Abstract
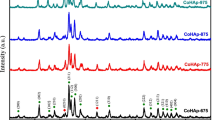
Complete carbonate substitution at A-sites (OH) of low-crystallinity hydroxyapatite with near stoichiometric composition (i.e. Ca/P ratio of 1.65) was achieved by heating in a dry carbon dioxide flow at 1173 K for 64 h. The carbonate content was analysed by thermogravimetry and infrared absorption spectrometry; the chemical composition was determined to be Ca9.9±0.1(PO4)6.00±0.1(CO3)0.9±0.1. The crystal structure and atomic configuration of the carbonate ion were determined by Rietveld refinement using X-ray powder diffraction data. This analysis revealed that the space group was monoclinic Pb with a = 0.9571(1), b = 1.9085(2), c = 0.68755(3) nm and γ = 119.847(7)°. The triangular planes of the carbonate ions were oriented parallel to the c- and a-axes, though there were two independent carbonate sites with occupancy factors of 0.56(1) and 0.34(1), where the triangles were oppositely rotated about the corresponding carbon atoms by 23° and −18°, respectively. The arrangement of the ions was disordered, which explains the lack of a thermal phase transition below 623 K.







Similar content being viewed by others
References
LeGeros RZ, Trautz OR, LeGeros JP, Klein E (1969) Cell Mol Life Sci 15:5
LeGeros RZ (1994) In: Brown PW, Constantz B (eds) Hydroxyapatite and related materials. CRC Press Inc., Boca Raton
Lee JC, Cho SB, Lee SJ, Rhee SH (2009) J Mater Sci 44:4531. doi:10.1007/s10853-009-3685-3s
Barralet JE, Fleming GJP, Campion C, Harris JJ, Wright AJ (2003) J Mater Sci 38:3979. doi:10.1023/A:1026258515285
Barinov SM, Rau JV, Cesaro SN, Durisin J, Fadeeva IV, Ferro D, Medvecky L, Trionfetti G (2006) J Mater Sci Mater Med 17:597
Komlev VS, Fadeeva IV, Gurin AN, Kovaleva ES, Smirnov VV, Gurin NA, Barinov SM (2009) Inorg Mater 45:329
Ikoma T, Kubo Y, Yamazaki A, Akao M, Tanaka J (2001) Key Eng Mater 192-5:191
Ito A (1988) Crystal growth and structure of apatitic phosphates. PhD Thesis, Waseda University
Suetsugu Y, Takahashi Y, Okamura FP, Tanaka J (2000) J Solid State Chem 155:292
Elliott JC (1980) J Appl Crystallogr 13:618
Fleet ME, Liu X (2003) J Solid State Chem 174:412
Fleet ME, Liu X (2005) Biomaterials 26:7548
Elliott JC (1994) Structure and chemistry of the apatites and other calcium orthophosphates. Elsevier Press, Amsterdam
Trombe JC (1973) Ann Chim (Paris) 14th Series 8:251
Bonel G (1972) Ann Chim (Paris) 14th Series 7:65
Bonel G (1972) Ann Chim (Paris) 14th Series 7:127
Ikoma T, Yamazaki A, Nakamura S, Akao M (1999) J Solid State Chem 144:272
Izumi F, Ikeda T (2000) Mater Sci Forum 321–324:198
Alberius-Henning P, Adolfsson E, Grins J, Fitch A (2001) J Mater Sci 36:663. doi:10.1023/A:1004876622105s
Rey C, Collins B, Goehl T, Dickson IR, Glimcher MJ (1989) Calcif Tissue Int 45:157
Rey C, Renugopalakrishnan V, Collins B, Glimcher MJ (1991) Calcif Tissue Int 49:251
Rey C, Shimizu M, Collins B, Glimcher MJ (1991) Calcif Tissue Int 49:259
Peroos S, Du Z, Henriette de Leeuw N (2006) Biomaterials 27:2150
Astala R, Stott MJ (2005) Chem Mater 17:4125
Elliott JC (1973) Science 180:1055
Kay MI, Young RA, Posner AS (1964) Nature 204:1050
Young RA, Bartlett ML, Spooner S, Mackie PE (1981) J Biol Phys 9:1
Suetsugu Y, Shimoya I, Tanaka J (1998) J Am Ceram Soc 81:746
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tonegawa, T., Ikoma, T., Yoshioka, T. et al. Crystal structure refinement of A-type carbonate apatite by X-ray powder diffraction. J Mater Sci 45, 2419–2426 (2010). https://doi.org/10.1007/s10853-010-4209-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-010-4209-x