Abstract
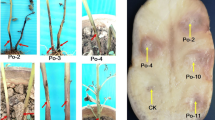
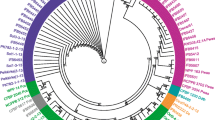
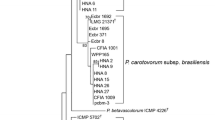
Pectolytic bacteria were isolated from potato tubers and stems showing tuber soft rot and blackleg symptoms. Approximately half (52 %) of the isolates could grow at both 27 and 37 °C while another half (48 %) failed to grow at 37 °C. All isolates could be amplified with primers specific to the pectate lyase (pel) gene. Carbon utilisation profiles could not conclusively identify these isolates. PCR amplification using primers specific for Pectobacterium carotovorum subsp. brasiliensis was positive for all isolates that grew at 37 °C. However, the group that did not grow at 37 °C failed to amplify with P. atrosepticum specific primers. To characterise this group of isolates, the intergenic transcribed spacer region (ITS) was amplified and PCR products digested with two restriction enzymes (RsaI and CfoI) to generate ITS-PCR-RFLP profiles. The profiles of these new isolates were compared to those of the type strains of other pectolytic bacteria. Profiles of five of the selected atypical strains generated with the enzyme CfoI appeared to be most similar to those of P. wasabiae type strain. Phylogenetic analysis using concatenated partial gene sequences of housekeeping genes mdh and gapA clustered these isolates together with those of P. wasabiae reference strains thus confirming their identity. These strains were virulent on potato tubers and stems but did not elicit hypersensitive response on tobacco plants. This is the first report of P. wasabiae causing soft rot and blackleg of potatoes in South Africa.





Similar content being viewed by others
References
Baghaee-Ravari, S., Rahimian, H., Shams-Bakhsh, M., Lopez-Solanilla, E., Antunez-Lamaz, M., & Rodriguez-Penzuela, P. (2011). Characterization of Pectobacterium species from Iran using biochemical and molecular methods. European Journal of Plant Pathology, 129, 413–425.
Charkowski, A., Blanco, C., Condemine, G., Expert, D., Franza, T., Hayes, C. et al. (2012) The role of secretion systems and small molecules in soft-rot Enterobacteriaceae pathogenicity. Annual Review of Phytopathology, 50, 425–449.
Clarke, R. C., Cai, R., Studholme, D. J., Guttman, D. S., & Vinatzer, B. A. (2010). Pseudomonas syringae strains naturally lacking the classical P. syringae hrp/hrc locus are common leaf colonizers equipped with an atypical Type III Secretion System. Molecular Plant Microbe Interaction, 23(2), 198–210.
Duarte, V., de Boer, S. H., Ward, T. L., & de Oliveora, A. M. R. (2004). Characterisation of atypical Erwinia carotovora subsp. atroseptica causing blackleg of potato in Brazil. Journal of Applied Microbiology, 96, 535–545.
Gardan, L., Gouy, C., Christen, R., & Samson, R. (2003). Elevation of three subsp. of Pectobacterium to subsp. level: Pectobacterium atrosepticum sp. nov., Pectobacterium betavasculorum sp. nov. and Pectobacterium wasabiaes p. nov. International Journal of Systematic and Evolutionary Microbiology, 53, 381–391.
Glasner, J. D., Marquez-Villavicencio, M., Kim, H. S., Jahn, C. E., Ma, B., Biehl, B. S., et al. (2008). Niche-specificity and the variable fraction of the Pectobacterium pan genome. Molecular Plant-Microbe Interactions, 21, 1549–1560.
Goto, M., & Matsumoto, K. (1987). Erwinia carotovora subsp. wasabiae subsp. nov. isolated from diseased rhizomes and fibrous roots of Japanese horseradish. International Journal of Systematic Bacteriology, 37, 130–135.
Hauben, L., Moore, E. R., Vauterin, L., Steenackers, M., Mergaert, J., Verdonck, L., et al. (1998). Phylogenetic position of phytopathogens within the Enterobacteriaceae. Systematic and Applied Microbiology, 21, 384–397.
Hyman, L. J., Sulivan, L., Toth, I. K., & Pérombelon, M. C. M. (2001). Modified crystal violet pectate medium (CVP) based on a new polypectate source (Slendid) for the detection and isolation of soft rot erwinias. Potato Research, 44, 265–270.
Kim, H. S., Ma, B., Perna, N. T., & Charkowski, A. O. (2009). Phylogeny and virulence of naturally occurring type III secretion system-deficient Pectobacterium strains. Applied and Environmental Microbiology, 75, 4539–4549.
Ma, B., Hibbing, M. E., Kim, H. S., Reedy, R. M., Yedidia, L., Breuer, J., et al. (2007). Host range and molecular phylogenies of the soft rot enterobacterial genera Pectobacterium and Dickeya. Phytopathology, 97, 1150–1163.
Marquez-Villavicencio, M. D. P., Groves, R. L., & Charkowski, A. O. (2011). Soft rot disease severity is affected by potato physiology and Pectobacterium taxa. Plant Disease, 95, 232–241.
Pérombelon, M. C. M., & Kelman, A. (1980). Ecology of soft rot erwinias. Annual Review of Phytopathology, 18, 361–387.
Pitman, A. R., Wright, P. J., Gailbraith, M. D., & Harrow, S. A. (2008). Biochemical and genetic diversity of pectolytic enterobacteria causing soft rot disease of potatoes in New Zealand. Australasian Plant Pathology, 37, 559–568.
Pitman, A. R., Harrow, S. A., & Visnovsky, S. B. (2010). Genetic characterisation of Pectobacterium wasabiae causing soft rot disease of potato in New Zealand. European Journal of Plant Pathology, 126, 423–435.
Sambrook, J., & Rusell, D. (2001). Molecular cloning: A laboratory manual (3rd ed.). Plainview: Cold Spring Harbor Lab Press.
Samson, R., Legendre, J. B., Christen, R., Fishcer-Le Saux, M., Achouak, W., & Gardan, L. (2005). Transfer of Pectobacterium chrysanthemi (Burkholder et al., 1953) Brenner et al. 1973 and chrysanthemi comb. Nov. and Dickeya paradisiaca comb.nov.and delineation of four novel species, Dickeya dadantii sp. nov., Dickeya dianthicola sp. nov. Dickeya dieffenbachiae sp. nov.and Dickeya zeae sp. nov. International Systematics Evolutionary Microbiology, 55, 1415–1427.
Serfontein, S., Logan, C., Swanepoel, A. E., Boelem, B. H., & Theron, D. J. (1991). A potato wilt disease in South Africa caused by Erwinia carotovora subspecies carotovora and E. chrysanthemi. Plant Pathology, 40, 382–386.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Masatoshi, N., & Kumar, S. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony. Methods Molecular Biology and Evolution, 28, 2731–2739.
Toth, I. K., Bertheau, Y., Hyman, L. J., Laplaze, L., López, M. M., McNicol, J., et al. (1999). Evaluation of phenotypic and molecular typing techniques for determining diversity in Erwinia carotovora subspp. atroseptica. Journal of Applied Microbiology, 87, 770–781.
Toth, I. K., Avrora, A. O., & Hyman, L. (2001). Rapid identification and differentiation of the soft rot erwinias by 16S–23S intergenic transcribed spacer PCR and restriction enzyme fragment polymorphism analyses. Applied and Environmental Microbiology, 67, 4070–4076.
Toth, I. K., van der Wolf, J. M., Saddler, G., Lojkowska, E., Hélias, E., Pirhonen, M., et al. (2011). Dickeya species: an emerging problem for potato production in Europe. Plant Pathology, 60, 385–399.
Tsror, L., Erlich, O., Lebiush, S., Hazanovsky, M., Zig, U., Slawiak, M., et al. (2009). Assessment of recent outbreaks of Dickeya sp. (syn. Erwinia chrysanthemi) slow wilt in potato crops in Israel. European Journal of Plant Pathology, 123, 311–320.
van der Merwe, J. J., Coutinho, T. A., Korsten, L., & van der Waals, J. E. (2010). Pectobacterium carotovorum subsp. brasiliensis causing blackleg on potatoes in South Africa. European Journal of Plant Pathology, 126, 175–185.
Acknowledgments
This work was funded by the National Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moleleki, L.N., Onkendi, E.M., Mongae, A. et al. Characterisation of pectobacterium wasabiae causing blackleg and soft rot diseases in South Africa. Eur J Plant Pathol 135, 279–288 (2013). https://doi.org/10.1007/s10658-012-0084-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-012-0084-4