Abstract
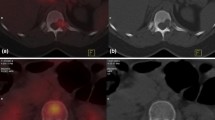
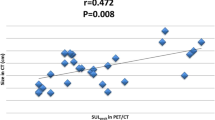
In India up to 50 % of breast cancer patients still present as locally advanced breast cancer (LABC). The conventional methods of metastatic work up include physical examination, bone scan, chest & abdominal imaging, and biochemical tests. It is likely that the conventional staging underestimates the extent of initial spread and there is a need for more sophisticated staging procedure. The PET/CT can detect extra-axillary and occult distant metastases and also aid in predicting response to chemotherapy at an early point in time. To evaluate the utility of FDG PET/CT in initial staging and response assessment of patients with LABC receiving NACT. A prospective study of all biopsy confirmed female patients diagnosed with LABC receiving NACT from April 2013 to May 2014. The conventional work up included serum chemistry, CECT chest and abdomen and bone scan. A baseline whole body PET/CT was done in all patients. A repeat staging evaluation and a whole body PET/CT was done after 2/3rd cycle of NACT in non-responders and after 3/4 cycles in clinical responders. The histopathology report of the operative specimen was used to document the pathological response. The FDG PET/CT reported distant metastases in 11 of 38 patients, where as conventional imaging revealed metastases in only 6. Almost all the distant lesions detected by conventional imaging were detected with PET/CT, which showed additional sites of metastasis in 3 patients. In 2 patients, PET/CT detected osteolytic bone metastasis which were not detected by bone scan. In 5 patients PET CT detected N3 disease which were missed on conventional imaging. A total of 14 patients had second PET/CT done to assess the response to NACT and 11 patients underwent surgery. Two patients had complete pathological response. Of these 1 patient had complete metabolic and morphologic response and other had complete metabolic and partial morphologic response on second PET/CT scan. The 18 FDG PET/CT can detect more number of metastasis as well as additional sites of metastasis compared to conventional methods. The response assessment resulted in change of treatment regimen in 14 % of patients.





Similar content being viewed by others
References
Agarwal G, Ramakant P (2008) Breast cancer care in India: the current scenario and the challenges for the future. Breast Care 3:21–27
AJCC (American Joint Committee on Cancer) (2010) In: Edge SB, Byrd DR, Compton CC et al (eds) Cancer staging manual, 7th edn. Springer, New York, p 347
Tham YL, Kramer R, Osborne CK (2010) Evaluation of patients for metastasis prior to primary therapy. In: Harris JR, Lippman ME, Morrow M, Osborne CK (eds) Diseases of the breast, 4th edn. Lippincott Williams & Wilkins, Philadelphia, p 483
Mahner S, Schirrmacher S, Brenner W, Jenicke L, Habermann CR, Avril N et al (2008) Comparison between positron emission tomography using 2-[fluorine-18] fluoro-2-deoxy-D-glucose, conventional imaging and computed tomography for staging of breast cancer. Ann Oncol 19:1249–1254
Al-Husaini H, Amir E, Fitzgerald B, Wright F, Dent R, Fralick J et al (2008) Prevalence of overt metastases in locally advanced breast cancer. Clin Oncol (R Coll Radiol) 20(5):340–344
Schirrmeister H, Kühn T, Guhlmann A, Santjohanser C, Hörster T, Nüssle K et al (2001) Fluorine-18 2-deoxy-2-fluoro-D-glucose PET in the preoperative staging of breast cancer: comparison with the standard staging procedures. Eur J Nucl Med 28:351–358
Groheux D, Moretti JL, Baillet G, Espie M, Giacchetti S, Hindie E et al (2008) Effect of (18) F-FDG PET/CT imaging in patients with clinical stage II and III breast cancer. Int J Radiat Oncol Biol Phys 71:695–704
Fuster D, Duch J, Paredes P, Velasco M, Muñoz M, Santamaría G et al (2008) Preoperative staging of large primary breast cancer with [18F] fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedures. J Clin Oncol 26:4746–4751
Van der Hoeven JJ, Krak NC, Hoekstra OS, Comans EF, Boom RP, Van Geldere D et al (2004) 18F-2-fluoro-2-deoxy-d-glucose positron emission tomography in staging of locally advanced breast cancer. J Clin Oncol 22:1253–1259
Von Minckwitz G, Sinn HP, Raab G, Loibl S, Blohmer JU, Eidtmann H, German Breast Group et al (2008) Clinical response after two cycles compared to HER2, Ki-67, p53, and bcl-2 in independently predicting a pathological complete response after preoperative chemotherapy in patients with operable carcinoma of the breast. Breast Cancer Res 10:R30
De Boer RH, Saini A, Johnston SR, O’Brien ME, Ellis PA, Verrill MW et al (2000) Continuous infusional combination chemotherapy in inflammatory breast cancer: a phase II study. Breast 9:149–155
Auclerc G, Borel C, Khayat D, Soubrane C (1992) Weil M [Primary chemotherapy in the treatment of breast cancer]. Ann Chir Plast Esthet 37:663–669
Fisher ER, Wang J, Bryant J, Fisher B, Mamounas E, Wolmark N (2002) Pathobiology of preoperative chemotherapy: findings from the national surgical adjuvant breast and bowel (NSABP) protocol B-18. Cancer 95:681–695
Van der Hage JA, Van de Velde CJ, Julien JP, Tubiana-Hulin M, Vandervelden C, Duchateau L (2001) Preoperative chemotherapy in primary operable breast cancer: results from the European organization for research and treatment of cancer trial 10902. J Clin Oncol 19:4224–4237
Bonadonna G, Valagussa P, Brambilla C, Ferrari L, Moliterni A, Terenziani M et al (1998) Primary chemotherapy in operable breast cancer: eight-year experience at the Milan cancer institute. J Clin Oncol 16:93–100
Feldman LD, Hortobagyi GN, Buzdar AU, Ames FC, Blumenschein GR (1986) Pathological assessment of response to induction chemotherapy in breast cancer. Cancer Res 46:2578–2581
NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. V 3.2014. National Comprehensive Cancer Network. Available at http://bit.ly/jOuSUf. Accessed October 2, 2014.
Groheux D, Giacchetti S, Delord M, Hindie E, Vercellino L, Cuvier C et al (2013) 18F FDG PET/CT in patients with locally advanced or inflammatory breast cancer: comparison to conventional staging. J Nucl Med 54:5–11
Wolfgang A (2009) Weber. Use of PET for monitoring cancer therapy and for predicting outcome. J Nucl Med 46:963–995
Wahl RL, Zasadny K, Helvie M, Hutchins GD, Weber B, Cody R (1993) Metabolic monitoring of breast cancer chemotherapy using positron emission tomography-initial evaluation. J Clin Oncol 11:2101–2111
Schelling M, Avril N, Nährig J, Kuhn W, Römer W, Sattler D et al (2000) Positron emission tomography using [(18) F] Fluorodeoxyglucose for monitoring primary chemotherapy in breast cancer. J Clin Oncol 18:1689–1695
Smith IC, Welch AE, Hutcheon AW, Miller ID, Payne S, Chilcott F et al (2000) Positron emission tomography using [(18) F]-fluorodeoxy-D-glucose to predict the pathologic response of breast cancer to primary chemotherapy. J Clin Oncol 18:1676–1688
Rousseau C, Devillers A, Sagan C, Ferrer L, Bridji B, Campion L et al (2006) Monitoring of early response to neoadjuvant chemotherapy in stage II and III breast cancer by [18F] fluorodeoxyglucose positron emission tomography. J Clin Oncol 24:5366–5372
McDermott GM, Welch A, Staff RT, Gilbert FJ, Schweiger L, Semple SI et al (2007) Monitoring primary breast cancer throughout chemotherapy using FDG-PET. Breast Cancer Res Treat 102:75–84
Schwarz-Dose J, Untch M, Tiling R, Sassen S, Mahner S, Kahlert S et al (2009) Monitoring primary systemic therapy of large and locally advanced breast cancer by using sequential positron emission tomography imaging with [18F] fluorodeoxyglucose. J Clin Oncol 27:535–541
Dunnwald LK, Gralow JR, Ellis GK (2008) Tumor metabolism and blood flow changes by positron emission tomography: relation to survival in patients treated with neoadjuvant chemotherapy for locally advanced breast cancer. J Clin Oncol 26:4449–4457
Groheux D, Hindie E, Giacchetti S, Delord M, Hamy AS, Roquancourt AD et al (2012) Triple negative breast cancer: early assessment with 18F-FDG PET/CT during neoadjuvant chemotherapy identifies patients who are unlikely to achieve a pathologic complete response and are at high risk of early relapse. J Nucl Med 53:249–254
Skoura EV, Datseris IE (2007) PET imaging in breast cancer. Hosp Chronicles 2:12–18
Conflict of Interest
None of the Authors have any conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hulikal, N., Gajjala, S.R., Kalawat, T.C. et al. Utility of [18F] Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography (FDG PET/CT) in the Initial Staging and Response Assessment of Locally Advanced Breast Cancer Patients Receiving Neoadjuvant Chemotherapy. Indian J Surg Oncol 6, 330–336 (2015). https://doi.org/10.1007/s13193-015-0421-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-015-0421-0