Abstract
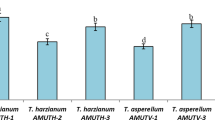
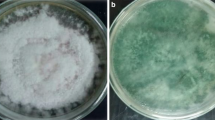
Two biological control practices are the use of suppressive growing media and the application of biological control agents (BCAs). The goals of this study were: (i) to screen 584 potential BCAs obtained from Fusarium wilt (FW) suppressive growing media; (ii) to evaluate in greenhouse conditions selected BCAs in three growing media with different degrees of suppressiveness of tomato FW. Two isolates selected after screening were identified as Fusarium solani (305) and Streptomyces sp. (A19). Results showed that tomato FW was reduced and total production was improved when both BCAs were applied to a conducive medium (coir fiber). In highly suppressive growing medium (grape marc compost), A19 and 305 inoculations did not improve suppressiveness. In moderately suppressive growing medium (cork compost), only A19 improved this compost to natural grape marc compost suppressiveness level. Therefore, compost suppressiveness of tomato FW depended on the nature of the compost and on the isolates applied.



Similar content being viewed by others
References
Abad M, Noguera P, Puchades R, Maquieira A, Noguera V (2002) Physico-chemical and chemical properties of some coconut coir dust for use as a peat substitute for containerised ornamental plants. Bioresour Technol 82:241–245
Alabouvette C, Olivain C, Steinberg C (2006) Biological control of plant diseases: the European situation. Eur J Plant Pathol 114:329–341
Alabouvette C, Olivain C, Migheli Q, Steinberg C (2009) Microbiological control of soil-borne phytopathogenic fungi with special emphasis on wilt-inducing Fusarium oxysporum. New Phytol 184:529–544
Anonymous (2010) Avances de superficies y producciones agrícolas. Ministerio de Medio Ambiente y Medio Rural y Marino, Spain, p 28
Avilés M, Borrero C, Trillas MI (2011) Review on compost as an inducer of disease suppression in plants grown in soilless culture. In: Special Issue Compost III—dynamic plant, dynamic soil. Global Science Books. 5 (Special Issue 2), pp 1–11
Bonanomi G, Antignani V, Capodilupo M, Scala F (2010) Identifying the characteristics of organic soil amendments that suppress soilborne plant diseases. Soil Biol Biochem 42:136–144
Borrero C, Trillas MI, Ordovás J, Tello JC, Avilés M (2004) Predictive factors for the suppression of Fusarium wilt of tomato in plant growth media. Phytopathology 94:1094–1101
Borrero C, Infantes MJ, González E, Tello JC, Avilés M (2005) Relation between suppressiveness to tomato Fusarium wilt and microbial populations in different growth media. Acta Hort 697:425–430
Borrero C, Ordovás J, Trillas MI, Avilés M (2006) Tomato Fusarium wilt suppressiveness. The relationship between the organic plant growth media and their microbial communities as characterised by Biolog®. Soil Biol Biochem 38:1631–1637
Borrero C, Trillas MI, Avilés M (2009) Carnation Fusarium wilt suppression in four composts. Eur J Plant Pathol 123:425–433
Brimmer T, Boland G (2003) A review of the non-target effects of fungi used to biologically control plant diseases. Agric Ecosyst Environ 100:3–16
Campbell CL, Madden LV (1990) Introduction to plant disease epidemiology. Wiley, New York, USA
Carmona E, Avilés M, Domínguez I, Moreno MT, Pajuelo P, Ordovás J (2004) Exploitation of composted agricultural wastes as growing media. In: Bernal MP, Moral R, Clemente R, Paredes C (ed) Sustainable organic waste management for environmental protection and food safety, vol I. Ramiran 2004. Proceedings of the 11th international conference of the FAO ESCORENA network on the recycling of agricultural residues in agriculture, Murcia, Spain, pp 141–144
Cassago A, Panepucci RA, Tordella-Baião AM, Henrique-Silva F (2002) Cellophane based mini-prep method for DNA extraction from the filamentous fungus Trichoderma ressei. BMC Microbiol 2:14–17
Castaño R, Borrero C, Avilés M (2011) Organic matter fractions by SP-MAS 13C NMR and microbial communities involved in the suppression of Fusarium wilt in organic growth media. Biol Control 58:286–293
Chen W, Hoitink HAJ, Schmitthenner AF, Tuovinen OH (1988) The role of microbial activity in suppression of damping-off caused by Pythium ultimum. Phytopathology 78:314–322
Chung YR, Hoitink HAJ (1990) Interactions between thermophilic fungi and Trichoderma hamatum in suppression of Rhizoctonia damping-off in a bark compost-amended container medium. Phytopathology 80:73–77
Coombs JT, Michelsen PP, Franco MM (2004) Evaluation of endophytic actinobacteria as antagonists of Gaeumannomyces graminis var. tritici in wheat. Biol Control 29:359–366
Cotxarrera L, Trillas-Gay MI, Steinberg C, Alabouvette C (2002) Use of sewage sludge compost and Trichoderma asperellum isolates to suppress Fusarium wilt of tomato. Soil Biol Biochem 34:467–476
De Boer W, Verheggen P, Klein Gunnewiek PJA, Kowalchuk GA, van Veen JA (2003) Microbial community composition affects soil fungistasis. Appl Environ Microbiol 69:835–844
Dekker J (1981) Resistance to fungicides in plant pathogens: abstracts of papers. Neth J Plant Pathol 87:233–255
Dhingra O, Sinclair J (1995) Basic plant pathology methods, 2nd edn. Lewis Publisher, Boca Raton, USA
Doumbou CL, Hamby Salove MK, Crawford DL, Beaulieu C (2002) Actinomycetes, promising tools to control plant diseases and to promote plant growth. Phytoprotection 82:85–102
El-Tarabily KA, Sivasithamparam K (2006) Non-streptomycete actinomycetes as biocontrol agents of soil-borne fungal plant pathogens and as plant growth promoters. Soil Biol Biochem 38:1505–1520
El-Tarabily KA, Soliman MH, Nassar AH, Al-Hassani HA, Sivasithamparam K, McKenna F, Hardy GESJ (2000) Biological control of Sclerotinia minor using a chitinolytic bacterium and actinomycetes. Plant Pathol 49:573–583
Garbeva P, Gera Hol WH, Termorshuizen AJ, Kowalchuk GA, de Boer W (2011) Fungistasis and general soil biostasis—a new synthesis. Soil Biol Biochem 43:469–477
Geiser DM, Jiménez-Gasco MM, Kang S, Makalowska I, Veeraraghavan N, Ward TJ, Zhang N, Kuldau GA, O’Donnell K (2004) Fusarium-ID v. 1.0: a DNA sequence database for identifying Fusarium. Eur J Plant Pathol 110:473–479
Hartman A, Schmid M, van Tuinen D, Berg G (2009) Plant-driven selection of microbes. Plant Soil 321:235–257
Kepner JR, Pratt JR (1994) Use of fluorochromes for direct enumeration of total bacterial in environmental samples: past and present. Microbiol Mol Biol Rev 58:603–615
Kim BS, Hwang B (2007) Microbial fungicides in the control of plant diseases. J Phytopathol 155:641–653
Komada H (1975) Development of a selective medium for quantitative isolation of Fusarium oxysporum from natural soil. Rev Plant Prot Res 8:114–125
LaMontagne MG, Schimel JP, Holden PA (2003) Comparison of subsurface and surface soil bacterial communities in California grassland as assessed by terminal restriction fragment length polymorphisms of PCR-amplified 16S rRNA genes. Microb Ecol 46:216–227
Larkin RP, Fravel DR (1998) Efficacy of various fungal and bacterial biocontrol organisms for control of Fusarium wilt of tomato. Plant Dis 82:1022–1028
Larkin RP, Fravel DR (1999) Mechanisms of action and dose-response relationships governing biological control of Fusarium wilt of tomato by nonpathogenic Fusarium spp. Phytopathology 89:1152–1161
Larkin RP, Fravel DR (2002) Effects of varying environmental conditions on biological control of Fusarium wilt of tomato nonpathogenic Fusarium spp. Phytopathology 92:1160–1166
Litterick AM, Harrier L, Wallace P, Watson CA, Wood M (2004) The role of uncomposted materials, composts, manures, and composts extracts in reducing pest and disease incidence and severity in sustainable temperate agricultural and horticultural crop production—a review. Crit Rev Plant Sci 23:453–479
Mandeel Q, Baker R (1991) Mechanisms involved in biological control of Fusarium wilt of cucumber with strains of nonpathogenic Fusarium oxysporum. Phytopathology 81:462–469
Nelson EB, Kuter GA, Hoitink HAJ (1983) Effects of fungal antagonists and compost age on suppression of Rhizoctonia damping-off in container media amended with composted hardwood bark. Phytopathology 73:1457–1462
Noble R, Coventry E (2005) Suppression of soil-borne plant diseases with composts: a review. Biocontrol Sci Technol 15:3–20
Paternoster T, Défago G, Duffy B, Gessler C, Pertot I (2010) Selection of a biocontrol agent based on a potential mechanism of action: degradation of nicotinic acid, a growth factor essential for Erwinia amylovora. Int Microbiol 13:195–206
Prabavathy VR, Mathivanan N, Murugesan K (2006) Control of blast and sheath blight diseases of rice using antifungal metabolites produced by Streptomyces sp. PM5. Biol Control 39:313–319
Punja ZK, Utkhede RS (2003) Using fungi and yeasts to manage vegetable crop diseases. Trends Biotechnol 21:400–407
Quecine MC, Araujo WL, Marcon J, Gai CS, Azevedo JL, Pizzirani-Kleiner AA (2008) Chitinolytic activity of endophytic Streptomyces and potential for biocontrol. Lett Appl Microbiol 47:486–491
Roy AK, Mandal NL, Singh AN (2009) Screening of maize rhizobacteria against aflatoxigenic Aspergillus flavus strains in relation to siderophore and HCN production. Indian Phytopathol 62:440–444
Sant D, Casanova E, Segarra G, Avilés M, Reis M, Trillas MI (2010) Effect of Trichoderma asperellum strain T34 on Fusarium wilt and water usage in carnation grown on compost-based growth medium. Biol Control 53:291–296
Singh PP, Shin YC, Park CS, Chung YR (1999) Biological control of Fusarium wilt of cucumber by chitinolytic bacteria. Phytopathology 89:92–99
Taurian T, Anzuay MS, Angelini JG, Tonelli ML, Ludueña L, Pena D, Ibáñez F, Fabra A (2010) Phosphate-solubilizing peanut associated bacteria: screening for plant growth-promoting activities. Plant Soil 329:421–431
Tondje PR, Roberts DP, Bon MC, Widmer T, Samuels GJ, Ismaiel A, Begoude AD, Tchana T, Nyemb-Tshomb E, Ndoumbe-Nkeng M, Bateman R, Fontem D, Hebbar KP (2007) Isolation and identification of mycoparasitic isolates of Trichoderma asperellum with potential for suppression of black pod disease of cacao in Cameroon. Biol Control 43:202–212
Trillas MI, Avilés M, Ordovás J, Bello A, Tello JC (2002) Using compost as a methyl bromide alternative. BioCycle 43:64–68
Trillas MI, Casanova E, Cotxarrera L, Ordovás J, Borrero C, Avilés M (2006) Composts from agricultural waste and the Trichoderma asperellum strain T-34 suppress Rhizoctonia solani in cucumber seedlings. Biol Control 39:32–38
Tuitert G, Szczech M, Bollen GJ (1998) Suppression of Rhizoctonia solani in potting mixtures amended with compost made from organic household waste. Phytopathology 88:764–773
Weller DM, Raaijmakers JM, McSpadden Gardener BB, Thomashow LS (2002) Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu Rev Phytopathol 40:309–348
Xia K, Kinkel LL, Samac DA (2002) Biological control of Phytophthora root rot on alfalfa and soybean with Streptomyces. Biol Control 23:285–295
Zheng Y, Xue QY, Xu LL, Xu Q, Lu S, Gu S, Guo JH (2011) A screening strategy of fungal biocontrol agents towards Verticillium wilt of cotton. Biol Control 56:209–216
Acknowledgments
This research was supported by grants from Ministerio de Educación y Ciencia (AGL2005-08137-C03-02), Consejería de Innovación, Ciencia y Empresa de la Junta de Andalucía (P06-AGR-02313) and Ministerio de Ciencia e Innovación (AGL2008-05414-C03-01) of Spain. We thank M.L. Castillo, S. Castillo and J. Rojo for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Monica Höfte
Rights and permissions
About this article
Cite this article
Castaño, R., Borrero, C., Trillas, M.I. et al. Selection of biological control agents against tomato Fusarium wilt and evaluation in greenhouse conditions of two selected agents in three growing media. BioControl 58, 105–116 (2013). https://doi.org/10.1007/s10526-012-9465-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-012-9465-z