Abstract
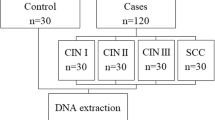
Cervical cancer (CC) is the second most common cancer in Mexican women. Human papillomavirus (HPV) infection is necessary but not sufficient for CC development. Furthermore, genetic factors as polymorphisms could be important susceptibility factors. Controversial results regarding TP53 polymorphisms specifically in codon 72 of CC have been reported. In the present work, the exon 4 sequence of TP53 gene in CC and healthy Mexican-mestizo women were analyzed. A group of 111 women with CC and 126 healthy women (control) were included. Peripheral blood cells for polymorphism analysis and cervical scrape for HPV detection were used. PCR of exon 4 of TP53 were subjected to denaturing high-performance liquid chromatography (DHPLC) analysis and sequencing. HPV detection was subjected to PCR and sequencing. The statistical analysis was carried out using the Arlequin software. Codon 72 Arg/Arg was the most common SNP detected, and Hardy–Weinberg analysis showed equilibrium in control and CC samples (P > 0.05). Wild type sequence of TP53 exon 4 was detected in 66 and 57% in control and CC samples, respectively. For codon 72 Arg/Arg, differences between control and CC women were found (P = 0.043). An association between HPV 16/18 infection and 72 Arg/Arg in woman with CC was found (P = 0.026). Haplotype GC (codon 36 and 72) was statistically significantly associated with CC (P = 0.011). HPV 16 was the most common viral type. Codon 72 Arg/Arg is the most common polymorphism in the Mexican population and could be associated to HPV 16 and/or HPV 18 infection in CC.


Similar content being viewed by others
References
http://www.inegi.org.mx/inegi/contenidos/espanol/prensa/Contenidos/estadisticas/2007/cancer07.pdf.
Walboomers JM, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189:12–9.
Castellsague X, Bosch FX, Muñoz N. Environmental co-factors in HPV carcinogenesis. Virus Res. 2002;89:191.
Kirk B, Feinsod M, Favis R, Kliman RM, Barany F. Single nucleotide polymorphism seeking long term association with complex disease. Nucleic Acids Res. 2002;30:3295–311.
Storey A, et al. Role of a p53 polymorphism in the development of human papillomavirus-associated cancer. Nature. 1998;393:229–34.
Thomas M, et al. Two Polymorphic variants of wild-type p53 differ biochemically and biologically. Mol Cel Biol. 1999;19:1092–100.
Jee SH, et al. Polymorphism p53 codon-72 and invasive cervical cancer: a meta-analysis. Int J Gynaecol Obstet. 2004;85:301–8.
Makni H, et al. P53 polymorphism in codon 72 and risk of human papillomavirus-induced cervical cancer: effect of inter-laboratory variation. Int J Cancer. 2000;87:528–33.
Cohen V, et al. Evaluation of denaturing high-performance liquid chromatography as a rapid detection method for identification of epidermal growth factor receptor mutations in nonsmall-cell lung cancer. Cancer. 2006;107:2858–65.
Gross E, Kiechle M, Arnold N. Mutation analysis of p53 in ovarian tumors by DHPLC. J Biochem Biophys Methods. 2001;47:73–81.
Terris MK, Peehl DM. Human papillomavirus detection by polymerase chain reaction in benign and malignant prostate tissue is dependent on the primer set utilized. Urology. 1997;50:150–6.
Manos M. Use of polymerase chain reaction amplification for the detection of genital human papillomaviruses. In: Forth M, Greaves M, et al., editors. Cancer Cell 7º molecular diagnostics of human cancer. New York: Cold Spring Harbor Laboratory; 1988. p. 209–14.
Sousa H, Santos AM, Pinto D, Medeiros R. Is the p53 codon 72 polymorphism a key biomarker for cervical cancer development? A meta-analysis review within European populations. Int J Mol Med. 2007;20:731–41.
Koushik A, Platt RW. Franco EL: p53 codon 72 polymorphism and cervical neoplasia: a meta-analysis review. Cancer Epidemiol Biomarkers Prev. 2004;13:11–22.
Klug SJ, et al. TP53 codon 72 polymorphism and cervical cancer: a pooled analysis of individual data from 49 studies. Lancet Oncol. 2009;10:772–84.
Zheng XZ, et al. Ethnicity determines association of p53Arg72Pro alleles with cervical cancer in China. Eur J Cancer Prev. 2008;17:460–6.
Suarez-Rincon AE, Moran-Moguel MC, Montoya-Fuentes H, Gallegos MP, Sánchez-Corona JL. Polymorphism in codon 72 of the p53 gene and cervico-uterine cancer risk in Mexico. Ginecol Obstet Mex. 2002;70:344–8.
Min-Min H, Ming-Rong X, Ze-yi C, Kai-xuan Y, Zhi-lin S. Analysis of p53 codon 72 polymorphism and its association with human papillomavirus 16 and 18 E6 in Chinese cervical lesions. Int J Gynecol Cancer. 2006;16:2004–8.
Saranath D, et al. HPV16/18 prevalence in cervical lesions/cancers and p53 genotypes in cervical cancer patients from India. Gynecol Oncol. 2002;86:157–62.
Brenna SM, et al. Prognostic value of P53 codon 72 polymorphism in invasive cervical cancer in Brazil. Gynecol Oncol. 2004;93(2):374–8.
Schwartz SM, et al. Human papillomavirus and prognosis of invasive cervical cancer: a population-based study. J Clin Oncol. 2001;19:1906–15.
Huang LW, Chao SL, Hwang JL. Human papillomavirus-31-related types predict better survival in cervical carcinoma. Cancer. 2004;100:327–34.
Shah W, et al. The prevalence of human papillomavirus type 58 in Chinese patients with cervical carcinoma and its influence on survival. Clin Oncol (R Coll Radiol). 2009;21:768–74.
Hiller T, Poppelreuther S, Stubenrauch F, Iftner T. Comparative analysis of 19 genital human papillomavirus types with regard to p53 degradation, immortalization, phylogeny, and epidemiologic risk classification. Cancer Epidemiol Biomarkers Prev. 2006;15:1262–7.
van Duin M, et al. Analysis of human papillomavirus type 16 E6 variants in relation to p53 codon 72 polymorphism genotypes in cervical carcinogenesis. J Gen Virol. 2000;81:317–25.
Bergamaschi D, et al. p53 polymorphism influences response in cancer chemotherapy via modulation of p73-dependent apoptosis. Cancer Cell. 2003;3:387–402.
Bojesen SE, Nordestgaard BG. The common germline Arg72Pro polymorphism of p53 and increased longevity in humans. Cell Cycle. 2008;7:158–63.
Koivusalo R, Hietanen S. The cytotoxicity of chemotherapy drugs varies in cervical cancer cells depending on the p53 status. Cancer Biol Ther. 2004;3:1177–83.
Varley JM, et al. Characterization of germline TP53 splicing mutations and their genetic and functional analysis. Oncogene. 2001;20:2647–54.
Tenti P, et al. p53 codon 72 polymorphism does not affect the risk of cervical cancer in patients from northern Italy. Cancer Epidemiol Biomarkers Prev. 2000;9:435–8.
Rosenthal AN, et al. p53 codon 72 polymorphism and risk of cervical cancer in UK. Lancet. 1998;352:871–2.
Tong D, et al. Detection of p53 polymorphism at codon 72 by PCR and allele-specific oligonucleotide hybridization on microtiter plates. Clin Chem. 2000;46:124–6.
Klug SJ, et al. TP53 Polymorphism, HPV Infection, and Risk of Cervical Cancer. Cancer Epidemiol Biomarkers Prev. 2001;10:1009–12.
Fernandes TA, Lima GL, De Souza FC, Fernandez JV, Meissner RV. Evaluation of the polymorphisms in the exons 2 to 4 of the TP53 in cervical carcinoma patients from a Brazilian population. Mol Biol 2008; 54 Suppl: OL1025–31.
Acknowledgments
This work was supported by grants from CONACyT 7114, and partially by SALUD-2006-COI-44633. During this work, P. Piña-Sáncez was recipient of fellowship from CONACyT and DGEP (Dirección General de Estudios de Posgrado, UNAM). This work was submitted in partial fulfillment of the requirements for the DSc degree in for P. Piña-Sánchez at Doctorado En Ciencias Biomédicas, Universidad Nacional Autonoma De Mexico. We are grateful to Psicofarma S.A. de C.V. for DHPLC equipment facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piña-Sánchez, P., Hernández-Hernández, D.M., Taja-Chayeb, L. et al. Polymorphism in exon 4 of TP53 gene associated to HPV 16 and 18 in Mexican women with cervical cancer. Med Oncol 28, 1507–1513 (2011). https://doi.org/10.1007/s12032-010-9599-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-010-9599-8