Abstract
The stability and other properties of the dipositronium molecule are discussed. It is pointed out that the dipositronium binding energy may be explained on the basis of a simplified model which satisfies the important requirement of providing a consistent description of the “atoms” and “molecule” of positronium. The importance of correlations in positronium and dipositronium is analyzed. In comparing positronium and dipositronium with atomic and molecular hydrogen, a remarkable parallelism is identified but also significant differences.
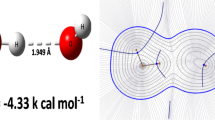
Graphical abstract

Similar content being viewed by others
References
J.A. Wheeler, Ann. N.Y. Acad Sci. 48, 219 (1946)
E.A. Hylleraas, A. Ore, Phys. Rev. 71, 493 (1947)
D.B. Cassidy, A.P. Mills Jr., Nature 449, 195 (2007)
D.B. Kinghorn, R.D. Poshusta, Phys. Rev. A 47, 3671 (1993)
L.J. Dunne, J.N. Murrell, Int. J. Quantum Chem. 96, 512 (2004)
A.J.C. Varandas, M. Brajczewska, J. da Providência, J.P. da Providência, Chem. Phys. Lett. 610-611, 167 (2014)
R.E. Peierls, J. Phys. A 24, 5273 (1991)
K.P. Huber, G. Herzberg, Molecular Spectra and Molecular Structure. IV. Constants of Diatomic Molecules (Van Nostrand Reinhold Company, New York, 1979), p. 716
K. Varga, J. Usukura, Y. Suzuki, Phys. Rev. Lett. 80, 1876 (1998)
D.C. Walker, J. Phys. Chem. 85, 3960 (1981)
J.D. Jackson, Phys. Rev. A 49, 132 (1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varandas, A., da Providência, J., Brajczewska, M. et al. On dipositronium and molecular hydrogen: similarities and differences. Eur. Phys. J. D 69, 114 (2015). https://doi.org/10.1140/epjd/e2015-50818-0
Received:
Revised:
Published:
DOI: https://doi.org/10.1140/epjd/e2015-50818-0