Abstract
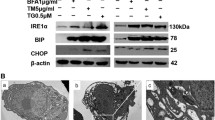
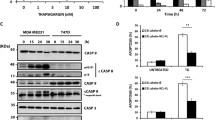
IRE1α endonuclease is a key regulator of endoplasmic reticulum (ER) stress that controls cell survival/apoptosis in cancers. Inhibition of IRE1α endonuclease leads to decreased splice XBP1 which decreases cell proliferation and increases cell death in cancer cells. Therefore, this study investigated the effects and mechanism of STF-083010 (an IRE1α inhibitor) on the cell growth/apoptosis of ovarian malignant cells via the XBP1-CHOP-Bim pathway following the induction of ER stress (ERS). ERS in OVCAR3 and SKOV3 cells was measured using Thioflavin T staining. The expression of ER stress response genes was evaluated by QRT-PCR. The levels of XBP1(s), PERK, phospho-PERK, p-PP2A, ATF4, BIP/GRP78, CHOP, and Bim proteins were evaluated using western blotting. Cell viability and apoptosis in STF-083010 and Tunicamycin (Tm) co-treated cells were assessed using BrdU, MTT, Annexin V-FITC/PI staining, and caspases-12 and -3 activity assays. The results showed increased XBP1, CHOP, and ATF-4 mRNA expression levels as well as high protein aggregation in STF-083010 and Tm co-treated cells. The IRE1α inhibitor down-regulated sXBP1 and BIP proteins, while XBP-1, p-PERK, ATF-4, CHOP, and Bim proteins were up-regulated. STF-083010 reduced cell proliferation and induced apoptosis through the activation of caspases-12 and -3 and Bax/Bcl-2 protein expression. In summary, the present data revealed the effects of STF-083010 in ER stress and apoptosis as well as signaling via XBP1/CHOP/Bim mediators. Thus, STF-083010 is proposed as a new target for the control of ERS in ovarian cancer cells.





Similar content being viewed by others
References
Atkins C, Liu Q, Minthorn E, Zhang S-Y, Figueroa DJ, Moss K et al (2013) Characterization of a novel PERK kinase inhibitor with antitumor and antiangiogenic activity. Cancer Res 73(6):1993–2002
Bagheri-Yarmand R, Sinha KM, Li L, Lu Y, Cote GJ, Sherman SI, Gagel RF (2019) Combinations of tyrosine kinase inhibitor and ERAD inhibitor promote oxidative stress-induced apoptosis through ATF4 and KLF9 in medullary thyroid Cancer. Mol Cancer Res 17(3):751–760. https://doi.org/10.1158/1541-7786.mcr-18-0354
Bakhshi J, Weinstein L, Poksay KS, Nishinaga B, Bredesen DE, Rao RV (2008) Coupling endoplasmic reticulum stress to the cell death program in mouse melanoma cells: effect of curcumin. Apoptosis 13(7):904–914. https://doi.org/10.1007/s10495-008-0221-x
Beriault DR, Werstuck GH (2013) Detection and quantification of endoplasmic reticulum stress in living cells using the fluorescent compound, Thioflavin T. Biochim Biophys Acta 1833(10):2293–2301. https://doi.org/10.1016/j.bbamcr.2013.05.020
Bian Z-M, Elner SG, Elner VM (2008) Regulated expression of caspase-12 gene in human retinal pigment epithelial cells suggests its immunomodulating role. Invest Ophthalmol Vis Sci 49(12):5593–5601. https://doi.org/10.1167/iovs.08-2116
Carrasco DR, Sukhdeo K, Protopopova M, Sinha R, Enos M, Carrasco DE et al (2007) The differentiation and stress response factor XBP-1 drives multiple myeloma pathogenesis. Cancer Cell 11(4):349–360. https://doi.org/10.1016/j.ccr.2007.02.015
Chen L, Li Q, She T, Li H, Yue Y, Gao S et al (2016) IRE1α-XBP1 signaling pathway, a potential therapeutic target in multiple myeloma. Leuk Res 49:7–12
Chen TH, Chiang YH, Hou JN, Cheng CC, Sofiyatun E, Chiu CH, Chen W. (2017) Xbp1-mediated Bip/GRP78 upregulation copes with oxidative stress in mosquito cells during dengue 2 virus infection. BioMed research international, 2017
Chen X, Iliopoulos D, Zhang Q, Tang Q, Greenblatt MB, Hatziapostolou M, Lim E, Tam WL, Ni M, Chen Y, Mai J, Shen H, Hu DZ, Adoro S, Hu B, Song M, Tan C, Landis MD, Ferrari M, Shin SJ, Brown M, Chang JC, Liu XS, Glimcher LH (2014) XBP1 promotes triple-negative breast cancer by controlling the HIF1alpha pathway. Nature 508(7494):103–107. https://doi.org/10.1038/nature13119
Chien W, Ding LW, Sun QY, Torres-Fernandez LA, Tan SZ, Xiao J et al (2014) Selective inhibition of unfolded protein response induces apoptosis in pancreatic cancer cells. Oncotarget 5(13):4881–4894. https://doi.org/10.18632/oncotarget.2051
Chiu TL, Su CC (2017) Tanshinone IIA increases protein expression levels of PERK, ATF6, IRE1alpha, CHOP, caspase3 and caspase12 in pancreatic cancer BxPC3 cellderived xenograft tumors. Mol Med Rep 15(5):3259–3263. https://doi.org/10.3892/mmr.2017.6359
Delie F, Petignat P, Cohen M (2012) GRP78 protein expression in ovarian cancer patients and perspectives for a drug-targeting approach. Journal of oncology, 2012
Dong D, Ni M, Li J, Xiong S, Ye W, Virrey JJ et al (2008) Critical role of the stress chaperone GRP78/BiP in tumor proliferation, survival, and tumor angiogenesis in transgene-induced mammary tumor development. Cancer Res 68(2):498–505
Gan PP, Zhou YY, Zhong MZ, Peng Y, Li L, Li JH (2017) Endoplasmic reticulum stress promotes autophagy and apoptosis and reduces chemotherapy resistance in mutant p53 lung Cancer cells. Cell Physiol Biochem 44(1):133–151. https://doi.org/10.1159/000484622
Guo F-J, Liu Y, Zhou J, Luo S, Zhao W, Li X, Liu C (2012) XBP1S protects cells from ER stress-induced apoptosis through Erk1/2 signaling pathway involving CHOP. Histochem Cell Biol 138(3):447–460
Han J, Back SH, Hur J, Lin YH, Gildersleeve R, Shan J, Yuan CL, Krokowski D, Wang S, Hatzoglou M, Kilberg MS, Sartor MA, Kaufman RJ (2013) ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death. Nat Cell Biol 15(5):481–490. https://doi.org/10.1038/ncb2738
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13(2):89–102. https://doi.org/10.1038/nrm3270
Hitomi J, Katayama T, Eguchi Y, Kudo T, Taniguchi M, Koyama Y, Manabe T, Yamagishi S, Bando Y, Imaizumi K, Tsujimoto Y, Tohyama M (2004) Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Abeta-induced cell death. J Cell Biol 165(3):347–356. https://doi.org/10.1083/jcb.200310015
Hübner A, Barrett T, Flavell RA, Davis RJ (2008) Multisite phosphorylation regulates Bim stability and apoptotic activity. Mol Cell 30(4):415–425. https://doi.org/10.1016/j.molcel.2008.03.025
Iurlaro R, Munoz-Pinedo C (2016) Cell death induced by endoplasmic reticulum stress. FEBS J 283(14):2640–2652. https://doi.org/10.1111/febs.13598
Jafari SM, Joshaghani HR, Panjehpour M, Aghaei M (2018) A2B adenosine receptor agonist induces cell cycle arrest and apoptosis in breast cancer stem cells via ERK1/2 phosphorylation. Cell Oncol 41(1):61–72
Jain BP (2017) An overview of unfolded protein response signaling and its role in cancer. Cancer Biother Radiopharm 32(8):275–281
Jin C, Jin Z, Chen NZ, Lu M, Liu CB, Hu WL, Zheng CG (2016) Activation of IRE1alpha-XBP1 pathway induces cell proliferation and invasion in colorectal carcinoma. Biochem Biophys Res Commun 470(1):75–81. https://doi.org/10.1016/j.bbrc.2015.12.119
Jung KJ, Min KJ, Bae JH, Kwon TK (2015) Carnosic acid sensitized TRAIL-mediated apoptosis through down-regulation of c-FLIP and Bcl-2 expression at the post translational levels and CHOP-dependent up-regulation of DR5, Bim, and PUMA expression in human carcinoma caki cells. Oncotarget 6(3):1556–1568. https://doi.org/10.18632/oncotarget.2727
Kalai M, Lamkanfi M, Denecker G, Boogmans M, Lippens S, Meeus A, Declercq W, Vandenabeele P (2003) Regulation of the expression and processing of caspase-12. J Cell Biol 162(3):457–467
Karagöz GE, Acosta-Alvear D, Nguyen HT, Lee CP, Chu F, Walter P (2017) An unfolded protein-induced conformational switch activates mammalian IRE1. Elife 6:e30700
Kim R, Emi M, Tanabe K, Murakami S (2006) Role of the unfolded protein response in cell death. Apoptosis 11(1):5–13. https://doi.org/10.1007/s10495-005-3088-0
Lee AH, Iwakoshi NN, Glimcher LH (2003) XBP-1 regulates a subset of endoplasmic reticulum resident chaperone genes in the unfolded protein response. Mol Cell Biol 23(21):7448–7459. https://doi.org/10.1128/mcb.23.21.7448-7459.2003
Lee AS (2005) The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35(4):373–381
Li X, Zhang K, Li Z (2011) Unfolded protein response in cancer: the Physician's perspective. J Hematol Oncol 4(1):8. https://doi.org/10.1186/1756-8722-4-8
Logue SE, McGrath EP, Cleary P, Greene S, Mnich K, Almanza A et al (2018) Inhibition of IRE1 RNase activity modulates the tumor cell secretome and enhances response to chemotherapy. Nat Commun 9(1):3267. https://doi.org/10.1038/s41467-018-05763-8
Luciano F, Jacquel A, Colosetti P, Herrant M, Cagnol S, Pages G, Auberger P (2003) Phosphorylation of Bim-EL by Erk1/2 on serine 69 promotes its degradation via the proteasome pathway and regulates its proapoptotic function. Oncogene 22(43):6785–6793. https://doi.org/10.1038/sj.onc.1206792
Luo S, Mao C, Lee B, Lee AS (2006) GRP78/BiP is required for cell proliferation and protecting the inner cell mass from apoptosis during early mouse embryonic development. Mol Cell Biol 26(15):5688–5697. https://doi.org/10.1128/MCB.00779-06
Luthra S, Fardin B, Dong J, Hertzog D, Kamjoo S, Gebremariam S, Butani V, Narayanan R, Mungcal JK, Kuppermann BD, Kenney MC (2006) Activation of Caspase-8 and Caspase-12 pathways by 7-Ketocholesterol in human retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 47(12):5569–5575. https://doi.org/10.1167/iovs.06-0333
Mandic A, Hansson J, Linder S, Shoshan MC (2003) Cisplatin induces endoplasmic reticulum stress and nucleus-independent apoptotic signaling. J Biol Chem 278(11):9100–9106. https://doi.org/10.1074/jbc.M210284200
Marciniak SJ, Yun CY, Oyadomari S, Novoa I, Zhang Y, Jungreis R et al (2004) CHOP induces death by promoting protein synthesis and oxidation in the stressed endoplasmic reticulum. Genes Dev 18(24):3066–3077. https://doi.org/10.1101/gad.1250704
Márquez S, Fernández JJ, Terán-Cabanillas E, Herrero C, Alonso S, Azogil A et al (2017) Endoplasmic reticulum stress sensor IRE1α enhances IL-23 expression by human dendritic cells. Front Immunol 8:639
Martinez JA, Zhang Z, Svetlov SI, Hayes RL, Wang KK, Larner SF (2010) Calpain and caspase processing of caspase-12 contribute to the ER stress-induced cell death pathway in differentiated PC12 cells. Apoptosis 15(12):1480–1493
Mimura N, Fulciniti M, Gorgun G, Tai Y-T, Cirstea D, Santo L et al (2012) Blockade of XBP1 splicing by inhibition of IRE1α is a promising therapeutic option in multiple myeloma. Blood 119(24):5772–5781
Ming J, Ruan S, Wang M, Ye D, Fan N, Meng Q et al (2015a) A novel chemical, STF-083010, reverses tamoxifen-related drug resistance in breast cancer by inhibiting IRE1/XBP1. Oncotarget 6(38):40692–40703. https://doi.org/10.18632/oncotarget.5827
Ming J, Ruan S, Wang M, Ye D, Fan N, Meng Q, Tian B, Huang T (2015b) A novel chemical, STF-083010, reverses tamoxifen-related drug resistance in breast cancer by inhibiting IRE1/XBP1. Oncotarget 6(38):40692–40703
Namba T, Chu K, Kodama R, Byun S, Yoon KW, Hiraki M, Mandinova A, Lee SW (2015) Loss of p53 enhances the function of the endoplasmic reticulum through activation of the IRE1α/XBP1 pathway. Oncotarget 6(24):19990–20001
Papandreou I, Denko NC, Olson M, Van Melckebeke H, Lust S, Tam A et al (2011a) Identification of an Ire1alpha endonuclease specific inhibitor with cytotoxic activity against human multiple myeloma. Blood 117(4):1311–1314. https://doi.org/10.1182/blood-2010-08-303099
Papandreou, Ioanna, Denko, Nicholas C, Olson, Michael, Van Melckebeke, Heleen, Lust, Sofie, Tam, Arvin, . . . Niwa, Maho. (2011b). Identification of an Ire1alpha endonuclease specific inhibitor with cytotoxic activity against human multiple myeloma. Blood, 117(4), 1311–1314
Pokhriyal R, Hariprasad R, Kumar L, Hariprasad G (2019) Chemotherapy Resistance in Advanced Ovarian Cancer Patients. Biomarkers in cancer, 11, 1179299X19860815-11179299X19860815. doi: 10.1177/1179299X19860815
Puthalakath H, O'Reilly LA, Gunn P, Lee L, Kelly PN, Huntington ND et al (2007) ER stress triggers apoptosis by activating BH3-only protein Bim. Cell 129(7):1337–1349
Racek T, Buhlmann S, Rüst F, Knoll S, Alla V, Pützer BM (2008) Transcriptional repression of the prosurvival endoplasmic reticulum chaperone GRP78/BIP by E2F1. J Biol Chem 283(49):34305–34314
Rao RV, Ellerby HM, Bredesen DE (2004) Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ 11(4):372–380. https://doi.org/10.1038/sj.cdd.4401378
Rasheva VI, Domingos PM (2009) Cellular responses to endoplasmic reticulum stress and apoptosis. Apoptosis 14(8):996–1007. https://doi.org/10.1007/s10495-009-0341-y
Ruiz-Vela A, Opferman JT, Cheng EH, Korsmeyer SJ (2005) Proapoptotic BAX and BAK control multiple initiator caspases. EMBO Rep 6(4):379–385. https://doi.org/10.1038/sj.embor.7400375
Sak K (2015) In vitro cytotoxic activity of flavonoids on human ovarian cancer cell lines. Cancer Sci Res 2(1):1–13
Saleh M, Mathison JC, Wolinski MK, Bensinger SJ, Fitzgerald P, Droin N, Ulevitch RJ, Green DR, Nicholson DW (2006) Enhanced bacterial clearance and sepsis resistance in caspase-12-deficient mice. Nature 440(7087):1064–1068. https://doi.org/10.1038/nature04656
Saleh M, Vaillancourt JP, Graham RK, Huyck M, Srinivasula SM, Alnemri ES, Steinberg MH, Nolan V, Baldwin CT, Hotchkiss RS, Buchman TG, Zehnbauer BA, Hayden MR, Farrer LA, Roy S, Nicholson DW (2004) Differential modulation of endotoxin responsiveness by human caspase-12 polymorphisms. Nature 429(6987):75–79. https://doi.org/10.1038/nature02451
Shahrestanaki MK, Arasi FP, Aghaei M (2019) Adenosine protects pancreatic beta cells against apoptosis induced by endoplasmic reticulum stress. J Cell Biochem 120(5):7759–7770
Sheng X, Arnoldussen YJ, Storm M, Tesikova M, Nenseth HZ, Zhao S et al (2015) Divergent androgen regulation of unfolded protein response pathways drives prostate cancer. EMBO Mol Med 7(6):788–801. https://doi.org/10.15252/emmm.201404509
Sheng X, Nenseth HZ, Qu S, Kuzu OF, Frahnow T, Simon L et al (2019) IRE1α-XBP1s pathway promotes prostate cancer by activating c-MYC signaling. Nat Commun 10(1):1–12
Storniolo A, Alfano V, Carbotta S, Ferretti E, Renzo D, Livia (2018) IRE1α deficiency promotes tumor cell death and eIF2α degradation through PERK dipendent autophagy. Cell death discovery 4(1):1–12
Suh DH, Kim M-K, Kim HS, Chung HH, Song YS (2012) Unfolded protein response to autophagy as a promising druggable target for anticancer therapy. Ann N Y Acad Sci 1271(1):20
Sun H, Lin DC, Guo X, Kharabi Masouleh B, Gery S, Cao Q et al (2016) Inhibition of IRE1alpha-driven pro-survival pathways is a promising therapeutic application in acute myeloid leukemia. Oncotarget 7(14):18736–18749. https://doi.org/10.18632/oncotarget.7702
Szegezdi E, Logue SE, Gorman AM, Samali A (2006) Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7(9):880–885
Tabas I, Ron D (2011) Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol 13(3):184–190. https://doi.org/10.1038/ncb0311-184
Tay KH, Jin L, Tseng HY, Jiang CC, Ye Y, Thorne RF, Liu T, Guo ST, Verrills NM, Hersey P, Zhang XD (2012) Suppression of PP2A is critical for protection of melanoma cells upon endoplasmic reticulum stress. Cell Death Dis 3:e337. https://doi.org/10.1038/cddis.2012.79
Tinhofer I, Anether G, Senfter M, Pfaller K, Bernhard D, Hara M, Greil R (2002) Stressful death of T-ALL tumor cells after treatment with the anti-tumor agent Tetrocarcin-a. FASEB J 16(10):1295–1297. https://doi.org/10.1096/fj.02-0020fje
Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD et al (2018) Ovarian cancer statistics, 2018. CA Cancer J Clin 68(4):284–296
Tungkum W, Jumnongprakhon P, Tocharus C, Govitrapong P, Tocharus J (2017) Melatonin suppresses methamphetamine-triggered endoplasmic reticulum stress in C6 cells glioma cell lines. J Toxicol Sci 42(1):63–71
Wang G, Yang Z-Q, Zhang K (2010) Endoplasmic reticulum stress response in cancer: molecular mechanism and therapeutic potential. Am J Transl Res 2(1):65–74
Wang M, Kaufman RJ (2014) The impact of the endoplasmic reticulum protein-folding environment on cancer development. Nat Rev Cancer 14(9):581–597
Wu J, He GT, Zhang WJ, Xu J, Huang QB (2016) IRE1α signaling pathways involved in mammalian cell fate determination. Cell Physiol Biochem 38(3):847–858. https://doi.org/10.1159/000443039
Acknowledgments
Parts of this project was financially helped by Isfahan University of Medical Sciences (Grant Number: 396511).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barez, S.R., Atar, A.M. & Aghaei, M. Mechanism of inositol-requiring enzyme 1-alpha inhibition in endoplasmic reticulum stress and apoptosis in ovarian cancer cells. J. Cell Commun. Signal. 14, 403–415 (2020). https://doi.org/10.1007/s12079-020-00562-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-020-00562-7