Abstract.
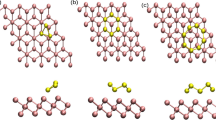
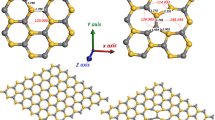
We showed in a recent density functional study that small palladium cluster on a MgO surface with F-centers can be oxidized to epitaxial PdxOy nano-oxides below room temperature [1]. Here, we employ density functional theory in order to explore different methods for an experimental verification of the PdxOy formation. The electronic density of states (DOS) of bare, O2-decorated and of oxidized palladium cluster was calculated. For many cluster sizes a clear difference in the DOS could be observed allowing for a detection of the oxidation with surface sensitive spectroscopic methods. In addition, adsorption sites and stretch frequencies of a single CO molecule on bare and oxidized Pd4 clusters were calculated. While CO prefers hollow sites on Pd4, top adsorption sites are found for Pd4O2. Markedly different CO stretch frequencies indicate a possible discrimination of bare clusters and oxides by Fourier transform infrared spectroscopy.
Similar content being viewed by others
References
B. Huber, P. Koskinen, H. Häkkinen, M. Moseler, Nature Materials 5, 44 (2006)
R.M. Lambert, G. Pacchioni, in Chemisorption and Reactivity on Supported Clusters and Thin Films (Kluwer, Dordrecht, 1997), pp. 395–424
H. Grönbeck, Top. Catalys. 28, 59 (2004)
U. Heiz, W.D. Schneider, in Metal Clusters at Surfaces (Springer, Berlin, 2000), pp. 237–273
C. Becker, C.R. Henry, Surf. Sci. 352, 457 (1996)
H.J. Freund, Adv. Catal. 45, 333 (2000)
S.H. Shaikhutdinov et al., Surf. Sci. 501, 270 (2002)
J. Mizei, J. Voutilainen, S. Saukko, V. Lantto, Thin Solid Films 391, 209 (2001)
M. Westphalen, U. Kreibig, J. Rostalski, H. Luth, D. Meissner, Sol. Ener. Mater. Sol. Cells. 61, 97 (2000)
S. Fukami, A. Ohno, A. Tanaka, Mater. Trans. 45, 2012 (2004)
U. Heiz, A. Sanchez, S. Abbet, W.D. Schneider, Chem. Phys. 262, 189 (2000)
S. Abbet, A. Sanchez, U. Heiz, W.D. Schneider, A.M. Ferrari, G. Pacchioni, N. Rösch, J. Am. Chem. Soc. 122, 3453 (2000)
S. Abbet, U. Heiz, H. Häkkinen, U. Landman, Phys. Rev. Lett. 86, 5950 (2001)
A. Eichler, F. Mittendorfer, J. Hafner, Phys. Rev. B 62, 4744 (2000)
B. Huber, H. Häkkinen, U. Landman, M. Moseler, Comp. Mat. Sci. 35, 371 (2006)
P. Mars, D.W. van Krevelen, Chem. Eng. Sci. 3, 41 (1954)
L.N. Kantorovich, A.L. Shluger, P.V. Sushko, J. Günster, P. Stracke, D.W. Goodman, V. Kempter, Faraday Discuss. 114, 173 (1999)
A.S. Wörz, K. Judai, S. Abbet, U. Heiz, J. Am. Chem. Soc. 125, 7964 (2003)
R.N. Barnett, U. Landman, Phys. Rev. B 48, 2081 (1993)
J.P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996)
N. Troullier, J.L. Martins, Phys. Rev. B 43, 1993 (1991)
The error caused by the finiteness of the Mg13O12 cluster was estimated by calculating the structure and binding energy of a Pd atom on the relevant O top positions of a defect-free Mg13O13 and Mg25O25 cluster. Neither the distance of the adsorption site from the centre nor the size of the substrate had a remarkable influence on the binding position of the Pd atom. Furthermore, the variation of binding energies was less than 5% indicating that finite size errors can be safely neglected
S. Abbet, E. Riedo, H. Brune, U. Heiz, A.M. Ferrari, L. Giordano, G. Pacchioni, J. Am. Chem. Soc. 123, 6172 (2001)
M. Moseler, B. Huber, H. Häkkinen, U. Landman, G. Wrigge, M.A. Hoffmann, B. von Issendorff, Phys. Rev. B 68, 165413 (2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huber, B., Moseler, M. Predicting experimental signatures for the oxidation of magnesia supported palladium clusters by density functional theory. Eur. Phys. J. D 45, 485–489 (2007). https://doi.org/10.1140/epjd/e2007-00178-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1140/epjd/e2007-00178-5