Abstract
Background
The prognosis of patients with cholangiocarcinoma is unsatisfactory. Therefore, evaluation of prognostic factors and establishment of new therapeutic strategies are needed to improve their long-term survival. The aim of this study was to identify useful prognostic factors for patients with intrahepatic, hilar, and distal cholangiocarcinoma.
Materials and Methods
Records of 127 patients with cholangiocarcinoma (21 with intrahepatic cholangiocarcinoma, 50 with hilar cholangiocarcinoma, and 56 with distal cholangiocarcinoma) who underwent surgical resection were reviewed retrospectively. Relationships between survival and clinicopathological factors including patient demographics and tumor characteristics were evaluated using univariate and multivariate analysis.
Results
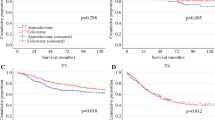
For all 127 patients, overall 1-, 3-, 5-year survival rates were 80, 51, and 40%, respectively. Univariate analysis revealed that adjuvant chemotherapy (P = .049), tumor differentiation (P = .014), lymph node metastasis (P < .001), surgical margin status (P < .001), UICC pT factor (P < .001), and UICC stage (P < .001) were associated significantly with survival. UICC pT factor (P = .007), adjuvant chemotherapy (P = .009), surgical margin status (P = .012), and lymph node metastasis (P = .014) remained independently associated with long-term survival by multivariate analysis. The 5-year survival rates of patients with or without positive surgical margins were 13 and 49%, respectively. The 5-year survival rates of patients treated with or without adjuvant chemotherapy were 47 and 36%, respectively.
Conclusions
R0 resection and adjuvant chemotherapy may be mandatory to achieve long-term survival for patients with cholangiocarcinoma.



Similar content being viewed by others
References
Khan SA, Taylor-Robinson SD, Toledano MB, Beck A, Elliott P, Thomas HC. Changing international trends in mortality rates for liver, biliary and pancreatic tumors. J Hepatol. 2002;37:806–13.
Furuse J. Postoperative adjuvant treatments for biliary tract cancer. J Hepatobiliary Pancreat Surg. 2008;15:463–7.
Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, et al. Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors. Ann Surg. 1996;22:463–75.
Shimada K, Sano T, Sakamoto Y, Esaki M, Kosuge T, Ojima H. Surgical outcomes of the mass-forming plus periductal infiltrating types of intrahepatic cholangiocarcinoma: a comparative study with the typical mass-forming type of intrahepatic cholangiocarcinoma. World J Surg. 2007;31:2016–22.
Konstadoulakis MM, Roayaie S, Gomatos IP, Labow D, Fiel MI, Miller CM, et al. Fifteen-year, single-center experience with the surgical management of intrahepatic cholangiocarcinoma: operative results and long-term outcome. Surgery. 2008;143:366–74.
Uenishi T, Kubo S, Yamazaki O, Yamada T, Sasaki Y, Nagano H, et al. Indications for surgical treatment of intrahepatic cholangiocarcinoma with lymph node metastases. J Hepatobiliary Pancreat Surg. 2008;15:417–22.
Nakagohri T, Kinoshita T, Konishi M, Takahashi S, Gotohda N. Surgical outcome and prognostic factors in intrahepatic cholangiocarcinoma. World J Surg. 2008;32:2675–80.
Guglielmi A, Ruzzenente A, Campagnaro T, Pachera S, Valdegamberi A, Nicoli P, et al. Intrahepatic cholangiocarcinoma: prognostic factors after surgical resection. World J Surg. 2009;33:1247–54.
Murakami Y, Uemura K, Sudo T, Hayashidani Y, Hashimoto Y, Nakamura H, et al. Gemcitabine-based adjuvant chemotherapy improves survival after aggressive surgery for hilar cholangiocarcinoma. J Gastrointest Surg. 2009;13:1470–9.
Igami T, Nishio H, Ebata T, Yokoyama Y, Sugawara G, Nimura Y, et al. Surgical treatment of hilar cholangiocarcinoma in the “new era”: the Nagoya University experience. J Hepatobiliary Pancreat Sci. 2010;17:449–54.
Hirano S, Kondo S, Tanaka E, Shichinohe T, Tsuchikawa T, Kato K, et al. Outcome of surgical treatment of hilar cholangiocarcinoma: a special reference to postoperative morbidity and mortality. J Hepatobiliary Pancreat Sci. 2010;17:455–62.
Lee SG, Song GW, Hwang S, Ha TY, Moon DB, Jung DH, et al. Surgical treatment of hilar cholangiocarcinoma in the new era: the Asian experience. J Hepatobiliary Pancreat Sci. 2010;17:476–89.
Unno M, Katayose Y, Rikiyama T, Yoshida H, Yamamoto K, Morikawa T, et al. Major hepatectomy for perihilar cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2010;17:463–9.
Sakamoto Y, Kosuge T, Shimada K, Sano T, Ojima H, Yamamoto J, et al. Prognostic factors of surgical resection in middle and distal bile duct cancer: an analysis of 55 patients concerning the significance of ductal and radial margins. Surgery. 2005;37:396–402.
Murakami Y, Uemura K, Hayashidani Y, Sudo T, Hashimoto Y, Ohge H, et al. Prognostic significance of lymph node metastasis and surgical margin status for distal cholangiocarcinoma. J Surg Oncol. 2007;95:207–12.
Ebata T, Nagino M, Nishio H, Igami T, Yokoyama Y, Nimura Y. Pancreatic and duodenal invasion in distal bile duct cancer: paradox in the tumor classification of the American Joint Committee on Cancer. World J Surg. 2007;31:2008–15.
Woo SM, Ryu JK, Lee SH, Yoo JW, Park JK, Kim YT, et al. Recurrence and prognostic factors of ampullary carcinoma after radical resection: comparison with distal extrahepatic cholangiocarcinoma. Ann Surg Oncol. 2007;14:3195–201.
Hong SM, Pawlik TM, Cho H, Aggarwal B, Goggins M, Hruban RH, et al. Depth of tumor invasion better predicts prognosis than the current American Joint Committee on Cancer T classification for distal bile duct carcinoma. Surgery. 2009;146:250–7.
Murakami Y, Uemura K, Hayashidani Y, Sudo T, Ohge H, Sueda T. Pancreatoduodenectomy for distal cholangiocarcinoma: prognostic impact of lymph node metastasis. World J Surg. 2007;31:337–42.
Murakami Y, Uemura K, Sudo T, Hayashidani Y, Hashimoto Y, Nakamura H, et al. Adjuvant gemcitabine plus S-1 chemotherapy improves survival after aggressive surgical resection for advanced biliary carcinoma. Ann Surg. 2009;250:950–6.
Murakami Y, Uemura K, Hayasidani Y, Sudo T, Hashimoto Y, Ohge H, et al. Indication for postoperative adjuvant therapy in biliary carcinoma based on analysis of recurrence and survival after surgical resection. Dig Dis Sci. 2009;54:1360–4.
Sobin LH, Gospodarowicz MK, Wittekind C, eds. International Union Against Cancer (UICC): TNM classification of malignant tumors, 7th edn. New York: Wiley-Blackwell; 2010.
DeOliveira ML, Cunningham SC, Cameron JL, Kamangar F, Winter JM, Lillemoe KD, et al. Cholangiocarcinoma: thirty-one-year experience with 564 patients at a single institution. Ann Surg. 2007;245:755–62.
Guglielmi A, Ruzzenente A, Campagnaro T, Pachera S, Valdegamberi A, Capelli P, et al. Does intrahepatic cholangiocarcinoma have better prognosis compared to perihilar cholangiocarcinoma? J Surg Oncol. 2010;101:111–5.
Nakagohri T, Asano T, Kinoshita H, Kenmochi T, Urashima T, Miura F, et al. Aggressive surgical resection for hilar-invasive and peripheral intrahepatic cholangiocarcinoma. World J Surg. 2003;27:289–93.
Gerhards MF, van Gulik TM, González González D, Rauws EA, Gouma DJ. Results of postoperative radiotherapy for resectable hilar cholangiocarcinoma. World J Surg. 2003;27:173–9.
Hughes MA, Frassica DA, Yeo CJ, Riall TS, Lillemoe KD, Cameron JL,et al. Adjuvant concurrent chemoradiation for adenocarcinoma of the distal common bile duct. Int J Radiat Oncol Biol Phys. 2007;68:178–82.
Serafini FM, Sachs D, Bloomston M, Carey LC, Karl RC, Murr MM, et al. Location, not staging, of cholangiocarcinoma determines the role for adjuvant chemoradiation therapy. Am Surg. 2001;67:839–44.
Sagawa N, Kondo S, Morikawa T, Okushiba S, Katoh H. Effectiveness of radiation therapy after surgery for hilar cholangiocarcinoma. Surg Today. 2005;35:548–52.
Takada T, Amano H, Yasuda H, Nimura Y, Matsushiro T, Kato H, et al. Study Group of Surgical Adjuvant Therapy for Carcinomas of the Pancreas and Biliary Tract. Is postoperative adjuvant chemotherapy useful for gallbladder carcinoma? A phase III multicenter prospective randomized controlled trial in patients with resected pancreaticobiliary carcinoma. Cancer. 2002;95:1685–95.
Penz M, Kornek GV, Raderer M, Ulrich-Pur H, Fiebiger W, Lenauer A, et al. Phase II trial of two-weekly gemcitabine in patients with advanced biliary tract cancer. Ann Oncol. 2001;12:183–6.
André T, Tournigand C, Rosmorduc O, Provent S, Maindrault-Goebel F, Avenin D, et al. GERCOR Group. Gemcitabine combined with oxaliplatin (GEMOX) in advanced biliary tract adenocarcinoma: a GERCOR study. Ann Oncol. 2004;15:1339–43.
Patt YZ, Hassan MM, Aguayo A, Nooka AK, Lozano RD, Curley SA, et al. Oral capecitabine for the treatment of hepatocellular carcinoma, cholangiocarcinoma, and gallbladder carcinoma. Cancer. 2004;101:578–86.
Ueno H, Okusaka T, Ikeda M, Takezako Y, Morizane C. Phase II study of S-1 in patients with advanced biliary tract cancer. Br J Cancer. 2004;91:1769–74.
Sasaki T, Isayama H, Nakai Y, Ito Y, Kogure H, Togawa O, et al. Multicenter, phase II study of gemcitabine and S-1 combination chemotherapy in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol. 2010;65:1101–7.
Todoroki T, Ohara K, Kawamoto T, Koike N, Yoshida S, Kashiwagi H, et al. Benefits of adjuvant radiotherapy after radical resection of locally advanced main hepatic duct carcinoma. Int J Radiat Oncol Biol Phys. 2000;46:581–7.
Cheng Q, Luo X, Zhang B, Jiang X, Yi B, Wu M. Predictive factors for prognosis of hilar cholangiocarcinoma: postresection radiotherapy improves survival. Eur J Surg. Oncol. 2007;33:202–7.
Chen XP, Lau WY, Huang ZY, Zhang ZW, Chen YF, Zhang WG, et al. Extent of liver resection for hilar cholangiocarcinoma. Br J Surg. 2009;96:1167–75.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murakami, Y., Uemura, K., Sudo, T. et al. Prognostic Factors After Surgical Resection for Intrahepatic, Hilar, and Distal Cholangiocarcinoma. Ann Surg Oncol 18, 651–658 (2011). https://doi.org/10.1245/s10434-010-1325-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1325-4