Abstract
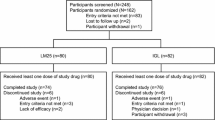
The aim of this study was to describe the efficacy and safety of two insulin intensification strategies in patients recruited in India with type 2 diabetes mellitus inadequately controlled on basal insulin glargine with metformin and/or pioglitazone. This multinational, open-label, randomized, parallel-arm, noninferiority, phase IV clinical trial evaluated insulin lispro low mixture (LM25) and basal insulin glargine administered with prandial insulin lispro (IGL) for 24 weeks. Patients were male and female, aged ≥18 to ≤75 years, with screening glycosylated hemoglobin (HbA1c) concentration ≥7.5 to ≤10.5 % and fasting plasma glucose ≤121 mg/dL. The primary efficacy end point was change in HbA1c from baseline to 24 weeks of treatment. Secondary efficacy end points included change in HbA1c from baseline to 12 weeks and change in fasting blood glucose (FBG) from baseline to 12 and 24 weeks. Safety and tolerability were measured by treatment-emergent adverse events and the incidence, rate, and severity of hypoglycemic episodes. Of 81 patients randomized to LM25 (n = 40) or IGL (n = 41), 80 patients completed the trial and one patient discontinued due to subject decision. Mean (SD) change in HbA1c from baseline to week 24 was −1.2 % (1.11) for the LM25 group and −1.0 % (1.18) for the IGL group. Safety profile, mean (FBG), glycemic variability, hypoglycemic episodes per patient-year, and health outcome measures were numerically similar between the two groups. The results of this post hoc analysis in an Indian subpopulation were consistent with results reported for the trial-level population and provide information to the consideration of LM25 as treatment option for intensification.


Similar content being viewed by others
References
Unger J. Insulin initiation and intensification in patients with T2DM for the primary care physician. Diabetes Metab Syndr Obes. 2011;4:253–61.
International Diabetes Federation. Diabetes surge hits every nation. 2013. http://www.idf.org/sites/default/files/Global_WDD_Final.pdf. Accessed 07 Apr 2014.
World Health Organization, International Diabetes Federation. Diabetes action now: an initiative of the World Health Organization and the International Diabetes Federation. 2004. http://www.who.int/diabetes/actionnow/en/DANbooklet.pdf. Accessed 23 Mar 2014.
Diabetes.co.uk. Diabetes in India. 2014. http://www.diabetes.co.uk/global-diabetes/diabetes-in-india.html. Accessed 15 Jul 2014.
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–53.
Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35:1364–79.
Owens DR. Stepwise intensification of insulin therapy in type 2 diabetes management—exploring the concept of the basal-plus approach in clinical practice. Diabet Med. 2013;30:276–88.
Mosenzon O, Raz I. Intensification of insulin therapy for type 2 diabetic patients in primary care: basal-bolus regimen versus premix insulin analogs: when and for whom? Diabetes Care. 2013;36 Suppl 2:S212–8.
Das AK, Sahay BK, Seshiah V, Mohan V, Muruganathan A, Kumar A, et al. Chapter 51: Indian National Consensus Group: National guidelines on initiation and intensification of insulin therapy with premixed insulin analogs. In: Muruganathan A, editor. Medicine update. The Association of Physicians of India; 2013. pp. 227–36.
Tinahones FJ, Gross JL, Onaca A, Cleall S, Rodríguez A. Insulin lispro low mixture twice daily versus basal insulin glargine once daily and prandial insulin lispro once daily in patients with type 2 diabetes requiring insulin intensification: a randomized phase IV trial. Diabetes Obes Metab. 2014. doi:10.1111/dom.12303.
World Health Organization. Definition, diagnosis and classification of diabetes mellitus and its complications, report of a WHO Consultation, part 1: diagnosis and classification of diabetes mellitus. Geneva: World Health Organization; 1999.
Vengurlekar S, Shukla P, Patidar P, Bafna R, Jain S. Prescribing pattern of antidiabetic drugs in Indore city hospital. Indian J Pharm Sci. 2008;70:637–40.
Kumar R, Kohli K, Kajal HL. A study of drug prescribing pattern and cost analysis among diabetic patients in a tertiary care teaching institute in north India. J Drug Deliv Ther. 2013;3:56–61.
Patel B, Oza B, Patel KP, Malhotra SD, Patel VJ. Pattern of antidiabetic drugs use in type-2 diabetic patients in a medicine outpatient clinic of a tertiary care teaching hospital. Int J Basic Clin Pharmacol. 2013;2:485–91.
Miser WF, Arakaki R, Jiang H, Scism-Bacon J, Anderson PW, Fahrbach JL. Randomized, open-label, parallel-group evaluations of basal-bolus therapy versus insulin lispro premixed therapy in patients with type 2 diabetes mellitus failing to achieve control with starter insulin treatment and continuing oral antihyperglycemic drugs: a noninferiority intensification substudy of the DURABLE trial. Clin Ther. 2010;32:896–908.
Rosenstock J, Ahmann AJ, Colon G, Scism-Bacon J, Jiang H, Martin S. Advancing insulin therapy in type 2 diabetes previously treated with glargine plus oral agents: prandial premixed (insulin lispro protamine suspension/lispro) versus basal/bolus (glargine/lispro) therapy. Diabetes Care. 2008;31:20–5.
Malone JK, Kerr LF, Campaigne BN, Sachson RA, Holcombe JH, Lispro Mixture-Glargine Study Group. Combined therapy with insulin lispro mix 75/25 plus metformin or insulin glargine plus metformin: a 16-week, randomized, open-label, crossover study in patients with type 2 diabetes beginning insulin therapy. Clin Ther. 2004;26:2034–44.
Malone JK, Bai S, Campaigne BN, Reviriego J, Augendre-Ferrante B. Twice-daily pre-mixed insulin rather than basal insulin therapy alone results in better overall glycaemic control in patients with type 2 diabetes. Diabet Med. 2005;22:374–81.
Acknowledgments
The study was funded by Eli Lilly and Company. The authors would like to thank Angel Rodríguez of Eli Lilly and Company for reviewing this manuscript and Amber Burns of Eli Lilly and Company and Shivanand Jigajinni of inVentiv Health Clinical for the medical writing support.
Conflict of interest
Simon Cleall and Shweta Uppal are employees of Eli Lilly and Company, and Steve Chen was an employee of Eli Lilly and Company during the writing of this manuscript. The authors report no conflicts of interest in this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prasanna Kumar, K.M., Shah, S., Shah, P. et al. Insulin lispro low mixture twice daily versus basal insulin glargine once daily and prandial insulin lispro once daily in patients with type 2 diabetes mellitus requiring insulin intensification—a randomized phase IV trial: Indian subpopulation analyses. Int J Diabetes Dev Ctries 37, 116–123 (2017). https://doi.org/10.1007/s13410-015-0387-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-015-0387-z