Abstract
Background
Pancreatic cysts are common incidental findings with malignant potential, raising diagnostic and treatment dilemmas.
Aims
To determine the added value of KRAS and GNAS mutation analysis on cyst classification and decision making.
Methods
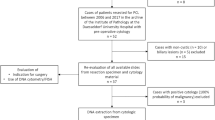
We analyzed 52 frozen samples of pancreatic cystic fluid obtained by EUS-FNA between 2008 and 2014. In addition to cytology and CEA, mutations of GNAS (exons 8 and 9) and KRAS (exons 2 and 3) genes were analyzed using Sanger sequencing.
Results
There were 52 patients, 67% females, with a mean age of 59 ± 15 years (29–91). Cysts were classified as mucinous in 21 patients (40%) (14 low-risk, seven malignant) and non-mucinous in 31 patients (60%). After EUS-FNA, 11 patients had surgery, six had chemotherapy or palliation, one had endoscopic drainage, and 34 are on follow-up after a mean of 57 months. KRAS mutation was detected in nine and GNAS in two samples. Patients harboring cysts with KRAS mutations were older (p = 0.01), cysts were more commonly mucinous (p = 0.001) and malignant (p = 0.01). KRAS mutations were present in both low-risk and malignant mucinous lesions. For identifying mucinous lesions, CEA > 192 ng/mL performed better (AUC ROC = 93%), whereas for malignant/high-risk mucinous lesions, EUS imaging had the best accuracy (AUC ROC = 88%). After molecular analysis, a modification in cyst classification occurred in ten patients, but was correct in only two, a pseudocyst re-classified as IPMN and a malignant cyst as a non-mucinous cyst.
Conclusions
In this cohort of patients with pancreatic cysts, KRAS and GNAS mutations had no significant diagnostic benefit in comparison with conventional testing.

Similar content being viewed by others
References
Lennon AM, Wolfgang CL, Canto MI, et al. The early detection of pancreatic cancer: What will it take to diagnose and treat curable pancreatic neoplasia? Cancer Res. 2014;74:3381–3389. https://doi.org/10.1158/0008-5472.CAN-14-0734.
Kromrey M, Bülow R, Hübner J, et al. Prospective study on the incidence, prevalence and 5-year pancreatic-related mortality of pancreatic cysts in a population-based study. Gut. 2018;67:1–8. https://doi.org/10.1136/gutjnl-2016-313127.
Palmucci S, Cappello G, Trombatore C, et al. Cystic pancreatic neoplasms: diagnosis and management emphasizing their imaging features. Eur Rev Med Pharmacol Sci.. 2014;18:1259–1268.
Brugge WR, Lewandrowski K, Lee-Lewandrowski E, et al. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology. 2004;126:1330–1336. https://doi.org/10.1053/j.gastro.2004.02.013.
Thornton GD, Mcphail MJW, Nayagam S, Hewitt MJ, Vlavianos P, Monahan KJ. Pancreatology endoscopic ultrasound guided fine needle aspiration for the diagnosis of pancreatic cystic neoplasms: a meta-analysis. Pancreatology. 2013;13:48–57. https://doi.org/10.1016/j.pan.2012.11.313.
Fasge VRM, Chandrasekhara V, Acosta RD, et al. The role of endoscopy in the diagnosis and treatment of cystic pancreatic neoplasms. Gastrointest Endosc. 2016;84:1–9. https://doi.org/10.1016/j.gie.2016.04.014.
Wang QX, Xiao J, Orange M, et al. EUS-guided FNA for diagnosis of pancreatic cystic lesions: a meta-analysis. Cell Physiol Biochem. 2015;36:1197–1209. https://doi.org/10.1159/000430290.
Rosenbaum MW, Jones M, Dudley JC, et al. Next-generation sequencing adds value to the preoperative diagnosis of pancreatic cysts. Cancer Cytopathol. 2017;125:41–47. https://doi.org/10.1002/cncy.21775.
Al-haddad M, Dewitt J, Sherman S, et al. Performance characteristics of molecular (DNA) analysis for the diagnosis of mucinous pancreatic cysts. Gastrointest Endosc. 2014;79:79–87. https://doi.org/10.1016/j.gie.2013.05.026.
Gillis A, Cipollone I, Cousins G, Conlon K. Does EUS-FNA molecular analysis carry additional value when compared to cytology in the diagnosis of pancreatic cystic neoplasm? A systematic review. HPB (Oxford). 2015;17:377–386. https://doi.org/10.1111/hpb.12364.
Singhi AD, Nikiforova MN, McGrath K. DNA testing of pancreatic cyst fluid: is it ready for prime time? Lancet Gastroenterol Hepatol. 2017;2:63–72. https://doi.org/10.1016/S2468-1253(16)30084-X.
Pitman MB, Centeno BA, Ali SZ, et al. Standardized terminology and nomenclature for pancreatobiliary cytology: the papanicolaou society of cytopathology guidelines. Diag Cytopathol. 2014;42:338–350. https://doi.org/10.1002/dc.23092.
Tanaka M, Chari S, Adsay V, et al. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms. Pancreatology. 2006;6:17–32. https://doi.org/10.1159/000090023.
Tanaka M, Fernández-del Castillo C, Adsay V, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12:183–197. https://doi.org/10.1016/j.pan.2012.04.004.
Springer S, Wang Y, Dal Molin M, et al. A combination of molecular markers and clinical features improve the classification of pancreatic cysts. Gastroenterology. 2015;149:1501–1510. https://doi.org/10.1053/j.gastro.2015.07.041.
Hata T, Molin MD, Hong S, et al. Predicting the grade of dysplasia of pancreatic cystic neoplasms using cyst fluid DNA methylation markers. Clin Cancer Res. 2017;23:3935–3944. https://doi.org/10.1158/1078-0432.CCR-16-2244.
Singhi AD, Nikiforova MN, Fasanella KE, et al. Preoperative GNAS and KRAS testing in the diagnosis of pancreatic mucinous cysts. Clin Cancer Res. 2014;20:4381–4389. https://doi.org/10.1158/1078-0432.CCR-14-0513.
Singhi AD, Mcgrath K, Brand RE, et al. Preoperative next-generation sequencing of pancreatic cyst fluid is highly accurate in cyst classification and detection of advanced neoplasia. Gut. 2017. https://doi.org/10.1136/gutjnl-2016-313586.
Jones M, Zheng Z, Wang J, et al. Impact of next-generation sequencing on the clinical diagnosis of pancreatic cysts. Gastrointest Endosc. 2016;83:140–148. https://doi.org/10.1016/j.gie.2015.06.047.
Levy P, Ohtsuka T, Salvia R, et al. Revisions of International Consensus Fukuoka Guidelines for the Management of IPMN of the Pancreas. International Association of Pancreatology and European Pancreatic Club. Pancreatology. 2017;17:738–753. https://doi.org/10.1016/j.pan.2017.07.007.
Khalid A, Zahid M, Finkelstein SD, Leblanc JK. Pancreatic cyst fluid DNA analysis in evaluating pancreatic cysts: a report of the PANDA study. Gastrointest Endosc. 2009;69:1095–1102. https://doi.org/10.1016/j.gie.2008.07.033.
Scheiman JM, Hwang JH, Moayyedi P. American gastroenterological association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015;148:824–848. https://doi.org/10.1053/j.gastro.2015.01.014.
Acknowledgments
We would like to thank Susan Marum, MD for the English language review of this manuscript.
Funding
The current study was supported by a Research Grant from Clube Português do Pâncreas, Bolsa, 2015, that had no role in any stage of the study, from design to submission for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Faias, S., Duarte, M., Albuquerque, C. et al. Clinical Impact of KRAS and GNAS Analysis Added to CEA and Cytology in Pancreatic Cystic Fluid Obtained by EUS-FNA. Dig Dis Sci 63, 2351–2361 (2018). https://doi.org/10.1007/s10620-018-5128-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5128-y