Abstract
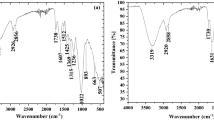
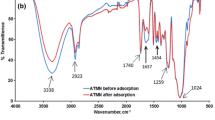
The adsorption capacity of ground hazelnut (HS) and almond (AS) shells towards Pb(II) and Cd(II) has been studied at pH = 5, in NaNO3 and NaCl ionic media, in the ionic strength range 0.05–0.5 mol L−1. Kinetic and equilibrium experiments were carried out by using the Differential Pulse Anodic Stripping Voltammetry technique to check the amount of the metal ion removed by HS and AS materials. Different kinetic and equilibrium equations were used to fit experimental data and a statistical study was done to establish the suitable model for the data fitting. A speciation study of the metal ions in solution was also done in order to evaluate the influence of the ionic medium on the adsorption process. TGA-DSC, FT-IR, and SEM-EDX techniques were used to characterize the adsorbent materials. The mechanism of metal ions adsorption was explained on the basis of the results obtained by the metal ions speciation study and the characterization of materials.







Similar content being viewed by others
References
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Autom Control 19(6):716–723. https://doi.org/10.1109/TAC.1974.1100705
Allouch M, Alami M, Boukhlifi F (2014) Kinetic and energy study of thermal degradation of biomass materials under oxidative atmosphere using TGA, DTA and DSC. JMEST 1(5):74–78
Baes CF, Mesmer RE (eds) (1976) The hydrolysis of cations. J. Wiley & Sons, New York
Blázquez G, Ronda A, Martín-Lara MA, Pérez A, Calero M (2015) Comparative study of isotherm parameters of lead biosorption by two wastes of olive-oil production. Water Sci Technol J Int Assoc Water Pollut Res 72(5):711–720. https://doi.org/10.2166/wst.2015.153
Bulut Y, Tez Z (2007) Adsorption studies on ground shells of hazelnut and almond. J Hazard Mater 149(1):35–41. https://doi.org/10.1016/j.jhazmat.2007.03.044
Cataldo S, Gianguzza A, Pettignano A et al (2012) Complex formation of copper(II) and cadmium(II) with pectin and polygalacturonic acid in aqueous solution. An ISE-H+ and ISE-Me2+ electrochemical study. Int J Electrochem Sci 7:6722–6737
Cataldo S, Cavallaro G, Gianguzza A, Lazzara G, Pettignano A, Piazzese D, Villaescusa I (2013a) Kinetic and equilibrium study for cadmium and copper removal from aqueous solutions by sorption onto mixed alginate/pectin gel beads. J Environ Chem Eng 1(4):1252–1260. https://doi.org/10.1016/j.jece.2013.09.012
Cataldo S, Gianguzza A, Pettignano A, Villaescusa I (2013b) Mercury(II) removal from aqueous solution by sorption onto alginate, pectate and polygalacturonate calcium gel beads. A kinetic and speciation based equilibrium study. React Funct Polym 73(1):207–217. https://doi.org/10.1016/j.reactfunctpolym.2012.10.005
Cataldo S, Muratore N, Orecchio S, Pettignano A (2015) Enhancement of adsorption ability of calcium alginate gel beads towards Pd(II) ion. A kinetic and equilibrium study on hybrid Laponite and Montmorillonite-alginate gel beads. Appl Clay Sci 118:162–170. https://doi.org/10.1016/j.clay.2015.09.014
Cataldo S, Gianguzza A, Milea D, Muratore N, Pettignano A (2016a) Pb(II) adsorption by a novel activated carbon–alginate composite material. A kinetic and equilibrium study. Int J Biol Macromol 92:769–778. https://doi.org/10.1016/j.ijbiomac.2016.07.099
Cataldo S, Gianguzza A, Pettignano A (2016b) Sorption of Pd(II) ion by calcium alginate gel beads at different chloride concentrations and pH. A kinetic and equilibrium study. Arab J Chem 9(5):656–667. https://doi.org/10.1016/j.arabjc.2014.10.031
Cimino G, Passerini A, Toscano G (2000) Removal of toxic cations and Cr(VI) from aqueous solution by hazelnut shell. Water Res 34(11):2955–2962. https://doi.org/10.1016/S0043-1354(00)00048-8
Crea F, Foti C, Milea D, Sammartano S (2013) Speciation of cadmium in the environment. Met Ions Life Sci 11:63–83. https://doi.org/10.1007/978-94-007-5179-8_3
Crompton TR (2007) Toxicants in aqueous ecosystems: a guide for the analytical and environmental chemist. Springer-Verlag, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-35741-4
De Stefano C, Sammartano S, Mineo P, Rigano C (1997) Computer tools for the speciation of natural fluids. In: Gianguzza A, Pelizzetti E, Sammartano S (eds) Marine chemistry—an environmental analytical chemistry approach. Kluwer Academic Publishers, Amsterdam, pp 71–83
De Stefano C, Lando G, Milea D, Pettignano A, Sammartano S (2010) Formation and stability of cadmium(II)/Phytate complexes by different electrochemical techniques. Critical analysis of results. J Solut Chem 39(2):179–195. https://doi.org/10.1007/s10953-009-9493-1
Demirbaş A (2002) Fuel characteristics of olive husk and walnut, hazelnut, sunflower, and almond shells. Energy Sources 24(3):215–221. https://doi.org/10.1080/009083102317243601
Demirbas A (2008) Heavy metal adsorption onto agro-based waste materials: a review. J Hazard Mater 157(2-3):220–229. https://doi.org/10.1016/j.jhazmat.2008.01.024
Fu F, Wang Q (2011) Removal of heavy metal ions from wastewaters: a review. J Environ Manag 92(3):407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
Haykiri-Acma H, Yaman S (2012) Comparison of the combustion behaviours of agricultural wastes under dry air and oxygen. WIT Trans Ecol Environ 163:145–151. https://doi.org/10.2495/WM120141
Ho YS, Ofomaja AE (2006) Pseudo-second-order model for lead ion sorption from aqueous solutions onto palm kernel fiber. J Hazard Mater 129(1-3):137–142. https://doi.org/10.1016/j.jhazmat.2005.08.020
International Nut&Dried Fruit (2015) Global statistical review 2014/2015. https://www.nutfruit.org/files/tech/global-statistical-review-2014-2015_101779.pdf
Kim N, Park M, Park D (2015) A new efficient forest biowaste as biosorbent for removal of cationic heavy metals. Bioresour Technol 175:629–632. https://doi.org/10.1016/j.biortech.2014.10.092
Kocabaş-Atakli ZÖ, Okyay-Öner F, Yürüm Y (2015) Combustion characteristics of Turkish hazelnut shell biomass, lignite coal, and their respective blends via thermogravimetric analysis. J Therm Anal Calorim 119(3):1723–1729. https://doi.org/10.1007/s10973-014-4348-4
Martin AR, Martins MA, da Silva ORRF, Mattoso LHC (2010) Studies on the thermal properties of sisal fiber and its constituents. Thermochim Acta 506(1-2):14–19. https://doi.org/10.1016/j.tca.2010.04.008
Mehrasbi MR, Farahmandkia Z, Taghibeigloo B, Taromi A (2009) Adsorption of lead and cadmium from aqueous solution by using almond shells. Water Air Soil Pollut 199(1-4):343–351. https://doi.org/10.1007/s11270-008-9883-9
Micke W (1996) Almond production manual. Regents of the University of California, Oakland. University of California, California
Militky J, Meloun M (2011) Statistical data analysis: a practical guide., AbeBooks. Woodhead publishing ltd, New Delhi
Monji AB, Ghoulipour V, Mallah MH (2016) Biosorption of toxic transition metals and radionuclides from aqueous solutions by agro-industrial byproducts. J Hazard Toxic Radioact Waste 20:1–9
Nguyen TAH, Ngo HH, Guo WS, Zhang J, Liang S, Yue QY, Li Q, Nguyen TV (2013) Applicability of agricultural waste and by-products for adsorptive removal of heavy metals from wastewater. Bioresour Technol 148:574–585. https://doi.org/10.1016/j.biortech.2013.08.124
Nurchi VM, Villaescusa I (2008) Agricultural biomasses as sorbents of some trace metals. Coord Chem Rev 252(10-11):1178–1188. https://doi.org/10.1016/j.ccr.2007.09.023
OriginLab (2004) Origin. OriginLab, Northampton
Park D, Yun Y-S, Park JM (2010) The past, present, and future trends of biosorption. Biotechnol Bioprocess Eng 15(1):86–102. https://doi.org/10.1007/s12257-009-0199-4
Pehlivan E, Altun T, Cetin S, Iqbal Bhanger M (2009) Lead sorption by waste biomass of hazelnut and almond shell. J Hazard Mater 167(1-3):1203–1208. https://doi.org/10.1016/j.jhazmat.2009.01.126
Ramiah MV (1970) Thermogravimetric and differential thermal analysis of cellulose, hemicellulose, and lignin. J Appl Polym Sci 14(5):1323–1337. https://doi.org/10.1002/app.1970.070140518
Ronda A, Martín-Lara MA, Dionisio E, Blázquez G, Calero M (2013) Effect of lead in biosorption of copper by almond shell. J Taiwan Inst Chem Eng 44(3):466–473. https://doi.org/10.1016/j.jtice.2012.12.019
Ruthven DM (1984) Principles of adsorption and adsorption processes. J. Wiley & Sons
Şen A, Pereira H, Olivella MA, Villaescusa I (2014) Heavy metals removal in aqueous environments using bark as a biosorbent. Int J Environ Sci Technol 12:391–404
Şencan A, Karaboyacı M, Kılıç M (2015) Determination of lead(II) sorption capacity of hazelnut shell and activated carbon obtained from hazelnut shell activated with ZnCl2. Environ Sci Pollut Res Int 22(5):3238–3248. https://doi.org/10.1007/s11356-014-2974-9
Skreiberg A, Skreiberg Ø, Sandquist J, Sørum L (2011) TGA and macro-TGA characterisation of biomass fuels and fuel mixtures. Fuel 90(6):2182–2197. https://doi.org/10.1016/j.fuel.2011.02.012
Taşar Ş, Kaya F, Özer A (2014) Biosorption of lead(II) ions from aqueous solution by peanut shells: equilibrium, thermodynamic and kinetic studies. J Environ Chem Eng 2(2):1018–1026. https://doi.org/10.1016/j.jece.2014.03.015
Volesky B (2003) Sorption and biosorption. BV Sorbex, Inc., Montréal
Wagenmakers E-J, Farrell S (2004) AIC model selection using Akaike weights. Psychon Bull Rev 11(1):192–196. https://doi.org/10.3758/BF03206482
Zhang Y, Zhao J, Jiang Z, Shan D, Lu Y (2014) Biosorption of Fe(II) and Mn(II) ions from aqueous solution by rice husk ash. Biomed Res Int 2014:973095. https://doi.org/10.1155/2014/973095
Funding
We thank the Italian Ministero dell’Istruzione, dell’Università e della Ricerca (MIUR, PRIN project n. 2015MP34H3_003 and 2015MP34H3_004) for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Guilherme L. Dotto
Electronic supplementary material
ESM 1
(DOCX 1.91 mb)
Rights and permissions
About this article
Cite this article
Cataldo, S., Gianguzza, A., Milea, D. et al. A critical approach to the toxic metal ion removal by hazelnut and almond shells. Environ Sci Pollut Res 25, 4238–4253 (2018). https://doi.org/10.1007/s11356-017-0779-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0779-3