Abstract
Aims/introduction
We investigated the effects of three different statins in patients with type 2 diabetes mellitus and hypercholesterolemia.
Materials and methods
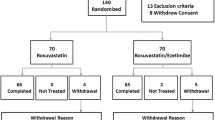
Patients with type 2 diabetes mellitus and low-density lipoprotein-cholesterol level of more than or equal to 100 mg/dL were treated at random with either atorvastatin (10 mg/day, n = 42), rosuvastatin (5 mg/day, n = 37) or pravastatin (10 mg/day, n = 38) for 3 months and changes in serum lipid profile and inflammatory markers were assessed.
Results
Low-density lipoprotein-cholesterol, small dense low-density lipoprotein and remnant-like particle cholesterol were significantly decreased from baseline in 3 groups. Atorvastatin and rosuvastatin significantly decreased high-sensitivity C-reactive protein. Rosuvastatin significantly decreased monocyte chemoattractant protein-1, while atorvastatin significantly increased estimated glomerular filtration rate (eGFR), but there were no significant intergroup differences in both parameters.
Conclusions
Atorvastatin and rosuvastatin significantly highly decreased low-density lipoprotein-cholesterol, remnant-like particle cholesterol, small dense low-density lipoprotein as well as high-sensitivity C-reactive protein than pravastatin and atorvastatin also had a potency in improve eGFR in patients with type 2 diabetes mellitus.


Similar content being viewed by others
References
Haffner SM, Lehto S, Rönnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339:229–34.
Grundy SM, Cleeman JI, Merz NB, et al. Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines. Circulation. 2004;110:227–39.
Graham I, Atar D, Borch-Johnsen K, et al. European guidelines on cardiovascular disease prevention in clinical practice: executive summary. Fourth Joint Task Force of the European Society of Cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts). Eur J Cardiovasc Prev Rehabil. 2007;14(Suppl 2):E1–40.
Teramoto T, Sasaki J, Ueshima H, et al. Diagnostic criteria for dyslipidemia. Executive summary of Japan Atherosclerosis Society (JAS) guideline for diagnosis and prevention of atherosclerotic cardiovascular diseases for Japanese. J Atheroscler Thromb. 2007;14:155–8.
Bahadir MA, Oguz A, Uzunlulu M, et al. Effects of different statin treatments on small dense low-density lipoprotein in patients with metabolic syndrome. J Atheroscler Thromb. 2009;16:684–90.
Otokozawa S, Ai M, Van Himbergen T, et al. Effects of intensive atorvastatin and rosuvastatin treatment on apolipoprotein B-48 and remnant lipoprotein cholesterol levels. Atherosclerosis. 2009;205:197–201.
Mason RP, Walter MF, Day CA, et al. Active metabolite of atorvastatin inhibits membrane cholesterol domain formation by an antioxidant mechanism. J Biol Chem. 2006;281:9337–45.
Thongtang N, Ai M, Otokozawa S, et al. Effects of maximal atorvastatin and rosuvastatin treatment on markers of glucose homeostasis and inflammation. Am J Cardiol. 2011;107:387–92.
Puccetti L, Santilli F, Pasqui AL, et al. Effects of atorvastatin and rosuvastatin on thromboxane-dependent platelet activation and oxidative stress in hypercholesterolemia. Atherosclerosis. 2011;214:122–8.
Shepherd J, Kastelein JJ, Bittner V, et al. Effect of intensive lipid lowering with atorvastatin on renal function in patients with coronary heart disease: the Treating to New Targets (TNT) study. Clin J Am Soc Nephrol. 2007;2:1131–9.
Zhang L, Zhang S, Jiang H, et al. Effect of statin treatment on cardiac function in patients with chronic heart failure: a meta-analysis of randomized controlled trials. Clin Cardiol. 2010;34:117–23.
Biffi A, Devan WJ, Anderson CD, et al. Statin treatment and functional outcome after ischemic stroke: case–control and meta-analysis. Stroke. 2011;42:1314–9.
Nissen SE, Nicholls SJ, Sipahi I, et al. Effect of very high-intensity statin therapy on regression of coronary atherosclerosis. The ASTEROID trial. JAMA. 2006;295:1556–65.
The Committee of Japan Diabetes Society on the diagnostic criteria of diabetes mellitus. Report of the Committee on the classification and diagnostic criteria of diabetes mellitus. J Diabetes Investig. 2010;1:212–228.
Lee E, Ryan S, Birmingham B, et al. Rosuvastatin pharmacokinetics and pharmacogenetics in white and Asian subjects residing in the same environment. Clin Pharmacol Ther. 2005;78:330–41.
Nicholls SJ, Brandrup-Wognsen G, Palmer M, et al. Meta-analysis of comparative efficacy of increasing dose of atorvastatin versus rosuvastatin versus Simvastatin on lowering levels of atherogenic lipids (from VOYAGER). Am J Cardiol. 2010;105:69–76.
Blasetto JW, Stein EA, Brown WV, et al. Efficacy of rosuvastatin compared with other statins at selected starting doses in hypercholesterolemic patients and in special population groups. Am J Cardiol. 2003;91(5A):3C–10C. discussion 10C.
Schneck DW, Knopp RH, Ballantyne CM, et al. Comparative effects of rosuvastatin and atorvastatin across their dose ranges in patients with hypercholesterolemia and without active arterial disease. Am J Cardiol. 2003;91:33–41.
Jones PH, Davidson MH, Stein EA, et al. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol. 2003;92:152–60.
Brunetti ND, Maulucci G, Casavecchia GP, et al. Improvement in endothelium dysfunction in diabetics treated with statins: a randomized comparison of atorvastatin 20 mg versus rosuvastatin 10 mg. J Interv Cardiol. 2007;20:481–7.
Yanagi K, Monden T, Ikeda S, et al. A crossover study of rosuvastatin and pitavastatin in patients with type 2 diabetes. Adv Ther. 2011;28:160–71.
Atorvastatin study group in Korea. Flexible initial dosing of atorvastatin based upon initial low-density lipoprotein cholesterol levels in type 2 diabetic patients. Korean J Intern Med. 2008;23:22–9.
Yamada S, Yanagawa T, Sasamoto K, et al. Atorvastatin lowers plasma low-density lipoprotein cholesterol and C-reactive protein in Japanese type 2 diabetic patients. Metabolism. 2006;55:67–71.
Takebayashi K, Matsumoto S, Wakabayashi S, et al. The effect of low-dose atorvastatin on circulating monocyte chemoattractant protein-1 in patients with type 2 diabetes complicated by hyperlipidemia. Metabolism. 2005;54:1225–9.
Forst T, Wilhelm B, Pfützner A, et al. Investigation of the vascular and pleiotropic effects of atorvastatin and pioglitazone in a population at high cardiovascular risk. Diab Vasc Dis Res. 2008;5:298–303.
Blanco-Colio LM, Martín-Ventura JL, de Teresa E, et al. Elevated ICAM-1 and MCP-1 plasma levels in subjects at high cardiovascular risk are diminished by atorvastatin treatment. Atorvastatin on Inflammatory Markers study: a substudy of Achieve Cholesterol Targets Fast with Atorvastatin Stratified Titration. Am Heart J. 2007;153:881–8.
Palaniswamy C, Selvaraj DR, Selvaraj T, et al. Mechanisms underlying pleiotropic effects of statins. Am J Ther. 2010;17:75–8.
Koh KK, Quon MJ, Han SH, et al. Atorvastatin causes insulin resistance and increases ambient glycemia in hypercholesterolemic patients. J Am Coll Cardiol. 2010;55:1209–16.
Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359:2195–207.
Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet. 2010;375:735–42.
Cachofeiro V, Goicochea M, de Vinuesa SG, et al. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int Suppl. 2008;111:S4–9.
Colhoun HM, Betteridge DJ, Durrington PN, et al. Effects of atorvastatin on kidney outcomes and cardiovascular disease in patients with diabetes: an analysis from the Collaborative Atorvastatin Diabetes Study (CARDS). Am J Kidney Dis. 2009;54:810–9.
Sawara Y, Takei T, Uchida K, et al. Effects of lipid-lowering therapy with rosuvastatin on atherosclerotic burden in patients with chronic kidney disease. Intern Med. 2008;47:1505–10.
Sorof J, Berne C, Siewert-Delle A, et al. Effect of rosuvastatin or atorvastatin on urinary albumin excretion and renal function in type 2 diabetic patients. Diabetes Res Clin Pract. 2006;72:81–7.
Acknowledgments
The authors thank Ms. N. Sakaguchi for the excellent technical assistance. This study is supported in part by a Research Grant-In-Aid for Scientific Research by the Ministry of Health, Labor and Welfare of Japan, the Ministry of Education, Culture, Sports, Science and Technology of Japan, and the University of Occupational and Environmental Health, Japan.
Conflict of interest
Dr. Tanaka has received consulting fees, speaking fees, and/or honoraria from Mitsubishi-Tanabe Pharma, Chugai Pharma, Eisai Pharma, Pfizer, Abbott Immunology Pharma, Daiichi-Sankyo, Janssen Pharma, Astra-Zeneca, Takeda Industrial Pharma, Astellas Pharma, Asahi-kasei Pharma and GlaxoSmithKline and has received research grant support from Mitsubishi-Tanabe Pharma, Bristol-Myers Squibb, Takeda Industrial Pharma, MSD, Astellas Pharma, Eisai Pharma, Chugai Pharma.
Author information
Authors and Affiliations
Corresponding author
Additional information
This trial was registered with University Hospital Medical Information Network (UMIN) (no. UMIN000007264).
About this article
Cite this article
Mori, H., Okada, Y. & Tanaka, Y. Effects of pravastatin, atorvastatin, and rosuvastatin in patients with type 2 diabetes mellitus and hypercholesterolemia. Diabetol Int 4, 117–125 (2013). https://doi.org/10.1007/s13340-012-0103-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-012-0103-x