Abstract
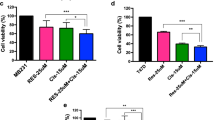
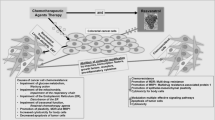
The objective of this study was to investigate, whether the naturally occurring polyphenol resveratrol (Res) enhances the anti-tumor activities of the chemotherapeutic agent oxaliplatin (Ox) in a cell culture model of colorectal cancer, also with regard to a possible inflammatory response and cytotoxic side-effects. Res and Ox in combination synergistically inhibit cell growth of Caco-2 cells, which seems to be due to the induction of different modes of cell death and further leads to an altered cytokine profile of cocultured macrophages. Moreover, combinatorial treatment does not affect non-transformed cells as severe cytotoxicity is not detected in human foreskin fibroblasts and platelets.







Similar content being viewed by others
Abbreviations
- BrdU:
-
Bromodeoxyuridine
- CI:
-
Combination index
- DMEM:
-
Dulbecco’s modified-Eagle’s medium
- DMSO:
-
Dimethylsulfoxide
- EDTA:
-
Ethylendiaminetetraacetic acid
- FCS:
-
Fetal calf serum
- FITC:
-
Fluorescein isothiocyanate
- 5-FU:
-
5-Fluorouracil
- HFF:
-
Human foreskin fibroblasts
- IAPs:
-
Inhibitor of apoptosis proteins
- IC50 :
-
Half maximal inhibitory concentration
- IL:
-
Interleukine
- LDH:
-
Lactate dehydrogenase
- Ox:
-
Oxaliplatin
- PARP:
-
Poly[ADP-ribose] polymerase
- PI:
-
Propidium iodide
- Res:
-
Resveratrol
- TAMs:
-
Tumor associated macrophages
- TNF-α:
-
Tumor necrosis factor-α
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62:10–29
Alcindor T, Beauger N (2011) Oxaliplatin: a review in the era of molecularly targeted therapy. Curr Oncol 18:18–25
Graham MA, Lockwood GF, Greenslade D, Brienza S, Bayssas M, Gamelin E (2000) Clinical pharmacokinetics of oxaliplatin: a critical review. Clin Cancer Res 6:1205–1218
Sarkar FH, Li Y (2006) Using chemopreventive agents to enhance the efficacy of cancer therapy. Cancer Res 66:3347–3350
Takaoka M (1939) Resveratrol, a new phenolic compound, from Veratrum grandiflorum. Nippon Kagaku Kaishi 60:1090–1100
Delmas D, Solary E, Latruffe N (2011) Resveratrol, a phytochemical inducer of multiple cell death pathways: apoptosis, autophagy and mitotic catastrophe. Curr Med Chem 18:1100–1121
Ulrich S, Wolter F, Stein JM (2005) Molecular mechanisms of the chemopreventive effects of resveratrol and its analogs in carcinogenesis. Mol Nutr Food Res 49:452–461
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239–257
Kroemer G, Martin SJ (2005) Caspase-independent cell death. Nat Med 11:725–730
Kroemer G, Galluzzi L, Brenner C (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87:99–163
Savill J, Dransfield I, Gregory C, Haslett C (2002) A blast from the past: clearance of apoptotic cells regulates immune responses. Nat Rev Immunol 2:965–975
Kono H, Rock KL (2008) How dying cells alert the immune system to danger. Nat Rev Immunol 8:279–289
Savill J, Fadok V (2000) Corpse clearance defines the meaning of cell death. Nature 407:784–788
Zitvogel L, Kepp O, Kroemer G (2010) Decoding cell death signals in inflammation and immunity. Cell 140:798–804
Basu S, Binder RJ, Suto R, Anderson KM, Srivastava PK (2000) Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-kappa B pathway. Int Immunol 12:1539–1546
Chou TC, Talalay P (1984) Quantitative analysis of dose–effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzym Regul 22:27–55
Carnesecchi S, Langley K, Exinger F, Gosse F, Raul F (2002) Geraniol, a component of plant essential oils, sensitizes human colonic cancer cells to 5-Fluorouracil treatment. J Pharmacol Exp Ther 301:625–630
Kaminski BM, Loitsch SM, Ochs MJ, Reuter KC, Steinhilber D, Stein J, Ulrich S (2010) Isothiocyanate sulforaphane inhibits protooncogenic ornithine decarboxylase activity in colorectal cancer cells via induction of the TGF-beta/Smad signaling pathway. Mol Nutr Food Res 54:1486–1496
Von Knethen AA, Brune B (2001) Delayed activation of PPARgamma by LPS and IFN-gamma attenuates the oxidative burst in macrophages. FASEB J 15:535–544
Weigert A, Tzieply N, von Knethen A, Johann AM, Schmidt H, Geisslinger G, Brune B (2007) Tumor cell apoptosis polarizes macrophages role of sphingosine-1-phosphate. Mol Biol Cell 18:3810–3819
Tamm I, Wang Y, Sausville E, Scudiero DA, Vigna N, Oltersdorf T, Reed JC (1998) IAP-family protein survivin inhibits caspase activity and apoptosis induced by Fas (CD95), Bax, caspases, and anticancer drugs. Cancer Res 58:5315–5320
Fujie Y, Yamamoto H, Ngan CY, Takagi A, Hayashi T, Suzuki R, Ezumi K, Takemasa I, Ikeda M, Sekimoto M, Matsuura N, Monden M (2005) Oxaliplatin, a potent inhibitor of survivin, enhances paclitaxel-induced apoptosis and mitotic catastrophe in colon cancer cells. Jpn J Clin Oncol 35:453–463
Khan Z, Khan N, Tiwari RP, Patro IK, Prasad GB, Bisen PS (2010) Down-regulation of survivin by oxaliplatin diminishes radioresistance of head and neck squamous carcinoma cells. Radiother Oncol 96:267–273
Eguchi Y, Shimizu S, Tsujimoto Y (1997) Intracellular ATP levels determine cell death fate by apoptosis or necrosis. Cancer Res 57:1835–1840
Wang XQ, Xiao AY, Sheline C, Hyrc K, Yang A, Goldberg MP, Choi DW, Yu SP (2003) Apoptotic insults impair Na+, K+-ATPase activity as a mechanism of neuronal death mediated by concurrent ATP deficiency and oxidant stress. J Cell Sci 116:2099–2110
Faragher RG, Burton DG, Majecha P, Fong NS, Davis T, Sheerin A, Ostler EL (2011) Resveratrol, but not dihydroresveratrol, induces premature senescence in primary human fibroblasts. Age (Dordr) 33:555–564
Capdevila J, Elez E, Peralta S, Macarulla T, Ramos FJ, Tabernero J (2008) Oxaliplatin-based chemotherapy in the management of colorectal cancer. Expert Rev Anticancer Ther 8:1223–1236
Rixe O, Ortuzar W, Alvarez M, Parker R, Reed E, Paull K, Fojo T (1996) Oxaliplatin, tetraplatin, cisplatin, and carboplatin: spectrum of activity in drug-resistant cell lines and in the cell lines of the National Cancer Institute’s Anticancer Drug Screen panel. Biochem Pharmacol 52:1855–1865
Smoliga JM, Baur JA, Hausenblas HA (2011) Resveratrol and health—a comprehensive review of human clinical trials. Mol Nutr Food Res 55:1129–1141
Vang O, Ahmad N, Baile CA, Baur JA, Brown K, Csiszar A, Das DK, Delmas D, Gottfried C, Lin HY, Ma QY, Mukhopadhyay P, Nalini N, Pezzuto JM, Richard T, Shukla Y, Surh YJ, Szekeres T, Szkudelski T, Walle T, Wu JM (2011) What is new for an old molecule? Systematic review and recommendations on the use of resveratrol. PLoS ONE 6:e19881
Kaminski BM, Steinhilber D, Stein JM, Ulrich S (2012) Phytochemicals resveratrol and sulforaphane as potential agents for enhancing the anti-tumor activities of conventional cancer therapies. Curr Pharm Biotechnol 13:137–146
Vinod BS, Maliekal TT, Anto RJ (2013) Phytochemicals as chemosensitizers: from molecular mechanism to clinical significance. Antioxid Redox Signal 18(11):1307–1348
El-Mowafy AM, El-Mesery ME, Salem HA, Al-Gayyar MM, Darweish MM (2010) Prominent chemopreventive and chemoenhancing effects for resveratrol: unraveling molecular targets and the role of C-reactive protein. Chemotherapy 56:60–65
Mao QQ, Bai Y, Lin YW, Zheng XY, Qin J, Yang K, Xie LP (2010) Resveratrol confers resistance against taxol via induction of cell cycle arrest in human cancer cell lines. Mol Nutr Food Res 54(11):1574–1584 (Epub ahead of print)
Harikumar KB, Kunnumakkara AB, Sethi G, Diagaradjane P, Anand P, Pandey MK, Gelovani J, Krishnan S, Guha S, Aggarwal BB (2010) Resveratrol, a multitargeted agent, can enhance antitumor activity of gemcitabine in vitro and in orthotopic mouse model of human pancreatic cancer. Int J Cancer 127:257–268
Colin D, Gimazane A, Lizard G, Izard JC, Solary E, Latruffe N, Delmas D (2009) Effects of resveratrol analogs on cell cycle progression, cell cycle associated proteins and 5fluoro-uracil sensitivity in human derived colon cancer cells. Int J Cancer 124:2780–2788
Jazirehi AR, Bonavida B (2004) Resveratrol modifies the expression of apoptotic regulatory proteins and sensitizes non-Hodgkin’s lymphoma and multiple myeloma cell lines to paclitaxel-induced apoptosis. Mol Cancer Ther 3:71–84
Chan JY, Phoo MS, Clement MV, Pervaiz S, Lee SC (2008) Resveratrol displays converse dose-related effects on 5-fluorouracil-evoked colon cancer cell apoptosis: the roles of caspase-6 and p53. Cancer Biol Ther 7:1305–1312
Kweon SH, Song JH, Kim TS (2010) Resveratrol-mediated reversal of doxorubicin resistance in acute myeloid leukemia cells via downregulation of MRP1 expression. Biochem Biophys Res Commun 395:104–110
Nabekura T, Kamiyama S, Kitagawa S (2005) Effects of dietary chemopreventive phytochemicals on P-glycoprotein function. Biochem Biophys Res Commun 327:866–870
Choi JS, Choi BC, Kang KW (2009) Effect of resveratrol on the pharmacokinetics of oral and intravenous nicardipine in rats: possible role of P-glycoprotein inhibition by resveratrol. Pharmazie 64:49–52
Frampton GA, Lazcano EA, Li H, Mohamad A, Demorrow S (2010) Resveratrol enhances the sensitivity of cholangiocarcinoma to chemotherapeutic agents. Lab Investig 90:1325–1338
LaCasse EC, Baird S, Korneluk RG, MacKenzie AE (1998) The inhibitors of apoptosis (IAPs) and their emerging role in cancer. Oncogene 17:3247–3259
Shin S, Sung BJ, Cho YS, Kim HJ, Ha NC, Hwang JI, Chung CW, Jung YK, Oh BH (2001) An anti-apoptotic protein human survivin is a direct inhibitor of caspase-3 and -7. Biochemistry 40:1117–1123
Altieri DC (2003) Validating survivin as a cancer therapeutic target. Nat Rev Cancer 3:46–54
Rodel F, Sprenger T, Kaina B, Liersch T, Rodel C, Fulda S, Hehlgans S (2012) Survivin as a prognostic/predictive marker and molecular target in cancer therapy. Curr Med Chem 19:3679–3688
Chao JI, Kuo PC, Hsu TS (2004) Down-regulation of survivin in nitric oxide-induced cell growth inhibition and apoptosis of the human lung carcinoma cells. J Biol Chem 279:20267–20276
Wall NR, O’Connor DS, Plescia J, Pommier Y, Altieri DC (2003) Suppression of survivin phosphorylation on Thr34 by flavopiridol enhances tumor cell apoptosis. Cancer Res 63:230–235
Rodel C, Haas J, Groth A, Grabenbauer GG, Sauer R, Rodel F (2003) Spontaneous and radiation-induced apoptosis in colorectal carcinoma cells with different intrinsic radiosensitivities: survivin as a radioresistance factor. Int J Radiat Oncol Biol Phys 55:1341–1347
Goyal L (2001) Cell death inhibition: keeping caspases in check. Cell 104:805–808
Igney FH, Krammer PH (2002) Death and anti-death: tumour resistance to apoptosis. Nat Rev Cancer 2:277–288
Tang HL, Yuen KL, Tang HM, Fung MC (2009) Reversibility of apoptosis in cancer cells. Br J Cancer 100:118–122
Ricci MS, Zong WX (2006) Chemotherapeutic approaches for targeting cell death pathways. Oncologist 11:342–357
Tesniere A, Schlemmer F, Boige V, Kepp O, Martins I, Ghiringhelli F, Aymeric L, Michaud M, Apetoh L, Barault L, Mendiboure J, Pignon JP, Jooste V, van Endert P, Ducreux M, Zitvogel L, Piard F, Kroemer G (2010) Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene 29:482–491
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331:1565–1570
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23:549–555
Lewis C, Murdoch C (2005) Macrophage responses to hypoxia: implications for tumor progression and anti-cancer therapies. Am J Pathol 167:627–635
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124:263–266
Crowther M, Brown NJ, Bishop ET, Lewis CE (2001) Microenvironmental influence on macrophage regulation of angiogenesis in wounds and malignant tumors. J Leukoc Biol 70:478–490
Bingle L, Brown NJ, Lewis CE (2002) The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 196:254–265
Mantovani A, Allavena P, Sica A (2004) Tumour-associated macrophages as a prototypic type II polarised phagocyte population: role in tumour progression. Eur J Cancer 40:1660–1667
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3:23–35
Sica A, Schioppa T, Mantovani A, Allavena P (2006) Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer 42:717–727
Chung EY, Kim BH, Hong JT, Lee CK, Ahn B, Nam SY, Han SB, Kim Y (2011) Resveratrol down-regulates interferon-gamma-inducible inflammatory genes in macrophages: molecular mechanism via decreased STAT-1 activation. J Nutr Biochem 22:902–909
Chuang CC, Martinez K, Xie G, Kennedy A, Bumrungpert A, Overman A, Jia W, McIntosh MK (2010) Quercetin is equally or more effective than resveratrol in attenuating tumor necrosis factor-{alpha}-mediated inflammation and insulin resistance in primary human adipocytes. Am J Clin Nutr 92:1511–1521
Reiter I, Krammer B, Schwamberger G (1999) Cutting edge: differential effect of apoptotic versus necrotic tumor cells on macrophage antitumor activities. J Immunol 163:1730–1732
Hagemann T, Lawrence T, McNeish I, Charles KA, Kulbe H, Thompson RG, Robinson SC, Balkwill FR (2008) “Re-educating” tumor-associated macrophages by targeting NF-kappaB. J Exp Med 205:1261–1268
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454:436–444
van Dongen M, Savage ND, Jordanova ES, Briaire-de Bruijn IH, Walburg KV, Ottenhoff TH, Hogendoorn PC, van der Burg SH, van der Burg SH, Gelderblom H, van Hall T (2010) Anti-inflammatory M2 type macrophages characterize metastasized and tyrosine kinase inhibitor-treated gastrointestinal stromal tumors. Int J Cancer 127:899–909
Acknowledgments
This work was supported by Graduate Scholarship Grants from the DFG to Bettina M. Kaminski and from the “Stiftung Polytechnische Gesellschaft Frankfurt am Main” to Maria-Christina Scherzberg. Bettina M. Kaminski was a member of the Frankfurt International Research Graduate School for Translational Biomedicine (FIRST), Frankfurt am Main. The authors thank Astrid Brüggerhoff for excellent technical assistance.
Conflict of interest
The authors have declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaminski, B.M., Weigert, A., Scherzberg, MC. et al. Resveratrol-induced potentiation of the antitumor effects of oxaliplatin is accompanied by an altered cytokine profile of human monocyte-derived macrophages. Apoptosis 19, 1136–1147 (2014). https://doi.org/10.1007/s10495-014-0988-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-0988-x