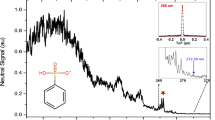
Abstract
The photoionization and dissociative photoionization of o-xylene have been studied using synchrotron radiation vacuum ultraviolet light with the photon energy in the range of 8.0–13.0 eV. The ionization energy of o-xylene and the appearance energies for the main fragment ions \(\hbox {C}_{\mathrm {8}}\hbox {H}_{\mathrm {9}}^{+}\), \(\hbox {C}_{\mathrm {7}}\hbox {H}_{\mathrm {7}}^{+}\) and \(\hbox {C}_{\mathrm {6}}\hbox {H}_{\mathrm {6}}^{+}\) were determined to be 8.45, 11.08, 11.47 and 11.17 eV, respectively, with photoionization efficiency spectra (PIES). Three dissociation pathways of parent ions are investigated by experimental photoionization and the quantum-chemical calculations. The fragment ions \(\hbox {C}_{\mathrm {7}}\hbox {H}_{\mathrm {7}}^{\mathrm {+}}\) derived from parent ions involved hydrogen atom transfer rather than direct dissociation. Particularly, detailed mechanisms of dissociation \(\hbox {C}_{\mathrm {6}}\hbox {H}_{\mathrm {6}}^{+}\) are presented for the first time in this work. In addition, the geometries of the intermediates (INTs), transition states (TSs) and products have been performed at B3LYP/6-311 \(+ \,+\) G (d, p) level. The mechanisms of dissociative photoionization of o-xylene and the INTs and TSs involved are discussed in detail.
Graphic Abstract









Similar content being viewed by others
Data Availability Statement
This manuscript has no associated data, or the data will not be deposited. [Author’s comment: All data generated during this study are contained in this published article.]
References
J. Chen, X. Chen, X. Chen, W. Xu, Z. Xu, H. Jia, J. Chen, Appl. Catal. B 224, 825 (2018)
M. Piumetti, D. Fino, N. Russo, Appl. Catal. B 163, 277 (2015)
C.W. Yu, J.T. Kim, Indoor Built Environ. 19, 30 (2010)
C. Song, N.A. Kwangsam, B. Warren, Q. Malloy, D.R. Cocker, Environ. Sci. Technol. 41, 7403 (2007)
K. Na, K.-C. Moon, Y.P. Kim, Atmos. Environ. 39, 5517 (2005)
A. Kansal, J. Hazard. Mater. 166, 17–26 (2009)
P. Zhang, J. Huang, J. Shu, B. Yang, Environ. Pollut. 245, 20 (2019)
F. Gaie-Levrel, C. Gutlé, H.W. Jochims, E. Rühl, M. Schwell, J. Phys. Chem. A 112, 5138 (2008)
C. Song, N.A. Kwangsam, B. Warren, B. Warren, Q. Malloy, D.R. Cocker, Environ. Sci. Technol. 41, 7403 (2007)
J.P. Maier, D.W. Turner, J. Chem. Soc. Faraday Trans. 54, 196 (1973)
F. Brogli, E. Giovannini, E. Heilbronner, R. Schurter, Eur. J. Inorg. Chem. 106, 961 (2010)
S.G. Lias, P. Ausloos, J. Am. Chem. Soc. 100, 6027 (1978)
J.O. Howell, J.M. Goncalves, C. Amatore, L. Klasinc, J. Am. Chem. Soc. 106, 3968 (1984)
A.G. Loudon, R.Z. Mazengo, J. Mass Spectr. 8, 179 (1974)
P. Nounou, J. Chim. Phys. 63, 994 (1966)
M.E. Akopyan, F.I. Vilesov, Khim. Vysokikh Energ. 2, 107 (1968)
C.Q. Jiao, S.F. Adams, Chem. Phys. Lett. 573, 24 (2013)
L. Zhao, Z.J. Cheng, L.L. Ye, F. Zhang, L.D. Zhang, F. Qi, Y.Y. Li, P. Combust. Inst. 35, 1745 (2015)
S.S. Wang, R.H. Kong, X.B. Shan, Y.W. Zhang, L.S. Sheng, Z.Y. Wang, L.Q. Hao, S.K. Zhou, J. Synchrotron. Radiat. 13, 415 (2006)
Y.J. Zhao, Y. Sun, J.D. Sun, W.Z. Fang, X.B. Shan, F.Y. Liu, L.S. Sheng, Z.Y. Wang, J. Electron. Spectrosc. Relat. Phenom. 173, 24 (2009)
Y.J. Zhao, X.B. Shan, L.S. Sheng, Z.Y. Wang, J. Zhang, C.R. Yu, Chin. Phys. B 20, 043201–1 (2011)
Y.J. Zhao, Y. Sun, R.H. Kong, Q. Du, W.Z. Fang, J.D. Sun, X.B. Shan, F.Y. Liu, L.S. Sheng, Z.Y. Wang, Nucl. Tech. 32, 561 (2009). (in Chinese)
A.D. Becke, J. Chem. Phys. 98, 5648 (1993)
C. Lee, W.T. Yang, G.P. Robert, Phys. Rev. B 37, 785 (1988)
C. Kosmidis, K.W.D. Ledingham, H.S. Kilic, T. McCanny, R.P. Singhal, A.J. Langley, W. Shaikh, J. Phys. Chem. A 101, 2264 (1997)
Y.J. Zhao, Y.S. Zhan, L. Li, X. Li, X.Y. Lian, P. Huang, L.S. Sheng, J. Chen, Chin. J. Chem. Phys. 030, 303 (2017)
S.J. Klippenstein, L.B. Harding, Y. Georgievskii, P. Combust. Inst. 31, 221 (2007)
M.E. Akopyan, F.I. Vilesov, Russ. J. Phys. Chem. 40, 63 (1966)
T.M. Ezzat, M.A. Rabbih, M.A. Fahmey, M.F. Hawash, Int. J. Mass. Spectr. 113, 133 (1992)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.11275006, No.11805032, No.11505027, No.11575178, No.U1532137), Nuclear Technology Application Engineering Research Center Open Foundation of Ministry of Education (No.HJSJYB2015-6, No.HJSJYB2018-6), the Chinese Scholarship Council (No.201608360053), the Graduate Students High-Quality Course Construction Program of Jiangxi Province (No.JXYYK2016-12), the China Postdoctoral Science Foundation(No.2013M531530), the Doctoral Foundation of East China University of Technology (No.DHBK201401) and the Provincial Natural Science Research Program of Higher Education Institutions of Anhui Province (No.KJ2012B086), Natural Science Foundation of Education Department of Guizhou Province ([2016]110) and Natural Science Foundation of Department of Science and Technology of Guizhou Province ([2020]QNSYXM03).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Y., Huang, P., Li, L. et al. VUV photoionization and dissociative photoionization of o-xylene: experimental and theoretical insights. Eur. Phys. J. D 75, 128 (2021). https://doi.org/10.1140/epjd/s10053-021-00135-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjd/s10053-021-00135-2