Abstract
Background
For the growing numbers of obese elderly with diabetes, the glucagon-like peptide-1 (GLP-1) receptor analogue (liraglutide) appears a safe way to promote and maintain substantial weight loss. Given this background, the aim of this study was to assess the effect of the liraglutide treatment, at doses up to 3.0 mg per day, on the body composition, focusing on sarcopenia, in overweight and obese elderly with type 2 diabetes mellitus (T2DM).
Methods
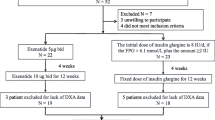
A perspective study was carried out in overweight and obese T2DM patients with HbA1c equal to 7.0 % (53 mmol/mol) ~10.0 % (86), under 3-month treatment (at least) of maximal dose of metformin at stable regime, and additional liraglutide at doses up to 3.0 mg per day. Body composition markers such as skeletal muscle index (SMI), android and gynoid fat mass, and arms and legs fat free mass, was measured by dual-energy X-ray densitometry (DXA) at baseline and after 24 weeks of liraglutide treatment. Glucose control was also carried out by glucose and HbA1c.
Results
Nine subjects (male/female 6/3, mean age 68.22 ± 3.86 years, BMI 32.34 ± 4.89 kg/m2) were evaluated. We noted a median decrease in BMI (−0.78 kg/m2), weight (−2000 g), fat mass (−1498 g) and android fat (−0.9 %), and a increase in SMI (+0.03 kg/m2) from baseline. Glycemic control also improved, with a median change HbA1c of −0.80 %.
Conclusions
Twenty-four weeks of liraglutide treatment was associated with reductions in fat mass and android fat. In addition, in order to prevent sarcopenia, it preserved the muscular tropism.

Similar content being viewed by others
References
Han TS, Wu FCW, Lean MEJ (2003) Obesity and weight management in the elderly: a focus on men. Best Pract Res Clin Endocrinol Metab 27:509–525
Drucker DJ, Philippe J, Mojsov S et al (1987) Glucagon-like peptide I stimulates insulin gene expression and increases cyclic AMP levels in a rat islet cell line. Proc Natl Acad Sci 84:3434–3438
Stoffers DA, Kieffer TJ, Hussain MA et al (2000) Insulinotropic glucagon-like peptide 1 agonists stimulate expression of homeodomain protein IDX-1 and increase islet size in mouse pancreas. Diabetes 49:741–748
Doyle ME, Egan JM (2007) Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol Ther 113:546–593
Buteau J (2008) GLP-1 receptor signaling: effects on pancreatic beta-cell proliferation and survival. Diab Metab 34:73–77
Werner U, Haschke G, Herling AW et al (2010) Pharmacological profile of lixisenatide: a new GLP-1 receptor agonist for the treatment of type 2 diabetes. Rev Regulat Peptides 164:58–64
Nikolaidis LA, Elahi D, Hentosz T et al (2004) Recombinant glucagonlike peptide-1 increases myocardial glucose uptake and improves left ventricular performance in conscious dogs with pacing-induced dilated cardiomyopathy. Circulation 110:955–961
Zhao T, Parikh P, Bhashyam S et al (2006) Direct effects of glucagonlike peptide-1 on myocardial contractility and glucose uptake in normal and postischemic isolated rat hearts. J Pharmacol Exp Ther 317:1106–1113
Basu A, Charkoudian N, Schrage W et al (2007) Beneficial effects of GLP-1 on endothelial function in humans: dampening by glyburide but not by glimepiride. Am J Physiol Endocrinol Metab 293:E1289–E1295
Nyström T (2008) The potential beneficial role of glucagon-like peptide-1 in endothelial dysfunction and heart failure associated with insulin resistance. Horm Metab Res 40:593–606
Chai W, Dong Z, Wang N et al (2012) Glucagon-like peptide. Recruits microvasculature and increases glu cose use in muscle via a nitric oxide-dependent mechanism. Diabetes 61:888–896
Luque MA, González N, Márquez L et al (2002) Glucagon-like peptide-1 (GLP-1) and glucose metabolism in human myocytes. J Endocrinol 173:465–473
Acitores A, Gonzalez N, Sancho V et al (2004) Cell signalling of glucagon-like peptide-1 action in rat skeletal muscle. J Endocrinol 180:389–398
Gonzalez N, Acitores A, Sancho V et al (2005) Effect of GLP-1 on glucose transport and its cell signalling in human myocytes. Regul Pept 126:203–211
Green CJ, Henriksen TI, Pedersen BK et al (2012) Glucagon-like peptide-1-induced glucose metabolism in differentiated human muscle satellite cells is attenuated by hyperglycemia. PloS One 7:e44284
Washburn RA, Smith KW, Jette AM et al (1993) The Physical Activity Scale for the Elderly (PASE): development and evaluation. J Clin Epidemiol 46:153–162
Janssen I, Baumgartner RN, Ross R et al (2004) Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am J Epidemiol 159:413–421
Baumgartner RN, Koehler KM, Gallagher D et al (1998) Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol 147:755–763
Lu N, Sun H, Yu J et al (2015) Glucagon-like peptide-1 receptor agonist Liraglutide has anabolic bone effects in ovariectomized rats without diabetes. PloSone 10:e0132744
Cetrone M, Mele A, Tricarico D (2104) Effects of the antidiabetic drugs on the age-related atrophy and sarcopenia associated with diabetes type II. Curr diabetes rev 10:231–237
Green CJ, Henriksen TI, Pedersen BK et al (2012) Glucagon like peptide-1-induced glucose metabolism in differentiated human muscle satellite cells is attenuated by hyperglycemia. PloS One 7:e44284
Yamamoto K, Amako M, Yamamoto Y et al (2013) therapeutic effect of exendin-4, a long-acting analogue of glucagon-like peptide-1 receptor agonist, on nerve regeneration after the crush nerve injury. Biomed Res Int 2013:315848
Jendle J, Nauck MA, Matthews DR et al (2009) Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes Metab 11:1163–1172
Inoue K, Maeda N, Kashine S et al (2011) Short-term effects of liraglutide on visceral fat adiposity, appetite, and food preference: a pilot study of obese Japanese patients with type 2 diabetes. Cardiovasc Diabetol 10:109
Nauck M, Frid A, Hermansen K et al (2009) Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (liraglutide effect and action in diabetes)-2 study. Diabetes Care 32:84–90
Li CJ, Yu Q, Yu P et al (2014) Changes in liraglutide-induced body composition are related to modifications in plasma cardiac natriuretic peptides levels in obese type 2 diabetic patients. Cardiovasc Diabetol 13:36
Wannamethee SG, Shaper AG, Lennon L et al (2007) Decreased muscle mass and increased central adiposity are independently related to mortality in older men. Am J Clin Nutr 86:1339–1346
Bode BW, Brett J, Falahati A et al (2011) Comparison of the efficacy and tolerability profile of liraglutide, a once-daily human GLP-1 analog, in patients with type 2 diabetes ≥65 and <65 years of age: a pooled analysis from phase III studies. Am J Geriatr Pharmacother 9:423–433
Peters KR (2013) Liraglutide for the treatment of type 2 diabetes: a clinical update. Am J Ther 20:178–188
Elashoff M, Matveyenko AV, Gier B et al (2011) Pancreatitis, pancreatic, and thyroid cancer with glucagon-like peptide-1-based therapies. Gastroenterology 141:150–156
Knezevich E, Crnic T, Kershaw S et al (2012) Liraglutide-associated acute pancreatitis. Am J Health Syst Pharm 69:386–389
Korkmaz H, Araz M, Alkan S et al (2015) Liraglutide-related cholelithiasis. Aging Clin Exp
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Ethical approval
The study was approved by the regional ethics committee at University of Pavia and was therefore performed in accordance with the ethical standards laid own in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
All subjects provided written consent to participate.
Rights and permissions
About this article
Cite this article
Perna, S., Guido, D., Bologna, C. et al. Liraglutide and obesity in elderly: efficacy in fat loss and safety in order to prevent sarcopenia. A perspective case series study. Aging Clin Exp Res 28, 1251–1257 (2016). https://doi.org/10.1007/s40520-015-0525-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-015-0525-y