Abstract
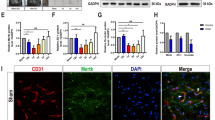
While electro-acupuncture (EA) has been well known to contribute towards neuroplasticity occurring in both the central and the peripheral nervous system after injury, the underlying mechanism remains largely unknown. This study evaluated the effects and the possible mechanism of EA on neuronal apoptosis in the spinal cords of cats subjected to the removal of L1–L5 and L7–S2 dorsal root ganglion, sparing the L6 dorsal root ganglion. EA treatment decreased the number of TUNEL-positive apoptotic cells in lamina II of the L3 and L6 cord segments at 7 and 14 days post operation (dpo). This EA-mediated neuroprotection is associated with a decrease in the number of Bax immunoreactive neurons and an increase in the number of Bcl-2 immunoreactive neurons. Furthermore, Western blot and RT-PCR analysis revealed a significant downregulation of Bax protein and its mRNA, but an upregulation of Bcl-2 in the dorsal horn of L3 and L6 cords at both 7 and 14 dpo. The present findings suggest that EA could inhibit neuronal apoptosis in dorsal root deafferentated cat spinal cords, possibly by Bax downregulation and Bcl-2 upregulation.




Similar content being viewed by others
References
Liu CN, Chambers WW (1958) Intraspinal sprouting of dorsal root axon. Arch Neurol Psychiatry 79:46–61
Lynch G, Gall C, Rose G et al (1976) Change in the distribution of the dentate gyrus associational system following unilateral or bilateral entorhinal lesions in the adult rat. Brain Res 110:57–71
Cotman CW, Nieto-Sampedro M, Harris EW (1981) Synapse replacement in the nervous system of adult vertebrates. Physiol Rev 61:684–784
Wu LF, Goldberger ME, Murray M (1986) Dorsal root terminal in lamina II of cat spinal cord: effects of partial deafferentation. Anat Rec 214:147A
Wu LF, Wang YM (1990) The plasticity of synaptic terminal in spinal lamina II of complete lumbosacral dorsal ganglionectomy cat: electron microscopic quantitative study. Chin J Anat 13(suppl):230A
Korsmever SJ (1992) Bcl-2 initiates a new category of oncogenes: regulators of cell death. J Blood 80:879–886
Liu XZ, Xu XM, Hu R et al (1997) Neuronal and glial apoptosis after traumatic spinal cord injury. Neuroscience 17:5395–5406
Emery E, Aldana P, Bunge MB et al (1998) Apoptosis after traumatic human spinal cord injury. J Neurosurg 89:911–920
Nesic-Taylor O, Cittelly D, Ye Z et al (2005) Exogenous Bcl-xL fusion protein spares neurons after spinal cord injury. J Neurosci Res 795:628–637
Okuno S, Saito A, Havashi T et al (2004) The c-Jun N-terminal protein kinase signaling pathway mediates Bax activation and subsequent neuronal apoptosis through interaction with Bim after transient focal cerebral ischemia. J Neurosci 24:7879–7887
Qiu J, Nesic O, Ye Z et al (2001) Bcl-xL expression after contusion to the rat spinal cord. J Neurotrauma 18:1267–1278
Cook SA, Sugden PH, Clerk A (1999) Regulation of bcl-2 family proteins during development and in response to oxidative stress in cardiac myocytes: association with changes in mitochondrial membrane potential. Circ Res 85:940–949
Shinoura N, Yoshida Y, Asai A et al (1999) Relative level of expression of Bax and Bcl-XL determines the cellular fate of apoptosis/necrosis induced by the overexpression of Bax. Oncogene 18:5703–5713
Lee JU, Hosotani R, Wada M et al (1999) Role of Bcl-2 family proteins (Bax, Bcl-2 and Bcl-X) on cellular susceptibility to radiation in pancreatic cancer cells. Eur J Cancer 35:1374–1380
Luo C, Zhu C, Jiang JY et al (2001) The alterations of bcl-xL and bax mRNA expression following traumatic brain injury in rats. Chin J Traumatol 17:209–211
Motoyama N, Wang F, Roth KA et al (1995) Massive cell death of immature hematopoietic cells and neurons in Bcl-x-deficient mice. J Sci 267:1506–1510
White FA, Keller-Peck CR, Knudson CM et al (1998) Widespread elimination of naturally occurring neuronal death in Bax-deficient mice. J Neurosci 18:1428–1439
Sun W, Winseck A, Vinsant S et al (2004) Programmed cell death of adult-generated hippocampal neurons is mediated by the proapoptotic gene Bax. J Neurosci 24:11205–11213
Jin ZG, Tao ZL (1997) Study on electroacupuncture treatment of experimental spinal injury by histochemistry and immunohistochemical in cats. Chin Acupunct 8:489–492
Gao XP (1985) Acupuncture therapy of traumatic paraplegia. Chin Acupunct 5:3–4
Li GR, Ji L, Zhang S (1985) Effect of acupuncture in 124 cases of traumatic paraplegia. J Tradit Chin Med 26:34–36
Saito K, Wang C, He W (1989) Experimental and ultrastructural study of synapses in the cat spinal cord after acupuncture in zusanli. Chin J Neuroanat 5:157–164
Wu LF (1993) Effects of acupuncture on terminal of synapse in spinal cord. J WCUMS 24:117–122
Wu LF, Bao TR (1994) Electro-acupuncture stimulation enhances the neurite outgrowth promoting effect of tissue extract from Clarke’s nucleus of L6 root in cat. Chin J Neurosci 1:13–18
Dong HX, Wu LF, Bao TR (1994) The effect of acupuncture on plasticity in lamina II of cat spinal cord after partial deafferentation—a quantitative electron microscopic study. Chin J Neuroanat 10:6–11
Wang TH, Wu LF, Liao DY (2002) The effect of acupuncture on the expression of BDNF in the process of spinal plasticity. Chin J Neuroanat 18:55–58
Wang TT, Yuan Y, Kang Y et al (2005) Effects of acupuncture on the expression of glial cell line-derived neurotrophic factor (GDNF) and basic fibroblast growth factor (FGF-2/bFGF) in the left sixth lumbar dorsal root ganglion following removal of adjacent dorsal root ganglia. Neurosci Lett 382:236–241
Wang TT, Yuan WL, Ke Q et al (2006) Effects of electro-acupuncture on the expression of c-jun and c-fos in spared dorsal root ganglion and associated spinal laminae following removal of adjacent dorsal root ganglia in cats. Neuroscience 140:1169–1176
Cheng PT, Wong MK, Chang PL (1998) A therapeutic trial of acupuncture in neurogenic bladder of spinal cord injured patients—a preliminary report. Spinal Cord 36:476–480
Pei J, Sun L, Chen R et al (2001) The effect of electroacupuncture on motor function recovery in patients with acute cerebral infarction: a randomly controlled trial. J Tradit Chin Med 21:270–272
Wong AM, Leong CP, Su TY et al (2003) Clinical trial of acupuncture for patients with spinal cord injuries. Am J Phys Med Rehabil 82:21–27
Wang JG, Zheng QX, Zhao M (2006) Neural cell apoptosis and significant expression of Caspase-3, Fas expression after spinal cord injury in rats. Chin J Rehabil Med 21:296–300
Zhang HT, Zou XL, Song XB et al (2007) Immunohistochemical distribution of NGF, BDNF, NT-3, NT-4 in the brains of adult Rhesus monkeys. J Histochem Cytochem 55:1–19
Zhang ZY, Dang RS, Zhang CS et al (2006) Inhibitory effect of electro-acupuncture on apoptosis after spinal cord injury. Chin Nerve Regen Res 1:239–243
Cho NH, Lee JD, Cheong BS et al (2004) Acupuncture suppresses intrastriatal hemorrhage-induced apoptotic neuronal cell death in rats. Neurosci Lett 362:141–145
Zhang ZY, Cui YH, Yan ZG (2002) The effect of electric-acupuncture on apoptosis in spinal cord injury. Chin J Clin Rehabil 6:818–819
Craig RW (1995) The bcl-2 gene family. Semin Cancer Biol 6:35–43
Xiao RY, Wu LF, Li GR (1989) Influence of acupuncture on the plasticity of lamina II of cat spinal dorsal horn—a quantitative ultrastructural study. Chin J Neuroanat 5:149–155
Dong HX, Wu LF, Bao TR (1992) Influence of acupuncture on the plasticity of lamina II of cat spinal cord. Physiol Commun 3:211–214
Putcha GV, Deshmukh M, Johnson EM (1999) Bax translocation is a critical event in neuronal apoptosis: regulation by neuroprotectants, BCL-2, and Caspases. J Neurosci 19:7476–7485
Xu J, Wu LF, Bao TR (1992) The effect of electric acupuncture on the plasticity of the synaptic terminals in Clarke’s nucleus after partial lumbosacral deafferentation of the cat spinal cord—a quantitative ultrastructural study. Chin J Neuroanat 8:47–51
Lou J, Lenle LJ, Xu F et al (1998) In vivo Bcl-2 oncogene neuronal expression in the rat spinal cord. Spine 23:517–523
Oltvai ZN, Milliman CL, Korsmeyer SJ (1993) Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74:609–619
Li SH, Hou TS (2001) The impact of direct-current stimulation on apoptotic adjustable gene expression of spinal cord motorneurons after sciatic nerve transection. Chin J Traumatol 17:398–401
Acknowledgements
We thank Dr. Kevin J. Foehrkolb and associate Professor Jiang-Fan Chen in Harvard Medical School for their very useful comments on the manuscript. A grant from the National Science Foundation (No. 30260125) and the New York-China Medical Grant (CMB00-722) supported this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Wei Zhao and Qi Zhao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, W., Zhao, Q., Liu, J. et al. Electro-acupuncture Reduces Neuronal Apoptosis Linked to Bax and Bcl-2 Expression in the Spinal Cords of Cats Subjected to Partial Dorsal Root Ganglionectomy. Neurochem Res 33, 2214–2221 (2008). https://doi.org/10.1007/s11064-008-9677-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9677-x