Abstract
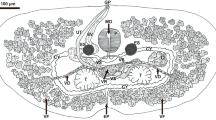
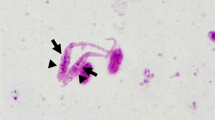
The nodular tapeworm, Raillietina echinobothrida is a well studied avian gastrointestinal parasite of family Davaineidae (Cestoda: Cyclophyllidea). It is reported to be the largest in size and second most prevalent species infecting chicken in north-east India. In the present study, morphometrical methods coupled with the molecular analysis of the second internal transcribed spacer (ITS2) region of ribosomal DNA were employed for precise identification of the parasite. The annotated ITS2 region was found to be 446 bp long and further utilized to elucidate the phylogenetic relationships and its species-interrelationships at the molecular level. In phylogenetic analysis similar topology was observed among the trees obtained by distance-based neighbor-joining as well as character-based maximum parsimony tree building methods. The query sequence R. echinobothrida is well aligned and placed within the Davaineidae group, with all Raillietina species well separated from the other cyclophyllidean (taeniid and hymenolepid) cestodes, while Diphyllobothrium latum (Pseudophyllidea: Diphyllobothriidae) was rooted as an out-group. Sequence similarities indeed confirmed our hypothesis that Raillietina spp. are neighboring the position with other studied species of order Cyclophyllidea against the out-group order Pseudophyllidea. The present study strengthens the potential of ITS2 as a reliable marker for phylogenetic reconstructions.


Similar content being viewed by others
References
Alvarez I, Wendel JF (2003) Ribosomal ITS sequences and plant phylogenetic inference. Mol Phylogenet Evol 29:417–434
Ashokan KV, Pillai MM (2008) In silico characterization of silk fibroin protein using computational tools and servers. Asian J Exp Sci 22:265–274
Beveridge I, Campbell RA, Palm HW (1999) Preliminary cladistic analysis of genera of the cestode order Trypanorhyncha Diesing, 1863. Syst Parasitol 42:29–49
Bowles J, Blair D, McManus DP (1995) A molecular phylogeny of the genus Echinococcus. Parasitology 110:317–328
Bray RA, Jones A, Hoberg EP (1999) Observations on the phylogeny of the cestode order Pseudophyllidea Carus, 1863. Syst Parasitol 42:13–20
Brooks DR, Hoberg EP (2001) Parasite systematics in the 21st century: opportunities and obstacles. Trends Parasitol 17:273–275
Coleman A (2003) ITS2 is a double-edged tool for eukaryotic evolutionary comparisons. Trends Genet 19:370–375
Engelmann JC, Rahmann S, Wolf M, Schultz J, Fritzilas E, Kneitz S, Dandekar T, Müller T (2009) Modeling cross-hybridization on phylogenetic DNA microarrays increases the detection power of closely related species. Mol Ecol Resour 9:83–93
Ghatani S, Shylla JA, Tandon V, Chatterjee A, Roy B (2012) Molecular characterization of pouched amphistome parasites (Trematoda: Gastrothylacidae) using ribosomal ITS2 sequence and secondary structures. J Helminthol 86:117–124
Gogoi AR, Hazarika RN (1975) Tetramorium tortosum (Rog), an intermediate host of poultry tapeworms Raillietina echinobothrida and Raillietina tetragona. Indian Vet J 52:939–941
Goswami LM, Prasad PK, Tandon V, Chatterjee A (2009) Molecular characterization of Gastrodiscoides hominis (Platyhelminthes: Trematoda: Digenea) inferred from ITS rDNA sequence analysis. Parasitol Res 104:1485–1490
Haukisalmi V, Wickstrom LM, Hantula J, Henttonen H (2001) Taxonomy, genetic differentiation and Holarctic biogeography of Paranoplocephala spp. (Cestoda: Anoplocephallidae) in collared lemmings (Dicrostonyx; Arvicolinae). Biol J Linn Soc 74:171–196
Hershkovitz M, Lewis L (1996) Deep-level diagnostic value of the rDNA-ITS region. Mol Biol Evol 13:1276–1295
Hoberg EP, Mariaux J, Justine JL, Brooks DR, Weekes PJ (1997) Phylogeny of the orders of the Eucestoda (Cercomeromorphae) based on comparative morphology: historical perspectives and a new working hypothesis. J Parasitol 83:1128–1147
Hoberg EP, Jones A, Bray RA (1999) Phylogenetic analysis among the families of the Cyclophyllidea (Eucestoda) based on comparative morphology, with new hypotheses for co-evolution in vertebrates. Syst Parasitol 42:51–73
Hoberg EP, Mariaux J, Brooks DR (2001) Phylogeny among orders of the Eucestoda (Cercomeromorphae): integrating morphology, molecules and total evidence. In: Littlewood DJT, Bray RA (eds) Interrelationships of the Platyhelminthes. Taylor & Francis, London, pp 112–126
Horsfall MW (1938) Observations on the Life History of Raillietina echinobothrida and of R. tetragona (Cestoda). J Parasitol 24:409–421
Huang A, Li JW, Shen ZQ, Wang XW, Jin M (2006) High-throughput identification of clinical pathogenic fungi by hybridization to oligonucleotide microarray. J Clin Microbiol 44:3299–3305
Jones MF, Horsfall MW (1935) Ants as intermediate hosts for two species of Raillietina parasitic in chickens. J Parasitol 21:442–443
Jyrwa DB, Tandon V, Lyngdoh RD (2009) Molecular characterization of Lytocestus spp. (Eucestoda: Caryophyllidea) from Clarias batrachus (L.) in Meghalaya. In: Tandon V, Yadav AK, Roy B (eds) Current trends in parasitology. Proceedings of the 20th national congress of parasitology, India, November 3–5, 2008. Panima Publishing Corporation, New Delhi, p 137–146
Khalil LF, Jones A, Bray RA (eds) (1994) Keys to the cestode parasites of vertebrates. Commonwealth Agriculture Bureaux International, Wallingford, pp 407–441
Literak F, Tenora V, Letkova M, Goldova N, Torres J, Olson PD (2006) Mesocestoides litteratus (Batsch, 1786) (Cestoda: Cyclophyllidea: Mesocestoidae) from the red fox: morphological and 18S rDNA characterization of European isolates. Helminthologia 43:191–195
Littlewood DTJ, Olson PD (2001) Small subunit rDNA and the phylum Platyhelminthes: signal, noise, conflict and compromise. In: Littlewood DTJ, Bray RA (eds) Interrelationships of the Platyhelminthes. Taylor & Francis, London, pp 262–278
Mariaux J (1996) Cestode systematics: any progress? Int J Parasitol 26:231–243
Mariaux J (1998) A molecular phylogeny of the Eucestoda. J Parasitol 84:114–124
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York, p 333
Park MH, Sim CJ, Baek J, Min GS (2007) Identification of genes suitable for DNA barcoding of morphologically indistinguishable Korean Halichondriidae sponges. Mol Cells 23:220–227
Prasad PK, Tandon V, Biswal DK, Goswami LM, Chatterjee A (2009a) Phylogenetic reconstruction using secondary structures and sequence motifs of ITS2 rDNA of Paragonimus westermani (Kerbert, 1878) Braun, 1899 (Digenea: Paragonimidae) and related species. BMC Genomics 10(Suppl 3):S25
Prasad PK, Tandon V, Biswal DK, Goswami LM, Chatterjee A (2009b) Use of sequence motifs as barcodes and secondary structures of internal transcribed spacer 2 (ITS2, rDNA) for identification of the Indian liver fluke, Fasciola (Trematoda: Fasciolidae). Bioinformation 3:314–320
Prasad PK, Goswami LM, Tandon V, Chatterjee A (2011) PCR-based molecular characterization and in silico analysis of food-borne trematode parasites Paragonimus westermani, Fasciolopsis buski and Fasciola gigantica from northeast India using ITS2 rDNA. Bioinformation 6:64–68
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sambrook J, Russel D (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Springs Harbour Press, New York
Sawada I (1965) On the genus Raillietina fuhrmann 1920 (II). J Nara Gakugei Univ (Nat) 13:5–38
Schmidt GD (1986) CRC Handbook of tapeworm identification. CRC Press, Boca Raton 675 pp
Sharma SK, Dkhar J, Kumaria S, Tandon P, Rao SR (2012) Assessment of phylogenetic inter-relationships in the genus Cymbidium (Orchidaceae) based on internal transcribed spacer region of rDNA. Gene 495:10–15
Soulsby EJL (1982) Helminths, Arthropods and Protozoa of domesticated animals, 7th edn. ELBS and Bailliere Tindall, London
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony method. Mol Biol Evol 28:2731–2739
Tandon V, Biswal DK, Prasad PK, Malsawmtluangi C (2010) Reconstructing the phylogenetic relationships of the cyclophyllidean cestodes: a case study using ITS2 rDNA and sequence structure alignment. In: Fred A, Filipe J, Gamboa H (eds) Biomedical engineering systems and technologies. BIOSTEC 2010, CCIS 127. Springer, Berlin, pp 309–321
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Waeschenbach A, Webster BL, Bray RA, Littlewood DTJ (2007) Added resolution among ordinal level relationships of tapeworms (Platyhelminthes: Cestoda) with complete small and large subunit nuclear ribosomal RNA genes. Mol Phylogenet Evol 45:311–325
Wiemers M, Keller A, Wolf M (2009) ITS2 secondary structure improves phylogeny estimation in a radiation of blue butterflies of the subgenus Agrodiaetus (Lepidoptera: Lycaenidae: Polyommatus). BMC Evol Biol 9:300
Woese CR, Fox GE (1977) Phylogenetic structure of the prokaryotic domain: the primary kingdoms. Proc Natl Acad Sci USA 74:5088–5090
Wolf M, Achtziger M, Schultz J, Dandekar T, Müller T (2005) Homology modeling revealed more than 20,000 rRNA internal transcribed spacer 2 (ITS2) secondary structures. RNA 11:1616–1623
Yadav AK, Tandon V (1991) Helminth parasitism of domestic fowl (Gallus domesticus L.) in a subtropical high-rainfall area of India. Beitr Trop Landwirtsch Veterinarmed 29:97–104
Yao H, Song J, Liu C, Luo K, Han J et al (2010) Use of ITS2 Region as the universal DNA barcode for plants and animals. Plos One 5:e13102
Acknowledgments
Authors thank the Department of Biotechnology (Government of India), New Delhi, for their financial support for the study. Thanks are also due to the Head, Department of Zoology, NEHU, for providing infrastructural facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramnath, Jyrwa, D.B., Dutta, A.K. et al. Molecular characterization of the Indian poultry nodular tapeworm, Raillietina echinobothrida (Cestoda: Cyclophyllidea: Davaineidae) based on rDNA internal transcribed spacer 2 region. J Parasit Dis 38, 22–26 (2014). https://doi.org/10.1007/s12639-012-0184-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-012-0184-2