakol PJ, Bonavolonta G, et al. An analysis of thirty cases of orbital lymphangiomas: pathophysiologic considerations and management recommendations. Ophthalmology. 1990;97:1583–92.
Barnacle AM, Theodorou M, Maling SJ, Abou-Rayyah Y. Sclerotherapy treatment of orbital lymphatic malformations: a large single-center experience. Br J Ophthalmol. 2016;100(2):204–8.
Wojno TH. Sotradecol (sodium tetradecyl sulfate) injection of orbital lymphangioma. Ophthal Plast Reconstr Surg. 1999;15:432–7.
Svendsen PA, Wikholm G, Rodriguez M, et al. Direct puncture and sclerotherapy with Sotradecol. Orbital lymphatic malformations. Interv Neuroradiol. 2001;30:193–9.
Poonyathalang A, Preechawat P, Jiarakongmun P, et al. Sclerosing therapy for orbital lymphangioma using sodium tetradecyl sulfate. Jpn J Ophthalmol. 2008;52:298–304.
Hill RH, Shiels WE, Foster JA, et al. Percutaneous drainage and ablation as first-line therapy for macrocystic and microcystic orbital lymphatic malformations. Ophthal Plast Recontr Surg. 2012;28:119–25.
Chiramel GK, Keshava SN, Moses V, Mammen S, David S, Sen S. Percutaneous sclerotherapy of congenital slow-flow vascular malformations of the orbit. Cardiovasc Intervent Radiol. 2015;38:270–9.
Patel KC, Kalantzis G, El-Hindy N, Chang BY. Sclerotherapy for orbital lymphangioma—case series and literature review. In Vivo. 2017;31(2):263–6.
Shen CY, Wu MC, Tyan YS, Ou CH, Chen TY, Wong HF. Preliminary experience of percutaneous intralesional bleomycin injection for the treatment of orbital lymphatic-venous malformation refractory to surgery. Clin Radiol. 2012;67:182–4.
Gooding C, Meyer D. Intralesional bleomycin: a potential treatment for refractory orbital lymphangiomas. Ophthal Plast Reconstr Surg. 2014;30:65–7.
Paramasivam S, Fay A, Fifi J, Berenstein A. O-015 Image guided bleomycin sclerotherapy for orbital lymphatic malformation. J Neuro Intervent Surg. 2014;6:A8–9.
Harmoush S, Chinnardurai P, El Salek K, et al. Multimodality image-guided sclerotherapy of low-flow orbital vascular malformations: report of single-center experience. J Vasc Interv Radiol. 2016;27(7):987–95.
Raichura N, Alam S, Noronha V, Mukherjee B. A prospective study of the role of intralesional bleomycin in orbital lymphangioma. J AAPOS. 2017;21:146–51.
Hanif AM, Saunders JA, Hawkins CM, Wojno TH, Kim HJ. Use of percutaneous bleomycin sclerotherapy for orbital lymphatic malformations. Orbit. 2018;14:1–7.
Suzuki Y, Obana A, Gohto Y, et al. Management of orbital lymphangioma using intralesional injection of OK-432. Br J Ophthalmol. 2000;84:614–7.
Woo YJ, Kim CY, Sgrignoli B, Yoon JS. Orbital lymphangioma: characteristics and treatment outcomes of 12 cases. Korean J Ophthalmol. 2017;31(3):194–201.
Yue H, Qian J, Elner VM, et al. Treatment of orbital vascular malformations with intralesional injection of pingyangmycin. Br J Ophthalmol. 2013;97:739–45.
Schwarcz RM, Ben Simon GJ, Cook T, et al. Sclerosing therapy as first-line treatment for low flow vascular lesions of the orbit. Am J Ophthalmol. 2006;141:333–9.
Thomas DM, Wieck MM, Grant CN, et al. Doxycycline sclerotherapy is superior in the treatment of pediatric lymphatic malformations. J Vasc Interv Radiol. 2016;27(12):1846–56.
Orford J, Barker A, Thonell S, King P, Murphy J. Bleomycin therapy for cystic hygroma. J Pediatr Surg. 1995;30:1282–7.
Baskin D, Tander B, Bankaoglu M. Local bleomycin injection in the treatment of lymphangioma. Eur J Pediatr Surg. 2005;15:383–6.
Muir T, Kirsten M, Fourie P, Dippenaar N, Ionescu GO. Intralesional bleomycin injection (IBI) treatment for haemangiomas and vascular malformations. Pediatr Surg Int. 2004;19:766–73.
Horbach SE, Rigter IM, Smitt JH, et al. Intralesional bleomycin injections for vascular malformations: a systematic review and meta-analysis. Plast Reconstr Surg. 2016;137:244–56.
Ferrara N, Hillan KJ, Gerber HP, Novotny W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat Rev Drug Discov. 2004;3(5):391–400.
Pasqualetti G, Danesi R, Del Tacca M, Bocci G. Vascular endothelial growth factor pharmacogenetics: a new perspective for anti-angiogenic therapy. Pharmacogenomics. 2007;8(1):49–66.
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419–31.
Arevalo JF, Lasave AF, Wu L, Diaz-Llopis M, Gallego-Pinazo R, Alezzandrini AA, et al. Intravitreal bevacizumab plus grid laser photocoagulation or intravitreal bevacizumab or grid laser photocoagulation for diffuse diabetic macular edema: results of the Pan-American Collaborative Retina Study Group at 24 months. Retina. 2013;33(2):403–13.
Wu L, Arevalo JF, Berrocal MH, Maia M, Roca JA, Morales-Cantón V, et al. Comparison of two doses of intravitreal bevacizumab as primary treatment for macular edema secondary to central retinal vein occlusion: results of the pan American collaborative retina study group at 24 months. Retina. 2010;30(7):1002–11.
Acknowledgements
The authors wish to acknowledge the help provided by Dr. Alex M Barnacle, Consultant Interventional Radiologist, Great Ormond Street Hospital for Children, NHS, London, UK. We would like to thank her for the expert advice and encouragement throughout this work. Her willingness to give her time so generously in editing the manuscript has been very much appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there are no conflicts of interest.
Ethical Standards
All procedures performed were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all legal guardians of the children.
Rights and permissions
About this article
Cite this article
Abdelaziz, O., Hassan, F., Elessawy, K. et al. Image-Guided Percutaneous Bleomycin and Bevacizumab Sclerotherapy of Orbital Lymphatic Malformations in Children. Cardiovasc Intervent Radiol 42, 433–440 (2019). https://doi.org/10.1007/s00270-018-2128-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-2128-4