Abstract
Objective
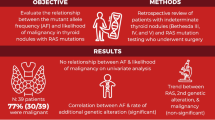
To investigate the frequency and ultrasonography (US) findings of RAS mutations and PAX8/PPARγ rearrangements between follicular thyroid adenomas (FTAs) and follicular thyroid carcinomas (FTCs) in a Korean population.
Methods
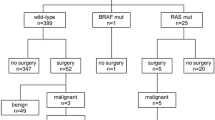
RAS mutations and PAX8/PPARγ rearrangements in 56 FTAs and 35 FTCs were analyzed. We also analyzed the US findings of FTCs and FTAs.
Results
16 nodules of 35 FTCs (45.7 %) and 19 nodules of 56 FTAs (33.9 %) harbored RAS mutations. Three FTCs and three FTAs showed two point mutations simultaneously. K-RAS codon 12–13 (n = 6, 31.6 %), N-RAS codon 61 (n = 5, 26.3 %), H-RAS codon 61 (n = 4, 21.1 %), K-RAS codon 61 (n = 3, 15.8 %), and N-RAS codon 12–13 (n = 1, 5.3 %) were found in FTCs, and N-RAS codon 61 (n = 10, 45 %), K-RAS codon 12–13 (n = 5, 22.7 %), H-RAS codon 61 (n = 5, 22.7 %), K-RAS codon 61 (n = 1, 4.5 %), and N-RAS codon 12–13 (n = 1, 4.5 %) were observed in FTAs. 4 of 56 (7.1 %) FTAs and 1 of 35 (2.9 %) FTCs represented PAX8/PPARγ rearrangements, respectively (P = 0.645). The absence of a hypoechoic rim (P = 0.021) and presence of calcifications (P = 0.049) were significantly associated with FTCs compared with FTAs.
Conclusions
RAS mutation frequency targeting the Korean population showed a 45.7 % in FTCs and 35.7 % in FTAs, and PAX8/PPARγ rearrangements were more frequently showed in FTAs. K-RAS codon 12–13 was the most common RAS mutation in FTCs, whereas N-RAS codon 61 was more frequent in FTAs. The presence of calcifications and absence of a hypoechoic rim showed more frequently in FTCs.





Similar content being viewed by others
References
Grebe SK, Hay ID (1995) Follicular thyroid cancer. Endocrinol Metab Clin North Am 24(4):761–801
DeLellis RA (2006) Pathology and genetics of thyroid carcinoma. J Surg Oncol 94(8):662–669
D’Avanzo A, Treseler P, Ituarte PH et al (2004) Follicular thyroid carcinoma: histology and prognosis. Cancer 100(6):1123–1129
Nikiforova MN, Lynch RA, Biddinger PW et al (2003) RAS point mutations and PAX8-PPAR gamma rearrangement in thyroid tumors: evidence for distinct molecular pathways in thyroid follicular carcinoma. J Clin Endocrinol Metab 88(5):2318–2326
Lin JD, Chao TC (2006) Follicular thyroid carcinoma: from diagnosis to treatment. Endocr J 53(4):441–448
Nikiforov YE (2008) Thyroid carcinoma: molecular pathways and therapeutic targets. Mod Pathol 21(2):S37–S43
Banito A, Pinto AE, Espadinha C, Marques AR, Leite V (2007) Aneuploidy and RAS mutations are mutually exclusive events in the development of well-differentiated thyroid follicular tumours. Clin Endocrinol (Oxf) 67(5):706–711
Namba H, Rubin SA, Fagin JA (1990) Point mutations of ras oncogenes are an early event in thyroid tumorigenesis. Mol Endocrinol 4(10):1474–1479
Suarez HG, du Villard JA, Severino M et al (1990) Presence of mutations in all three ras genes in human thyroid tumors. Oncogene 5(4):565–570
Esapa CT, Johnson SJ, Kendall-Taylor P, Lennard TW, Harris PE (1999) Prevalence of ras mutations in thyroid neoplasia. Clin Endocrinol (Oxf) 50(4):529–535
Marques AR, Espadinha C, Catarino AL et al (2002) Expression of PAX8-PPAR gamma 1 rearrangements in both follicular thyroid carcinomas and adenomas. J Clin Endocrinol Metab 87(8):3947–3952
Marques AR, Espadinha C, Frias MJ et al (2004) Underexpression of peroxisome proliferator-activated receptor (PPAR)gamma in PAX8/PPARgamma-negative thyroid tumours. Br J Cancer 91(4):732–738
Mazzaferri EL (1993) Management of a solitary thyroid nodule. N Engl J Med 328(8):553–559
Fernández-Medarde A, Santos E (2011) Ras in cancer and developmental diseases. Genes Cancer 2(3):344–358
Forbes SA, Bindal N, Bamford S et al (2011) COSMIC: mining complete cancer genomes in the catalogue of somatic mutations in cancer. Nucleic Acids Res 39:D945–D950
Park KY, Koh JM, Kim YI et al (1998) Prevalences of Gs alpha, ras, p53 mutations and ret/PTC rearrangement in differentiated thyroid tumours in a Korean population. Clin Endocrinol (Oxf) 49(3):317–323
Kim HJ, Jang HW, Sohn SY et al (2012) Frequency of RAS mutations and PAX8/PPARgamma rearrangement in follicular thyroid tumors in Korea. Endocrinol Metab 27(1):45–53
Garcia-Rostan G, Zhao H, Camp RL et al (2003) Ras mutations are associated with aggressive tumor phenotypes and poor prognosis in thyroid cancer. J Clin Oncol 21(17):3226–3235
Fukahori M, Yoshida A, Hayashi H et al (2012) The associations between RAS mutations and clinical characteristics in follicular thyroid tumors: new insights from a single center and a large patient cohort. Thyroid 22(7):683–689
Vasko V, Ferrand M, Di Cristofaro J, Carayon P, Henry JF, de Micco C (2003) Specific pattern of RAS oncogene mutations in follicular thyroid tumors. J Clin Endocrinol Metab 88(6):2745–2752
Yoshimoto K, Iwahana H, Fukuda A et al (1992) Ras mutations in endocrine tumors: mutation detection by polymerase chain reaction-single strand conformation polymorphism. Jpn J Cancer Res 83(10):1057–1062
Liu RT, Hou CY, You HL et al (2004) Selective occurrence of ras mutations in benign and malignant thyroid follicular neoplasms in Taiwan. Thyroid 14(8):616–621
Manenti G, Pilotti S, Re FC, Della Porta G, Pierotti MA (1994) Selective activation of ras oncogenes in follicular and undifferentiated thyroid carcinomas. Eur J Cancer 30A(7):987–993
McIver B, Grebe SK, Eberhardt NL (2004) The PAX8/PPAR gamma fusion oncogene as a potential therapeutic target in follicular thyroid carcinoma. Curr Drug Targets Immune Endocr Metabol Disord 4(3):221–234
Acknowledgments
This work was supported by the Soonchunhyang University Research Fund. We are grateful to Ji Sung Lee, PhD (Director, Biostatistical Consulting Unit at Soonchunhyang University Medical Center) for statistical analysis.
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent of the present retrospective study was waived.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeong, S.H., Hong, H.S., Kwak, J.J. et al. Analysis of RAS mutation and PAX8/PPARγ rearrangements in follicular-derived thyroid neoplasms in a Korean population: frequency and ultrasound findings. J Endocrinol Invest 38, 849–857 (2015). https://doi.org/10.1007/s40618-015-0311-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0311-x