Abstract
Introduction
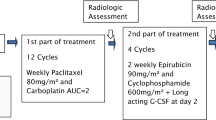
The objective was to evaluate a dose-dense schedule of docetaxel followed by doxorubicin and cyclophosphamide (AC) as neoadjuvant treatment for patients with locally advanced breast cancer.
Patients and methods
Ninety-nine patients were included and received 100 mg/m2 of docetaxel every two weeks for four cycles followed by 60 mg/m2 of doxorubicin and 600 mg/m2 of cyclophosphamide every two weeks for four cycles. Primary prophylaxis with granulocyte colony-stimulating factor (G-CSF) was administered systematically to all patients.
Results
Efficacy and toxicity analyses were carried out on an intention-to-treat basis. After treatment, complete pathological response in the breast and lymph nodes was confirmed in 15 patients (15%, 95% confidence interval [CI]: 8.4–22.9). Clinical response rate was 74% (95% CI: 65–82), of which 19% were complete responses. Breastconserving surgery could be performed in 41% of patients. The dose-dense schedule was generally well tolerated. The most important grade 3/4 toxicities per patient were cutaneous toxicity (12.1%) and hepatic dysfunction (9.1%) during docetaxel administration, and neutropenia (28.1%) and leucopenia (8.3%) with AC.
Conclusion
A dose-dense schedule of docetaxel followed by AC as neoadjuvant treatment is an effective and safe treatment for locally advanced breast cancer. Primary prophylaxis with G-CSF, and possibly the change in the sequence of drug administration, appears to play a major role in avoiding the excessive toxicity of dose-dense schedules.
Similar content being viewed by others
References
Kaufmann M, von Minckwitz G, Smith R et al (2003) International expert panel on the use of primary (preoperative) systemic treatment of operable breast cancer: review and recommendations. J Clin Oncol 21:2600–2608
Formenti SC, Volm M, Skinner KA et al (2003) Preoperative twice-weekly paclitaxel with concurrent radiation therapy followed by surgery and postoperative doxorubicin-based chemotherapy in locally advanced breast cancer: a phase I/II trial. J Clin Oncol 21:864–870
van der Hage JA, van de Velde CJ, Julien JP et al (2001) Preoperative chemotherapy in primary operable breast cancer: results from the European Organization for Research and Treatment of Cancer trial 10902. J Clin Oncol 19:4224–4237
Bonadonna G, Valagussa P, Brambilla C et al (1998) Primary chemotherapy in operable breast cancer: eight-year experience at the Milan Cancer Institute. J Clin Oncol 16:93–100
Belembaogo E, Feillel V, Chollet P et al (1992) Neoadjuvant chemotherapy in 126 operable breast cancers. Eur J Cancer 28A:896–900
Kuerer HM, Newman LA, Smith TL et al (1999) Clinical course of breast cancer patients with complete pathologic primary tumor and axillary lymph node response to doxorubicin-based neoadjuvant chemotherapy. J Clin Oncol 17:460–469
Scholl SM, Fourquet A, Asselain B et al (1994) Neoadjuvant versus adjuvant chemotherapy in premenopausal patients with tumours considered too large for breast conserving surgery: preliminary results of a randomised trial: S6. Eur J Cancer 30A:645–652
Makris A, Powles TJ, Ashley SE et al (1998) A reduction in the requirements for mastectomy in a randomized trial of neoadjuvant chemoendocrine therapy in primary breast cancer. Ann Oncol 9:1179–1184
Mamounas EP (1998) Overview of National Surgical Adjuvant Breast Project neoadjuvant chemotherapy studies. Semin Oncol 25:31–35
Fisher B, Brown A, Mamounas E et al (1997) Effect of preoperative chemotherapy on localregional disease in women with operable breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-18. J Clin Oncol 15: 2483–2493
Fisher B, Bryant J, Wolmark N et al (1998) Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol 16:2672–2685
Nabholtz JM, Gligorov J (2005) Docetaxel in the treatment of breast cancer: current experience and future prospects. Expert Rev Anticancer Ther 5: 613–633
Costa SD, von Minckwitz G, Raab G et al (1999) The role of docetaxel (Taxotere) in neoadjuvant chemotherapy of breast cancer. Semin Oncol 26: 24–31
Goble S, Bear HD (2003) Emerging role of taxanes in adjuvant and neoadjuvant therapy for breast cancer: the potential and the questions. Surg Clin North Am 83:943–971
Hutcheon AW, Heys SD, Sarkar TK (2003) Neoadjuvant docetaxel in locally advanced breast cancer. Breast Cancer Res Treat 79[Suppl 1]: S19–24
Bear HD, Anderson S, Brown A et al (2003) The effect on tumor response of adding sequential preoperative docetaxel to preoperative doxorubicin and cyclophosphamide: preliminary results from National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol 21:4165–4174
Bear HD, Anderson S, Smith RE et al (2006) Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer: National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol 24:2019–2027
von Minckwitz G, Raab G, Caputo A et al (2005) Doxorubicin with cyclophosphamide followed by docetaxel every 21 days compared with doxorubicin and docetaxel every 14 days as preoperative treatment in operable breast cancer: the GEPARDUO study of the German Breast Group. J Clin Oncol 23:2676–2685
Norton L (1997) Evolving concepts in the systemic drug therapy of breast cancer. Semin Oncol 24:S10-3–S10-10
Norton L (1999) Kinetic concepts in the systemic drug therapy of breast cancer. Semin Oncol 26: 11–20
Norton L (2001) Theoretical concepts and the emerging role of taxanes in adjuvant therapy. Oncologist 6[Suppl 3]:30–35
Gianni L, Mariani G, Mariani P (2004) Role of dose in the treatment of breast cancer. Ann Oncol 15[Suppl 4]:iv31–35
von Minckwitz G, Costa SD, Eiermann W et al (1999) Maximized reduction of primary breast tumor size using preoperative chemotherapy with doxorubicin and docetaxel. J Clin Oncol 17:1999–2005
(1999) Common Toxicity Criteria, Version 2.0. National Cancer Institute, Cancer Therapy Evaluation Program, Bethesda, MD, June 1
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Cristofanilli M, Buzdar AU, Sneige N et al (2001) Paclitaxel in the multimodality treatment for inflammatory breast carcinoma. Cancer 92:1775–1782
Smith IC, Heys SD, Hutcheon AW et al (2002) Neoadjuvant chemotherapy in breast cancer: significantly enhanced response with docetaxel. J Clin Oncol 20:1456–1466
Cooper BW, Radivoyevitch T, Overmoyer BA et al (2006) Phase II study of dose-dense sequential doxorubicin and docetaxel for patients with advanced operable and inoperable breast cancer. Breast Cancer Res Treat 97:311–318
Miller KD, McCaskill-Stevens W, Sisk J et al (1999) Combination versus sequential doxorubicin and docetaxel as primary chemotherapy for breast cancer: A randomized pilot trial of the Hoosier Oncology Group. J Clin Oncol 17:3033–3037
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antolín, S., Mel, R., Ramos, M. et al. A dose-dense schedule of docetaxel followed by doxorubicin and cyclophosphamide as neoadjuvant treatment for breast cancer: results from a phase II study. Clin Transl Oncol 13, 686–691 (2011). https://doi.org/10.1007/s12094-011-0715-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-011-0715-9