Abstract
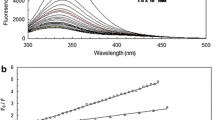
The aim of the present study is to characterize the competition between propranolol (PROP) and taxifolin (TAX) in binding to human serum albumin (HSA) in a physiological buffer (pH 7.4) using multiple spectroscopic, chemometrics and molecular dynamics simulation measurements. Fluorescence analysis was used to determine the binding and quenching properties of HSA–ligand complexes in binary and ternary systems. These spectral data were further analyzed by the multivariate curve resolution-alternating least squares method. In addition, the concentration profiles and pure spectra of three species (HSA, ligand and HSA–ligand complex) and the apparent equilibrium constants K app were evaluated. Fluorescence spectroscopy showed that in the presence of TAX, the binding constant of HSA–PROP increased. The effect of ligands on the secondary structure of the protein has been analyzed by using Fourier transform infrared spectra. The conformational change of the protein was analyzed using synchronous fluorescence spectroscopy, three-dimensional fluorescence spectra and molecular dynamics (MD) simulation. The results of synchronous fluorescence and three-dimensional fluorescence spectra show that PROP alters the microenvironment around the tryptophan (Trp) and tyrosine (Tyr) residues in the presence of TAX. According to the MD simulation, these ligands can interact with the protein, affecting the secondary structure of HSA and modifying its tertiary structure. MD simulations and experimental data support each other.









Similar content being viewed by others
References
Huang, B.X., Kim, H.Y., Dass, C.: Probing three-dimensional structure of bovine serum albumin by chemical cross-linking and mass spectrometry. J. Am. Soc. Mass Spectr. 15, 1237–1247 (2004)
Martini, S., Bonechi, C., Rossi, C.: Interaction between vine pesticides and bovine serum albumin studied by nuclear spin relaxation data. J. Agr. Food. Chem. 58, 10705–10709 (2010)
Kalanur, S.S., Seetharamappa, J., Kalalbandi, V.K.L.: Characterization of interaction and the effect of carbamazepine on the structure of human serum albumin. J. Pharm. Biomed. Anal. 53, 660–666 (2010)
Bourassa, P., Dubeau, S., Maharvi, GhM, Fauq, A.H., Thomas, T.J., TajmirRiahi, H.A.: Binding of antitumor tamoxifen and its metabolites 4-hydroxytamoxifen and endoxifen to human serum albumin. Biochimie 93, 1089–1101 (2011)
Zhu, L., Yang, F., Chen, L., Meehan, E.J., Huang, M.: A new drug binding subsite on human serum albumin and drug–drug interaction studied by X-ray crystallography. J. Struct. Biol. 162, 40–49 (2008)
Zohoorian-Abootorabi, T., Sanee, H., Iranfar, H., Saberi, M.R., Chamani, J.: Separate and simultaneous binding effects through a non-cooperative behavior between cyclophosphamide hydrochloride and fluoxymesterone upon interaction with human serum albumin: multi-spectroscopic and molecular modeling approaches. Spectrochim. Acta A 88, 177–191 (2012)
Huang, X., Xiong, P., Xiong, C., Cai, Y., Wei, A., Wang, J., Liang, X., Ruan, J.: In vitro and in vivo antitumor activity of Macrothelypteris torresiana and its acute/subacute oral toxicity. Phytomedicine 17, 930–934 (2010)
Wang, T., Xiang, B.R., Li, Y., Chen, C.Y., Zhou, X.H., Wang, Z.M., Dong, Y., Wan, Y., Fang, H.S.: Studies on the binding of a carditionic agent to human serum albumin by two-dimensional correlation fluorescence spectroscopy and molecular modeling. J. Mol. Struct. 921, 188–198 (2009)
Anzenbacher, P., Zanger, U.M.: Metabolism of drugs and other xenobiotics. Wiley Online Library, New York (2012)
Papadopoulou, A., Green, R.J., Frazier, R.A.: Interaction of flavonoids with bovine serum albumin: a fluorescence quenching study. J. Agr. Food Chem. 53, 158–163 (2005)
Shi, S., Zhang, Y., Xiong, X., Huang, K., Chen, X., Peng, M.: The influence of flavonoids on the binding of pantoprazole to bovine serum albumin by spectroscopic methods: with the viewpoint of food/drug interference. Food Chem. 135, 1083–1090 (2012)
Yang, Y., Hu, Q., Fan, Y., Shen, H.: Study on the binding of luteolin to bovine serum albumin. Spectrochim. Acta A 69, 432–436 (2008)
Gotardo, M.A., Tognolli, J.O., Pezza, H.R., Pezza, L.: Detection of propranolol in pharmaceutical formulations by diffuse reflectance spectroscopy. Spectrochim. Acta A 69, 1103–1109 (2008)
Li, H.M., Hu, Y.Z.: Spectroscopic investigation of inner filter effect by magnolol solutions. Spectrochim. Acta A 68, 1263–1268 (2007)
Shi, X., Li, X., Sun, Y., Wei, W., Yang, R., Zhang, H., Jin, Y.: In vitro study of the binding of taxifolin to bovine serum albumin and the influence of common ions on the binding. J. Solution Chem. 39, 482–494 (2010)
Madrakian, T., Afkhami, A., Mohammadnejad, M.: Simultaneous spectrofluorimetric determination of levodopa and propranolol in urine using feed-forward neural networks assisted by principal component analysis. Talanta 78, 1051–1055 (2009)
Lackowicz, J.R.: Principles of Fluorescence Spectroscopy, pp. 111–150. Plenum Press, New York (1983)
de Juan, A., Tauler, R.: Chemometrics applied to unravel multicomponent processes and mixtures revisiting latest trends in multivariate resolution. Anal. Chim. Acta 500, 195–210 (2003)
Zachariassen, C.B., Larsen, J., van den Berg, F., Bro, R., de Juan, A., Tauler, R.: Comparison of PARAFAC2 and MCR-ALS for resolution of an analytical liquid dilution system. Chemom. Intell. Lab. Syst. 83, 13–25 (2006)
Maeder, M.: Evolving factor analysis for the resolution of overlapping chromatographic peaks. Anal. Chem. 59, 527–530 (1987)
Golub, G.H., van Loan, C.F.: Matrix Computations, 2nd edn. John Hopkins University Press, Baltimore (1989)
Tauler, R., Smilde, A., Kowalski, B.: Selactivity, local rank, three-way data analysis and ambiguity in multivariant curve resolution. J. Chemom. 9, 31–58 (1995)
Lindahl, E., Hess, B., Van Der Spoel, D.: GROMACS 3.0: a package for molecular simulation and trajectory analysis. J. Mol. Model. 7, 306–317 (2001)
Díaz-Vergara, N., Piñeiro, Á.: Molecular dynamics study of triosephosphate isomerase from Trypanosoma cruzi in water/decane mixtures. J. Phys. Chem. B 112, 3529–3539 (2008)
Micaêlo, N.M., Soares, C.M.: Protein structure and dynamics in ionic liquids. Insights from molecular dynamics simulation studies. J. Phys. Chem. B 112, 2566–2572 (2008)
Todorova, N., Legge, F.S., Treutlein, H., Yarovsky, I.: Systematic comparison of empirical forcefields for molecular dynamic simulation of insulin. J. Phys. Chem. B 112, 11137–11146 (2008)
Schuttelkopf, A.W., Van Aalten, D.M.: PRODRG: a tool for high-throughput crystallography of protein–ligand complexes. Acta Crystallogr. D 60, 1355–1363 (2004)
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., Elbert, S.T., Gordon, M.S., Jensen, J.H., Koseki, S., Matsunaga, N., Nguyen, K.A., Su, S.: General atomic and molecular electronic structure system. J. Comput. Chem. 14, 1347–1363 (1993)
Hess, B., Bekker, H., Berendsen, H.J., Fraaije, J.G.: LINCS: a linear constraint solver for molecular simulations. J. Comput. Chem. 18, 1463–1472 (1997)
Darden, T., York, D., Pedersen, L.: Particle mesh Ewald: an N·log (N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993)
Berendsen, H.J., Postma, J.P.M., van Gunsteren, W.F., DiNola, A., Haak, J.: Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684–3690 (1984)
Danciulescu, C., Nick, B., Wortmann, F.J.: Structural stability of wild type and mutated α-keratin fragments: molecular dynamics and free energy calculations. Biomacromolecules 5, 2165–2175 (2004)
Chamani, J., Tafrishi, N., Momen-Heravi, M.: Characterization of the interaction between human lactoferrin and lomefloxacin at physiological condition: multi-spectroscopic and modeling description. J. Lumin. 130, 1160–1168 (2010)
Lakowicz, J.: Principles of Fluorescence Spectroscopy. Springer, New York (2006)
Qin, P., Liu, R., Pan, X., Fang, X., Mou, Y.: Impact of carbon chain length on binding of perfluoroalkyl acids to bovine serum albumin determined by spectroscopic methods. J. Agr. Food Chem. 58, 5561–5567 (2010)
Yue, Y., Chen, X., Qin, J., Yao, X.: A study of the binding of CI Direct Yellow 9 to human serum albumin using optical spectroscopy and molecular modeling. Dyes Pigments 79, 176–182 (2008)
Housaindokht, M.R., Rouhbakhsh Zaeri, Z., Bahrololoom, M., Chamani, J., Bozorgmehr, M.R.: Investigation of the behavior of HSA upon binding to amlodipine and propranolol: spectroscopic and molecular modeling approaches. Spectrochim. Acta A 85, 79–84 (2012)
Subramanyam, R., Gollapudi, A., Bonigala, P., Chinnaboina, M., Amooru, D.G.: Betulinic acid binding to human serum albumin: a study of protein conformation and binding affinity. J. Photoch. Photobio. B 94, 8–12 (2009)
Mohseni-Shahri, F.S., Housaindokht, M.R., Bozorgmehr, M.R., Moosavi-Movahedi, A.A.: The influence of theflavonoid quercetin on the interaction of propranolol with human serum albumin: experimental and theoretical approaches. J. Lumin. 154, 229–240 (2014)
Zhang, G., Que, Q., Pan, J., Guo, J.: Study of the interaction between icariin and human serum albumin by fluorescence spectroscopy. J. Mol. Struct. 881, 132–138 (2008)
Yang, L., Huo, D., Hou, C., Yang, M., Fa, H., Luo, X.: Interaction of monosulfonate tetraphenyl porphyrin (H2TPPS1) with plant-esterase: determination of the binding mechanism by spectroscopic methods. Spectrochim. Acta A 78, 1349–1355 (2011)
Abdollahpour, N., Asoodeh, A., Saberi, M.R., Chamani, J.: Separate and simultaneous binding effects of aspirin and amlodipine to human serum albumin based on fluorescence spectroscopic and molecular modeling characterizations: a mechanistic insight for determining usage drugs doses. J. Lumin. 131, 1885–1899 (2011)
He, L., Wang, X., Liu, B., Wang, J., Sun, Y., Gao, E., Xu, S.: Study on the interaction between promethazine hydrochloride and bovine serum albumin by fluorescence spectroscopy. J. Lumin. 131, 285–290 (2011)
Caruso, Í.P., Vilegas, W., de Souza, F.P., Fossey, M.A., Cornélio, M.L.: Binding of antioxidant flavone isovitexin to human serum albumin investigated by experimental and computational assays. J. Pharmaceut. Biomed. 98, 100–106 (2014)
Zhang, Y.Z., Zhou, B., Zhang, X.P., Huang, P., Li, C.H., Liu, Y.: Interaction of malachite green with bovine serum albumin: determination of the binding mechanism and binding site by spectroscopic methods. J. Hazard. Mater. 163, 1345–1352 (2009)
Rahmelow, K., Hubner, W.: Secondary structure determination of proteins in aqueous solution by infrared spectroscopy: a comparison of multivariate data analysis methods. Anal. Biochem. 241, 5–13 (1996)
Jung, C.: Insight into protein structure and protein–ligand recognition by Fourier transform infrared spectroscopy. J. Mol. Recognit. 13, 325–351 (2000)
Housaindokht, M.R., Bozorgmehr, M.R., Bahrololoom, M.: Analysis of ligand binding to proteins using molecular dynamics simulations. J. Theor. Biol. 254, 294–300 (2008)
Vassura, M., Margara, L., Di Lena, P., Medri, F., Fariselli, P., Casadio, R.: FT-COMAR: fault tolerant three-dimensional structure reconstruction from protein contact maps. Bioinformatics 24, 1313–1315 (2008)
Ding, F., Diao, J.X., Sun, Y., Sun, Y.: Bioevaluation of human serum albumin–hesperidin bioconjugate: insight into protein vector function and conformation. J. Agr. Food Chem. 60, 7218–7228 (2012)
Acknowledgments
The financial support for this work was provided by Research Council of Ferdowsi University of Mashhad (Research Project No. 3/24357-91/10/12).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohseni-Shahri, F.S., Housaindokht, M.R., Bozorgmehr, M.R. et al. Influence of Taxifolin on the Human Serum Albumin–Propranolol Interaction: Multiple Spectroscopic and Chemometrics Investigations and Molecular Dynamics Simulation. J Solution Chem 45, 265–285 (2016). https://doi.org/10.1007/s10953-016-0435-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-016-0435-4