Abstract
Background
Cisplatin is a potent anti-tumor compound. Nephrotoxicity-inducing oxidative stress is a common side effect. This study was conducted to find out whether, the triterpenoid saponin of Terminalia arjuna (TA), Arjunolic acid which is a natural antioxidant, could prevent cisplatin-induced renal toxicity and if so, explore its possible renoprotective mechanism.
Methods
Thirty male Sprague–Dawley rats were divided into three groups: Control group: rats received saline injection, cisplatin group: rats injected intraperitoneally with 7 mg/kg cisplatin and Arjunolic acid group: rats received 20 mg/kg Arjunolic acid daily for 10 days with cisplatin injection on day 5. Serum creatinine and blood urea nitrogen (BUN) were determined and kidney sections were obtained for histopathology. Oxidative stress was evaluated in kidney homogenates by measuring malondialdehyde (MDA), reduced glutathione (GSH) and nitric oxide (NO) levels. Renal gene expressions of transforming growth factor-beta (TGF-β), nuclear factor-kappa B (NF-κB), kidney injury molecule-1 (Kim-1) and B cell lymphoma-2 (Bcl-2) were estimated.
Results
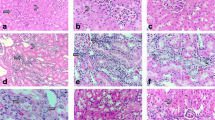
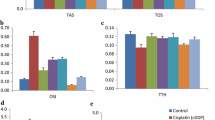
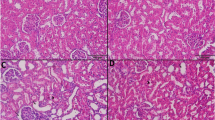
Cisplatin-treated rats showed a significant reduction in renal GSH and a significant elevation of serum creatinine, BUN, MDA and NO renal levels when compared with control. Moreover, upregulation of TGF-β, NF-κB and Kim-1 along with downregulation of Bcl-2 renal expressions were also observed in cisplatin-treated rats in comparison to control. All these markers were significantly reversed by TA triterpenoid saponin administration.
Conclusion
Arjunolic acid ameliorated the nephrotoxic biochemical changes induced by cisplatin supporting its renoprotective effects which may be mediated by attenuation of oxidative stress markers, downregulation of renal expressions of fibrotic (TGF-β), inflammatory (NF-κB) and kidney injury (Kim-1) markers along with upregulation of renal antiapoptotic marker (Bcl-2) gene expressions.


Similar content being viewed by others
References
Schrier RW. Cancer therapy and renal injury. J Clin Invest. 2002;110(6):743–5.
Yao X, Panichpisal K, Kurtzman N, Nugent K. Cisplatin nephrotoxicity: a review. Am J Med Sci. 2007;334(2):115–24.
Launay-Vacher V, Rey J-B, Isnard-Bagnis C, Deray G, Daouphars M. Prevention of cisplatin nephrotoxicity: state of the art and recommendations from the European Society of Clinical Pharmacy Special Interest Group on Cancer Care. Cancer Chemother Pharmacol. 2008;61(6):903–9.
Miyawaki Y, Ueki M, Ueno M, Asaga T, Tokuda M, Shirakami G. d-Allose ameliorates cisplatin-induced nephrotoxicity in mice. Tohoku J Exp Med. 2012;228(3):215–21.
Mukhopadhyay P, Horváth B, Zsengellér Z, Zielonka J, Tanchian G, Holovac E, Kechrid M, Patel V, Stillman IE, Parikh SM. Mitochondrial-targeted antioxidants represent a promising approach for prevention of cisplatin-induced nephropathy. Free Radic Biol Med. 2012;52(2):497–506.
Mukhopadhyay P, Horváth B, Kechrid M, Tanchian G, Rajesh M, Naura AS, Boulares AH, Pacher P. Poly (ADP-ribose) polymerase-1 is a key mediator of cisplatin-induced kidney inflammation and injury. Free Radic Biol Med. 2011;51(9):1774–88.
Ahmad MS, Ahmad S, Gautam B, Arshad M, Afzal M. Terminalia arjuna, a herbal remedy against environmental carcinogenicity: an in vitro and in vivo study. Egypt J Med Hum Genet. 2014;15(1):61–7.
Ghosh J, Sil PC. Arjunolic acid: a new multifunctional therapeutic promise of alternative medicine. Biochimie. 2013;95(6):1098–109.
Manna P, Sinha M, Sil PC. Aqueous extract of Terminalia arjuna prevents carbon tetrachloride induced hepatic and renal disorders. BMC Complement Altern Med. 2006;6(1):33.
Sinha M, Manna P, Sil PC. Aqueous extract of the bark of Terminalia arjuna plays a protective role against sodium-fluoride-induced hepatic and renal oxidative stress. J Nat Med. 2007;61(3):251–60.
Ulu R, Dogukan A, Tuzcu M, Gencoglu H, Ulas M, İlhan N, Muqbil I, Mohammad RM, Kucuk O, Sahin K. Regulation of renal organic anion and cation transporters by thymoquinone in cisplatin induced kidney injury. Food Chem Toxicol. 2012;50(5):1675–9.
Sinha M, Manna P, Sil PC. Arjunolic acid attenuates arsenic-induced nephrotoxicity. Pathophysiology. 2008;15(3):147–56.
Song J, Liu D, Feng L, Zhang Z, Jia X, Xiao W. Protective effect of standardized extract of Ginkgo biloba against cisplatin-induced nephrotoxicity. Evid Based Complement Altern Med. 2013;2013:1–11.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25(4):402–8.
Ali B, Al-Moundhri M, Eldin MT, Nemmar A, Al-Siyabi S, Annamalai K. Amelioration of cisplatin-induced nephrotoxicity in rats by tetramethylpyrazine, a major constituent of the Chinese herb Ligusticum wallichi. Exp Biol Med. 2008;233(7):891–6.
Rashed LA, Hashem RM, Soliman HM. Oxytocin inhibits NADPH oxidase and P38 MAPK in cisplatin-induced nephrotoxicity. Biomed Pharmacother. 2011;65(7):474–80.
Talebi A, Nasri H, Khazaei M, Baradaran-Mahdavi M-M, Jafapisheh A, Olia B, Pirhaji O, Hashemi-Nia SJ, Eshraghi F, Pezeshki Z. A combination of Vitamin C and Losartan for cisplatin-induced Nephrotoxicity in Rats. Iran J Kidney Dis. 2012;6(5):361–5.
Özen S, Akyol Ö, Iraz M, Söğüt S, Özuğurlu F, Özyurt H, Odacı E, Yıldırım Z. Role of caffeic acid phenethyl ester, an active component of propolis, against cisplatin-induced nephrotoxicity in rats. J Appl Toxicol. 2004;24(1):27–35.
Chirino YI, Pedraza-Chaverri J. Role of oxidative and nitrosative stress in cisplatin-induced nephrotoxicity. Exp Toxicol Pathol. 2009;61(3):223–42.
Chirino YI, Trujillo J, Sánchez-González DJ, Martínez-Martínez CM, Cruz C, Bobadilla NA, Pedraza-Chaverri J. Selective iNOS inhibition reduces renal damage induced by cisplatin. Toxicol Lett. 2008;176(1):48–57.
El-Sayed E-SM, Abd-Ellah MF, Attia SM. Protective effect of captopril against cisplatin-induced nephrotoxicity in rats. Pak J Pharm Sci. 2008;21(3):255–61.
Yildirim Z, Sogut S, Odaci E, Iraz M, Ozyurt H, Kotuk M, Akyol O. Oral erdosteine administration attenuates cisplatin-induced renal tubular damage in rats. Pharmacol Res. 2003;47(2):149–56.
Mansour MA, Mostafa AM, Nagi MN, Khattab MM, Al-Shabanah OA. Protective effect of aminoguanidine against nephrotoxicity induced by cisplatin in normal rats. Comp Biochem Physiol C Toxicol Pharmacol. 2002;132(2):123–8.
Atasayar S, Gürer-Orhan H, Orhan H, Gürel B, Girgin G, Özgüneş H. Preventive effect of aminoguanidine compared to vitamin E and C on cisplatin-induced nephrotoxicity in rats. Exp Toxicol Pathol. 2009;61(1):23–32.
Palipoch S, Punsawad C. Biochemical and histological study of rat liver and kidney injury induced by cisplatin. J Toxicolog Pathol. 2013;26(3):293.
Ghosh J, Das J, Manna P, Sil PC. Acetaminophen induced renal injury via oxidative stress and TNF-α production: therapeutic potential of Arjunolic acid. Toxicology. 2010;268(1):8–18.
El-Naga RN. Pre-treatment with cardamonin protects against cisplatin-induced nephrotoxicity in rats: impact on NOX-1, inflammation and apoptosis. Toxicol Appl Pharmacol. 2014;274(1):87–95.
Chaney SG, Campbell SL, Temple B, Bassett E, Wu Y, Faldu M. Protein interactions with platinum—DNA adducts: from structure to function. J Inorg Biochem. 2004;98(10):1551–9.
Pal S, Pal PB, Das J, Sil PC. Involvement of both intrinsic and extrinsic pathways in hepatoprotection of Arjunolic acid against cadmium induced acute damage. Toxicology. 2011;283(2):129–39.
Manna P, Sil PC. Arjunolic acid: beneficial role in type 1 diabetes and its associated organ pathophysiology. Free Radic Res. 2012;46(7):815–30.
Manna P, Sil PC. Impaired redox signaling and mitochondrial uncoupling contributes vascular inflammation and cardiac dysfunction in type 1 diabetes: protective role of Arjunolic acid. Biochimie. 2012;94(3):786–97.
Luo J, Tsuji T, Yasuda H, Sun Y, Fujigaki Y, Hishida A. The molecular mechanisms of the attenuation of cisplatin-induced acute renal failure by N-acetylcysteine in rats. Nephrol Dial Transplant. 2008;23(7):2198–205.
Liu W, Zhang X, Liu P, Shen X, Lan T, Li W, Jiang Q, Xie X, Huang H. Effects of berberine on matrix accumulation and NF-kappa B signal pathway in alloxan-induced diabetic mice with renal injury. Eur J Pharmacol. 2010;638(1–3):150–5.
Ilbey YO, Ozbek E, Cekmen M, Simsek A, Otunctemur A, Somay A. Protective effect of curcumin in cisplatin-induced oxidative injury in rat testis: mitogen-activated protein kinase and nuclear factor-kappa B signaling pathways. Hum Reprod. 2009;24(7):1717–25.
Moraes PA, Yonamine CY, Pinto DC Jr, Esteves JV, Machado UF, Mori RC. Insulin acutely triggers transcription of Slc2a4 gene: participation of the AT-rich. E-box and NFKB-binding sites. Life Sci. 2014;114(1):36–44.
Ross JW, Ashworth MD, Mathew D, Reagan P, Ritchey JW, Hayashi K, Spencer TE, Lucy M, Geisert RD. Activation of the transcription factor, nuclear factor kappa-B, during the estrous cycle and early pregnancy in the pig. Reprod Biol Endocrinol. 2010;8:39.
Pérez-Rojas JM, Cruz C, García-López P, Sánchez-González DJ, Martínez-Martínez CM, Ceballos G, Espinosa M, Melendez-Zajgla J, Pedraza-Chaverri J. Renoprotection by α-mangostin is related to the attenuation in renal oxidative/nitrosative stress induced by cisplatin nephrotoxicity. Free Radic Res. 2009;43(11):1122–32.
Wolf G. Renal injury due to renin–angiotensin–aldosterone system activation of the transforming growth factor-beta pathway. Kidney Int. 2006;70(11):1914–9.
Kusumoto M, Kamobayashi H, Sato D, Komori M, Yoshimura M, Hamada A, Kohda Y, Tomita K, Saito H. Alleviation of cisplatin-induced acute kidney injury using phytochemical polyphenols is accompanied by reduced accumulation of indoxyl sulfate in rats. Clin Exp Nephrol. 2011;15(6):820–30.
Al-Kharusi N, Babiker H, Al-Salam S, Waly M, Nemmar A, Al-Lawati I, Yasin J, Beegam S, Ali B. Ellagic acid protects against cisplatin-induced nephrotoxicity in rats: a dose-dependent study. Eur Rev Med Pharmacol Sci. 2013;17(3):299–310.
Zhou Y, Vaidya VS, Brown RP, Zhang J, Rosenzweig BA, Thompson KL, Miller TJ, Bonventre JV, Goering PL. Comparison of kidney injury molecule-1 and other nephrotoxicity biomarkers in urine and kidney following acute exposure to gentamicin, mercury, and chromium. Toxicol Sci. 2008;101(1):159–70.
Prozialeck WC, Edwards JR, Lamar PC, Liu J, Vaidya VS, Bonventre JV. Expression of kidney injury molecule-1 (Kim-1) in relation to necrosis and apoptosis during the early stages of Cd-induced proximal tubule injury. Toxicol Appl Pharmacol. 2009;238:306–14.
van Timmeren MM, Van Den Heuvel MC, Bailly V, Bakker SJ, van Goor H, Stegeman CA. Tubular kidney injury molecule-1 (Kim-1) in human renal disease. J Pathol. 2007;212:209–17.
Qi S, Wu D. Bone marrow-derived mesenchymal stem cells protect against cisplatin-induced acute kidney injury in rats by inhibiting cell apoptosis. Int J Mol Med. 2013;32(6):1262–72.
Ognjanović BI, Djordjević NZ, Matić MM, Obradović JM, Mladenović JM, Štajn AS, Saičić ZS. Lipid peroxidative damage on cisplatin exposure and alterations in antioxidant defense system in rat kidneys: a possible protective effect of selenium. Int J Mol Sci. 2012;13(2):1790–803.
Tanabe K, Tamura Y, Lanaspa MA, Miyazaki M, Suzuki N, Sato W, Maeshima Y, Schreiner GF, Villarreal FJ, Johnson RJ, Nakagawa T. Epicatechin limits renal injury by mitochondrial protection in cisplatin nephropathy. Am J Physiol Renal Physiol. 2012;303:F1264–74.
Acknowledgments
The author acknowledges Dr. Azza Abdel-Aziz, Associate Professor of Pathology, Faculty of Medicine, Mansoura University, for histopathological examination of the kidney. This research received no specific Grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The author declares no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sherif, I.O. Amelioration of cisplatin-induced nephrotoxicity in rats by triterpenoid saponin of Terminalia arjuna . Clin Exp Nephrol 19, 591–597 (2015). https://doi.org/10.1007/s10157-014-1056-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-1056-0