Abstract
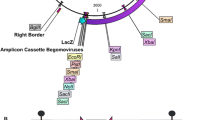
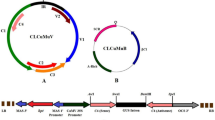
Several important biological processes are performed by distinct functional domains found on replication-associated protein (Rep) encoded by AC1 of geminiviruses. Two truncated forms of replicase (tAC1) gene, capable of expressing only the N-terminal 669 bp (5′AC1) and C-terminal 783 bp (3′AC1) nucleotides cloned under transcriptional control of the CaMV35S were introduced into cotton (Gossypium hirsutum L.) using LBA4404 strain of Agrobacterium tumefaciens to make use of an interference strategy for impairing cotton leaf curl virus (CLCuV) infection in transgenic cotton. Compared with nontransformed control, we observed that transgenic cotton plants overexpressing either N-terminal (5′AC1) or C-terminal (3′AC1) sequences confer resistance to CLCuV by inhibiting replication of viral genomic and β satellite DNA components. Molecular analysis by Northern blot hybridization revealed high transgene expression in early and late growth stages associated with inhibition of CLCuV replication. Of the eight T1 transgenic lines tested, six had delayed and minor symptoms as compared to nontransformed control lines which developed disease symptoms after 2–3 weeks of whitefly-mediated viral delivery. Virus biological assay and growth of T2 plants proved that transgenic cotton plants overexpressing 5′- and 3′AC1 displayed high resistance level up to 72, 81%, respectively, as compared to non-transformed control plants following inoculation with viruliferous whiteflies giving significantly high cotton seed yield. Progeny analysis of these plants by polymerase chain reaction (PCR), Southern blotting and virus biological assay showed stable transgene, integration, inheritance and cotton leaf curl disease (CLCuD) resistance in two of the eight transgenic lines having single or two transgene insertions. Transgenic cotton expressing partial AC1 gene of CLCuV can be used as virus resistance source in cotton breeding programs aiming to improve virus resistance in cotton crop.






Similar content being viewed by others
References
S. Mansoor, R.W. Briddon, Y. Zafar, J. Stanley, Trends Plant Sci. 8, 128 (2003)
A.S. Moffat, Science 286, 835 (1999)
M.I. Boulton, Ann. Appl. Biol. 142, 143 (2003)
M. Rahman, D. Hussain, Y. Zafar, Crop Sci. 42, 2137 (2002)
R.W. Briddon, S. Mansoor, I.D. Bedford, M.S. Pinner, K. Saunders, J. Stanley, Y. Zafar, K.A. Malik, P.G. Markham, Virology 285, 234 (2001)
R.N. Beachy, Curr. Opin. Biotechnol. 8, 215 (1997)
P.V. Shivaprasad, V.B. Thillaichidambaram, K. Valuthambi, Virus Genes 33, 365 (2006)
S. Asad, W.A.A. Haris, A. Bashir, Y. Zafar, K.A. Malik, C.P. Lichtenstein, Arch. Virol. 148, 2341 (2003)
U. Ganeson, S.S. Suri, S. Rajasubraminiam, M.V. Rajam, I. Dasgupta, Virus Genes 39, 113 (2009)
Y. Hong, J. Stanley, Mol. Plant Microbe Interact. 9, 219 (1996)
P. Chellappan, M.V. Masona, R. Vanitharani, N.J. Taylor, C.M. Fauquet, Plant Mol. Biol. 56, 601 (2004)
S.F. Hanson, D.P. Maxwell, Phytopathology 89, 480 (1999)
A. Sangare, D. Deng, C.M. Fauquet, R.N. Beachy, Mol. Biol. Rep. 5, 95 (1999)
A.G. Day, E.R. Bejarano, K.W. Buck, M. Burrell, C.P. Lichtenstein, Proc. Natl. Acad. Sci. USA 88, 6721 (1991)
E.R. Bejarano, C.P. Lichtenstein, Plant Mol. Biol. 24, 241 (1994)
M. Bendahmane, B. Gronenborn, Plant Mol. Biol. 33, 351 (1997)
F.J.L. Aragao, S.G. Ribeiro, L.M.G. Barros, A.C.M. Brasiliero, D.P. Maxwell, E.L. Rech, J.C. Faria, Mol. Breed. 4, 491 (1998)
E. Noris, G.P. Accotto, R. Tavazza, A. Brunetti, S. Crespi, M. Tavazza, Virolology 224, 130 (1996)
T.A. Wilkins, R. Mishra, N.L. Trolinder, J. Food Agric. Environ. 2, 179 (2004)
J. Sambrook, F.F. Fritsch, T. Maniatis, Moleculat Cloning: A Laboratory Manual, 2nd edn. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1989), pp. 1.3–1.4
S. Asad, Z. Mukhtar, F. Nazir, J.A. Hashmi, S. Mansoor, Y. Zafar, M. Arshad, Mol. Biotechnol. 40, 161 (2008)
J.D.G. Jones, Curr. Opin. Plant Biol. 4, 281 (2001)
R.M. Solomon-Blackburn, J.E. Bradshaw, Potato Res. 58, 87 (2007)
M. Rahman, D. Hussain, K.A. Malik, Y. Zafar, Plant Path. 54, 764 (2005)
M. Padidam, S. Sawyer, C.M. Fauquet, Virology 265, 218 (1999)
R.W. Briddon, Mol. Plant Pathol. 4, 427 (2003)
J.M. Anderson, D.L. Bucholtz, A.E. Greene, M.G. Francki, S.M. Gray, H. Sharma, H.W. Ohm, K.L. Perry, Phytopathology 88, 851 (1998)
M. Prins, M. Laimer, E. Noris, J. Schubert, M. Wassenger, M. Tepfer, Mol. Plant Pathol. 9, 73 (2008)
F.J. Perlak, R.W. Deaton, T.A. Armstrong, R.L. Fuchs, S.R. Sims, J.T. Greenplate, D.A. Fischhoff, Bio/Technology 8, 939 (1990)
J.C. Sanford, S.A. Johnston, J. Theor. Biol. 113, 395 (1985)
N. Taylor, P. Chavariaga, K. Raemakers, D. Siritunga, P. Zhang, Plant Mol. Biol. 56, 671 (2004)
C. Desbeiz, H. Lecoq, Plant Pathol. 46, 809 (1997)
J. Stanley, Vet. Microbiol. 88, 121 (2004)
A. Lucioli, E. Noris, A. Brunetti, R. Tavazza, V. Ruzza, A.G. Castillo, E.R. Bejarano, G.P. Accoto, M. Tavaza, J. Virol. 77, 6785 (2003)
P. Hanson, S.K. Green, G. Kuo, Tomato Genet. Coop. Rep. 56, 17 (2006)
A. Brunetti, M. Tavazza, E. Noris, R. Tavazza, P. Caciagli, G. Ancora, S. Crespi, G.P. Acotto, Mol. Plant Microbe Interact. 10, 571 (1997)
R. Goldbach, E. Bucher, M. Prins, Virus Res. 92, 207 (2003)
F. Heyraud, V. Matzeit, M. Kammann, S. Schaefer, J. Schell, B. Gronenborn, EMBO J. 12, 4445 (1993)
A.I. Sanz, A. Fraile, F.G. Arnel, X. Zhou, D.J. Robinson, S. Khalid, T. Butt, B.D. Harrison, J. Gen. Virol. 81, 1839 (2000)
Acknowledgments
We would like to thank Miss. Shahnaz Akhtar, Mrs. Kalsoom Akhtar, Mr Saeed Faisal, and Mr. Karim Bukhsh for their help in tissue culture and glasshouse trials, respectively. We also acknowledge PSF and HEC for providing financial support to carry out this work. We also extend our thanks to Dr. John P. Carr, Department of Plant Sciences, University of Cambridge for providing access to his Lab and to carry out some hybridization work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hashmi, J.A., Zafar, Y., Arshad, M. et al. Engineering cotton (Gossypium hirsutum L.) for resistance to cotton leaf curl disease using viral truncated AC1 DNA sequences. Virus Genes 42, 286–296 (2011). https://doi.org/10.1007/s11262-011-0569-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-011-0569-9