Abstract
Introduction
Chemotherapy is increasingly administered prior to resection in patients with early-stage pancreatic adenocarcinoma, but the national prevalence of this practice is poorly understood. Our objectives were to (1) describe the utilization of upfront chemotherapy management of stage I pancreatic cancer; (2) define factors associated with the use of upfront chemotherapy and subsequent resection; and (3) assess hospital-level variability in upfront chemotherapy and subsequent resection.
Methods
The National Cancer Database was used to identify patients treated for clinical stage I pancreatic adenocarcinoma. Outcomes were receipt of upfront chemotherapy and surgical resection after upfront chemotherapy. Associations between patient/hospital factors and both initial management and subsequent resection were assessed by multivariable logistic regression.
Results
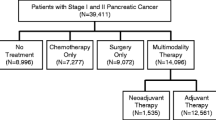
A total of 17,495 patients were included, with 26.6% receiving upfront chemotherapy. Upfront chemotherapy was more likely in patients who were ≥ 80 years of age (odds ratio [OR] 1.64, 95% confidence interval [CI] 1.39–1.93), had T2 tumors (OR 2.56, 95% CI 2.36–2.78), or were treated at a low-volume center (OR 2.10, 95% CI 1.63–2.71). Among patients receiving upfront chemotherapy, only 33.5% underwent subsequent resection. Resection was more likely in patients with T1 tumors (OR 1.22, 95% CI 1.04–1.43) and in those treated at high-volume centers (OR 4.03, 95% CI 2.90–5.60). Only 20.4% of hospitals performed resection in > 50% of patients after upfront chemotherapy.
Conclusion
Rates of surgical resection after upfront chemotherapy are relatively low, and the proportion of patients who eventually undergo resection varies considerably between hospitals. The use of surgery after upfront chemotherapy in resectable pancreatic cancer should be considered as an internal quality-of-cancer-care measure.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin 2019;69:7–34.
Miller KD, Siegel RL, Lin CC, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 2016;66:271–89.
Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol 2008;15:683–90.
Riall TS, Nealon WH, Goodwin JS, et al. Pancreatic cancer in the general population: Improvements in survival over the last decade. J Gastrointest Surg. 2006;10:1212–23; discussion 23–4.
Schnelldorfer T, Ware AL, Sarr MG, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Ann Surg. 2008;247:456–62.
Riall TS, Cameron JL, Lillemoe KD, et al. Resected periampullary adenocarcinoma: 5-year survivors and their 6- to 10-year follow-up. Surgery. 2006;140:764–72.
Piatek M, Kusnierz K, Bienkowski M, Peksa R, Kowalczyk M, Nawrocki S. Primarily resectable pancreatic adenocarcinoma—to operate or to refer the patient to an oncologist? Crit Rev Oncol/Hematol 2019;135:95–102.
Tempero MA, Malafa MP, Al-Hawary M, et al. Pancreatic adenocarcinoma, version 2.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2017;15:1028–61.
Khorana AA, Mangu PB, Katz MHG. Potentially curable pancreatic cancer: American society of clinical oncology clinical practice guideline update summary. J Oncol Pract 2017;13:388–91.
Belli C, Cereda S, Anand S, Reni M. Neoadjuvant therapy in resectable pancreatic cancer: a critical review. Cancer Treat Rev 2013;39:518–24.
Dhir M, Malhotra GK, Sohal DPS, et al. Neoadjuvant treatment of pancreatic adenocarcinoma: a systematic review and meta-analysis of 5520 patients. World J Surg Oncol 2017;15:183.
Bilimoria KY, Bentrem DJ, Ko CY, Stewart AK, Winchester DP, Talamonti MS. National failure to operate on early stage pancreatic cancer. Ann Surg 2007;246:173–80.
Zhan HX, Xu JW, Wu D, et al. Neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of prospective studies. Cancer Med 2017;6:1201–19.
About the Commission on Cancer. 2018. Available at: https://www.facs.org/quality-programs/cancer/coc/about. Accessed 21 Dec 2018.
Cancer Staging Manual. 2018. Available at: https://cancerstaging.org/references-tools/deskreferences/Pages/default.aspx. Accessed 21 Dec 2018.
Participant User Files. 2018. Available at: https://www.facs.org/quality-programs/cancer/ncdb/puf. Accessed 21 Dec 2018.
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol 1992;45:613–9.
Merlo J, Yang M, Chaix B, Lynch J, Rastam L. A brief conceptual tutorial on multilevel analysis in social epidemiology: investigating contextual phenomena in different groups of people. J Epidemiol Commun Health 2005;59:729–36.
Merlo J, Chaix B, Ohlsson H, et al. A brief conceptual tutorial of multilevel analysis in social epidemiology: using measures of clustering in multilevel logistic regression to investigate contextual phenomena. J Epidemiol Commun Health 2006;60:290–7.
Merkow RP, Bilimoria KY, Tomlinson JS, et al. Postoperative complications reduce adjuvant chemotherapy use in resectable pancreatic cancer. Ann Surg 2014;260:372–7.
Swords DS, Mulvihill SJ, Brooke BS, Skarda DE, Firpo MA, Scaife CL. Disparities in utilization of treatment for clinical stage I-II pancreatic adenocarcinoma by area socioeconomic status and race/ethnicity. Surgery 2019;165(4):751–759.
Khawja SN, Mohammed S, Silberfein EJ, Musher BL, Fisher WE, Van Buren G. Pancreatic cancer disparities in African Americans. Pancreas 2015;44:522–7.
Moaven O, Richman JS, Reddy S, Wang T, Heslin MJ, Contreras CM. Healthcare disparities in outcomes of patients with resectable pancreatic cancer. Am J Surg 2019;217:725–31.
Tohme S, Kaltenmeier C, Bou-Samra P, Varley PR, Tsung A. Race and health disparities in patient refusal of surgery for early-stage pancreatic cancer: an NCDB cohort study. Ann Surg Oncol 2018;25:3427–35.
Funding
As an organization, the American College of Surgeons had no role in the design and conduct of the study; analysis and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The views expressed in this work represent those of the authors only. RJE (Agency for Healthcare Research and Quality [AHRQ] 5T32HS000078) was supported by a postdoctoral research fellowship and the American College of Surgeons Clinical Scholars in Residence Program. RPM is supported by the Agency for Healthcare Quality (K12HS023011) and an Institutional Research Grant from the American Cancer Society (IRG-18-163-24). ADY is supported by the National Heart, Lung, and Blood Institute (NHLBI) of the National Institutes of Health (K08HL145139).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors report no conflicts of interest, financial or otherwise, related to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ellis, R.J., Ho, J.W., Schlick, C.J.R. et al. National Use of Chemotherapy in Initial Management of Stage I Pancreatic Cancer and Failure to Perform Subsequent Resection. Ann Surg Oncol 27, 909–918 (2020). https://doi.org/10.1245/s10434-019-08023-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-08023-1