Abstract
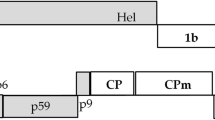
We investigated virus infection rates in Japanese garlic plants and found that a potyvirus, onion yellow dwarf virus (OYDV) was always associated with another potyvirus, leek yellow stripe virus (LYSV). When the OYDV HC-Pro genes from those viral isolates were PCR-amplified and sequenced, we found that all the HC-Pros isolated from Japanese garlic lacked a long stretch of the N-terminal amino acids (~ 100 residues) including the KITC motif, suggesting possible loss of viral aphid transmission. Our phylogenetic analysis revealed that HC-Pros were divided into three major groups and that they did not branch from a single common ancestor, suggesting different mechanisms for generation of the short HC-Pros from the intact ones. Because HC-Pro is a well-known RNA silencing suppressor (RSS), we then analyzed the RSS activity of HC-Pro using the conventional agroinfiltration assay with Nicotiana benthamiana and also with onion, which is a natural host of OYDV. The results demonstrated that the short HC-Pro had reduced RSS activity compared to the long HC-Pro while both type HC-Pros were well expressed in the agroinfiltrated tissues. In addition, the tested OYDV HC-Pros exhibited more efficient RSS activity in onion than in N. benthamiana. We presume that OYDV with reduced RSS activity of HC-Pro may take advantage of mixed infection with LYSV to survive.




Similar content being viewed by others
References
Ahn JW, Lee JS, Davarpanah SJ, Jeon JH, Park YI, Liu JR, Jeong WJ (2011) Host-dependent suppression of RNA silencing mediated by the viral suppressor p19 in potato. Planta 234:1065–1072
Arya M, Baranwal VK, Ahlawat YS, Singh L (2006) RT-PCR detection and molecular characterization of Onion yellow dwarf virus associated with garlic and onion. Curr Sci 91:1230–1234
Atreya CD, Pirone TP (1993) Mutational analysis of the helper component-proteinase gene of a potyvirus: effects of amino acid substitutions, deletions, and gene replacement on virulence and aphid transmissibility. Proc Natl Acad Sci USA 90:11919–11923
Bagi F, Stojšin V, Budakov D, El Swaeh SMA, Gvozdanović-Varga J (2012) Effect of onion yellow dwarf virus (OYDV) on yield components of fall garlic (Allium sativum L.) in Serbia. Afr J Agric Res 7:2386–2390
Blanc S, Ammar ED, Garcia-Lampasona S, Dolja VV, Llave C, Baker J, Pirone TP (1998) Mutations in the potyvirus helper component protein: effects on interactions with virions and aphid stylets. J Gen Virol 79:3119–3122
Bucher E, Hemmes H, de Haan P, Goldbach R, Prins M (2004) The influenza A virus NS1 protein binds small interfering RNAs and suppresses RNA silencing in plants. J Gen Virol 85:983–991
Cheng CP, Nagy PD (2003) Mechanism of RNA recombination in carmo- and tombusviruses: evidence for template switching by the RNA-dependent RNA polymerase in vitro. J Virol 77:12033–12047
Conci VC, Canavelli A, Lunello P, Di Rienzo J, Nome SF, Zumelzu G, Italia R (2003) Yield losses associated with virus-infected garlic plants during five successive years. Plant Dis 87:1411–1415
Cronin S, Verchot J, Haldeman-Cahill R, Schaad MC, Carrington JC (1995) Long-distance movement factor: a transport function of the potyvirus helper component proteinase. Plant Cell 7:549–559
Dolja VV, Herndon KL, Pirone TP, Carrington JC (1993) Spontaneous mutagenesis of a plant potyvirus genome after insertion of a foreign gene. J Virol 67:5968–5975
Dovas CI, Hatziloukas E, Salomon R, Barg E, Shiboleth Y, Katis NI (2001) Incidence of viruses infecting Allium spp. in Greece. Eur J Plant Pathol 107:677–684
Drake CJ, Tate HD, Harris HM (1933) The relationship of aphids to the transmission of yellow dwarf of onions. J Econ Entomol 26:841–846
El-Wahab ASA (2009) Aphid-transmission efficiency of two main viruses on garlic in Egypt, Onion yellow dwarf virus (OYDV-G) and Leek yellow stripe virus (LYSV-G). Acad J Entomol 2:40–42
Fajardo TVM, Nishijima M, Buso JA, Torres AC, Ávila AC, Resende RO (2001) Garlic viral complex: identification of potyviruses and carlavirus in central Brazil. Fitopatol Bras 26:619–626
Fayad-André MDS, Dusi AN, Resende RO (2011) Spread of viruses in garlic fields cultivated under different agricultural production systems in Brazil. Trop Plant Pathol 36:341–349
Gal-On A (2000) A point mutation in the FRNK motif of the potyvirus helper component-protease gene alters symptom expression in cucurbits and elicits protection against the severe homologous virus. Phytopathology 90:467–473
Ivanov KI, Eskelin K, Bašić M, De S, Lõhmus A, Varjosalo M, Mäkinen K (2016) Molecular insights into the function of the viral RNA silencing suppressor HCPro. Plant J 85:30–45
Jamous RM, Boonrod K, Fuellgrabe MW, Ali-Shtayeh MS, Krczal G, Wassenegger M (2011) The helper component-proteinase of the Zucchini yellow mosaic virus inhibits the Hua Enhancer 1 methyltransferase activity in vitro. J Gen Virol 92:2222–2226
Kok KH, Jin DY (2006) Influenza A virus NS1 protein does not suppress RNA interference in mammalian cells. J Gen Virol 87:2693–2644
Kumar S, Baranwal VK, Joshi S, Arya M, Majumder S (2010) Simultaneous detection of mixed infection of Onion yellow dwarf virus and an Allexivirus in RT-PCR for ensuring virus free onion bulbs. Indian J Virol 21:64–68
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Lakatos L, Csorba T, Pantaleo V, Chapman EJ, Carrington JC, Liu YP, Dolja VV, Calvino LF, López-Moya JJ, Burgyán J (2006) Small RNA binding is a common strategy to suppress RNA silencing by several viral suppressors. EMBO J 25:2768–2780
Manglli A, Mohammed HS, El Hussein AA, Agosteo GE, Albanese G, Tomassoli L (2014) Molecular analysis of the 3′ terminal region of Onion yellow dwarf virus from onion in southern Italy. Phytopathol Mediterr 53:438–450
Mohammed HS, Zicca S, Manglli A, Mohamed ME, El Siddig MAR, El Hussein AA, Tomassoli L (2013) Occurrence and phylogenetic analysis of potyviruses, carlaviruses and allexiviruses in garlic in Sudan. J Phytopathol 161:642–650
Peng Y, Kadoury D, Gal-On A, Huet H, Wang Y, Raccah B (1998) Mutations in the HC-Pro gene of zucchini yellow mosaic potyvirus: effects on aphid transmission and binding to purified virions. J Gen Virol 79:897–904
Plisson C, Drucker M, Blanc S, German-Retana S, Le Gall O, Thomas D, Bron P (2003) Structural characterization of HC-Pro, a plant virus multifunctional protein. J Biol Chem 278:23753–23761
Senda M, Masuta C, Ohnishi S, Goto K, Kasai A, Sano T, Hong JS, MacFarlane S (2004) Patterning of virus-infected Glycine max seed coat is associated with suppression of endogenous silencing of chalcone synthase genes. Plant Cell 16:807–818
Shiboleth YM, Haronsky E, Leibman D, Arazi T, Wassenegger M, Whitham SA, Gaba V, Gal-On A (2007) The conserved FRNK box in HC-Pro, a plant viral suppressor of gene silencing, is required for small RNA binding and mediates symptom development. J Virol 81:13135–13148
Soitamo AJ, Jada B, Lehto K (2011) HC-Pro silencing suppressor significantly alters the gene expression profile in tobacco leaves and flowers. BMC Plant Biol 11:68
Suzuki M, Hibi T, Masuta C (2003) RNA recombination between cucumoviruses: possible role of predicted stem-loop structures and an internal subgenomic promoter-like motif. Virology 306:77–86
Takaki F, Sano T, Yamashita K (2006) The complete nucleotide sequence of attenuated onion yellow dwarf virus: a natural poyvirus deletion mutant lacking the N-terminal 92 amino acids of HC-Pro. Arch Virol 151:1439–1445
Takaki F, Sano T, Yamashita K, Fujita T, Ueda K, Kato T (2005) Complete nucleotide sequences of attenuated and severe isolates of Leek yellow stripe virus from garlic in northern Japan: identification of three distinct virus types in garlic and leek world-wide. Arch Virol 150:1135–1149
Valli AA, Gallo A, Rodamilans B, López-Moya JJ, García JA (2018) The HCPro from the Potyviridae family: an enviable multitasking helper component that every virus would like to have. Mol Plant Pathol 19:744–763
Várallyay É, Havelda Z (2013) Unrelated viral suppressors of RNA silencing mediate the control of ARGONAUTE1 level. Mol Plant Pathol 14:567–575
Verma RK, Mishra R, Petrov NM, Stoyanova M, Stoev A, Bakardjieva N, Gaur RK (2015) Molecular characterization and recombination analysis of an Indian isolate of onion yellow dwarf virus. Eur J Plant Pathol 143:437–445
Wu HW, Lin SS, Chen KC, Yeh SD, Chua NH (2010) Discriminating mutations of HC-Pro of Zucchini yellow mosaic virus with differential effects on small RNA pathways involved in viral pathogenicity and symptom development. Mol Plant Microbe Interact 23:17–28
Xu K, Huang X, Wu M, Wang Y, Chang Y, Liu K, Zhang J, Zhang Y, Zhang F, Yi L, Li T, Wang R, Tan G, Li C (2014) A rapid, highly efficient and economical method of Agrobacterium-mediated in planta transient transformation in living onion epidermis. PLoS ONE 9:e83556
Yoshida N, Shimura H, Yamashita K, Suzuki M, Masuta C (2012) Variability in the P1 gene helps to refine phylogenetic relationships among leek yellow stripe virus isolates from garlic. Arch Virol 157:147–153
Acknowledgements
We are grateful to Dr. Reika Isoda for her helpful advice on phylogenetic analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
This article does not contain any studies with human participants or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10327_2020_926_MOESM1_ESM.pptx
Supplementary file1 Fig. S1. Multiple alignment of the N-terminal region of the onion yellow dwarf virus (OYDV) HC-Pro nucleotide sequences. The alignment was created using MEGA v7. Arrowheads on the left indicate the OYDV HC-Pro genes sequenced in this study. All the OYDV HC-Pro sequences in the NCBI database were included in the analysis (PPTX 652 kb)
10327_2020_926_MOESM2_ESM.pptx
Supplementary file2 Fig. S2. Agroinfiltration assay of tobacco etch virus (TEV) HC-Pro in onion epidermal cells. (a) RSS activity of TEV HC-Pro in cells. Agrobacterium carrying the 35S:GFP construct was used to coinfiltrate three onion bulb scales with the 35S:TEV-HC-Pro construct. Inocula were prepared as described for Fig. 4. Bright field (BF) and GFP (UV) images were taken using an epifluorescence microscope (Leica DMI 6000B) at 3 dpi. (b) Fold change in GFP signals in areas in onion epidermis agroinfiltrated with different HC-Pro isolates. GFP signal intensities were quantified as described for Fig. 4. Means (±SE) among isolates were analyzed for significant differences using Student’s t-test (*P < 0.05) (PPTX 787 kb)
10327_2020_926_MOESM3_ESM.pdf
Supplementary file3 Fig. S3 Prediction of 3D structures of onion yellow dwarf virus (OYDV) HC-Pro proteins. Ab initio structural prediction of OYDV HC-Pro used in this study was done using the I-TASSER v5.1 server (https://zhanglab.ccmb.med.umich.edu/I-TASSER/), and the prediction models were visualized using UCSF chimera program v1.9. The two blue arrowheads in China HC-Pro indicate the start and end sites of the unique long α-helix motif. The schematic structures above each model show a simplified image of the HC-Pro proteins. Dotted boxes represent the deletion in the HC-Pro N-terminal region (PDF 37 kb)
Rights and permissions
About this article
Cite this article
Kim, H., Aoki, N., Takahashi, H. et al. Reduced RNA silencing suppressor activity of onion yellow dwarf virus HC-Pro with N-terminal deletion may be complemented in mixed infection with another potyvirus in garlic. J Gen Plant Pathol 86, 300–309 (2020). https://doi.org/10.1007/s10327-020-00926-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-020-00926-2