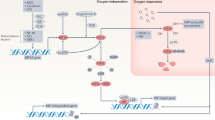
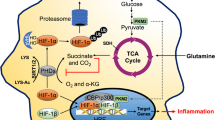
Abstract
Recent studies suggest a novel role of HIF-1α under non-hypoxic conditions, including antibacterial and antiviral innate immune responses. However, the identity of the pathogen-associated molecular pattern which triggers HIF-1α activation during the antiviral response remains to be identified. Here, we demonstrate that cellular administration of double-stranded nucleic acids, the molecular mimics of viral genomes, results in the induction of HIF-1α protein level as well as the increase in HIF-1α target gene expression. Whole-genome DNA microarray analysis revealed that double-stranded nucleic acid treatment triggers induction of a number of hypoxia-inducible genes, and induction of these genes are compromised upon siRNA-mediated HIF-1α knock-down. Interestingly, HIF-1α knock-down also resulted in down-regulation of a number of genes involved in antiviral innate immune responses. Our study demonstrates that HIF-1α activation upon nucleic acid-triggered antiviral innate immune responses plays an important role in regulation of genes involved in not only hypoxic response, but also immune response.
Similar content being viewed by others
References
Akira, S., Uematsu, S., and Takeuchi, O. (2006). Pathogen recognition and innate immunity. Cell 124, 783–801.
An, J., and Rettig, M.B. (2005). Mechanism of von Hippel-Lindau protein-mediated suppression of nuclear factor kappa B activity. Mol. Cell. Biol. 25, 7546–7556.
An, J., Fisher, M., and Rettig, M.B. (2005). VHL expression in renal cell carcinoma sensitizes to bortezomib (PS-341). through an NF-kappaB-dependent mechanism. Oncogene 24, 1563–1570.
Bolstad, B.M., Irizarry, R.A., Astrand, M., and Speed, T.P. (2003). A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19, 185–193.
Bosco, M.C., Puppo, M., Santangelo, C., Anfosso, L., Pfeffer, U., Fardin, P., Battaglia, F., and Varesio, L. (2006). Hypoxia modifies the transcriptome of primary human monocytes: modulation of novel immune-related genes and identification of CC-chemokine ligand 20 as a new hypoxia-inducible gene. J. Immunol. 177, 1941–1955.
Chi, J.T., Wang, Z., Nuyten, D.S., Rodriguez, E.H., Schaner, M.E., Salim, A., Wang, Y., Kristensen, G.B., Heiland, A., Borresen-Dale, A.L., et al. (2006). Gene expression programs in response to hypoxia: cell type specificity and prognostic significance in human cancers. PLoS Med. 3, e47.
Choi, J.W., Park, S.C., Kang, G.H., Liu, J.O., and Youn, H.D. (2004). Nur77 activated by hypoxia-inducible factor-1alpha overproduces proopiomelanocortin in von Hippel-Lindau-mutated renal cell carcinoma. Cancer Res. 64, 35–39.
Dennis, G., Jr., Sherman, B.T., Hosack, D.A., Yang, J., Gao, W., Lane, H.C., and Lempicki, R.A. (2003). DAVID: database for annotation, visualization, and integrated discovery. Genome Biol. 4, P3.
Dery, M.A., Michaud, M.D., and Richard, D.E. (2005). Hypoxia-inducible factor 1: regulation by hypoxic and non-hypoxic activators. Int. J. Biochem. Cell Biol. 37, 535–540.
Ebert, B.L, Firth, J.D., and Ratcliffe, P.J. (1995). Hypoxia and mitochondrial inhibitors regulate expression of glucose transporter-1 via distinct Cis-acting sequences. J. Biol. Chem. 270, 29083–29089.
Franovic, A., Gunaratnam, L., Smith, K., Robert, I., Patten, D., and Lee, S. (2007). Translational up-regulation of the EGFR by tumor hypoxia provides a nonmutational explanation for its over-expression in human cancer. Proc. Natl. Acad. Sei. USA 104, 13092–13097.
Frede, S., Stockmann, C., Freitag, P., and Fandrey, J. (2006). Bacterial lipopolysaccharide induces HIF-1 activation in human monocytes via p44/42 MAPK and NF-kappaB. Biochem. J. 396, 517–527.
Gotea, V., and Ovcharenko, I. (2008). DiRE: identifying distant regulatory elements of co-expressed genes. Nucleic Acids Res. 36, W133–139.
Gross, C., Dubois-Pot, H., and Wasylyk, B. (2008). The ternary complex factor NefElk-3 participates in the transcriptional response to hypoxia and regulates HIF-1 alpha. Oncogene 27, 1333–1341.
Hong, S.W., Kim, S., and Lee, D.K. (2008). The role of Bach2 in nucleic acid-triggered antiviral innate immune responses. Biochem. Biophys. Res. Commun. 365, 426–432.
Hwang, I.I., Watson, I.R., Der, S.D., and Ohh, M. (2006). Loss of VHL confers hypoxia-inducible factor (HIF).-dependent resistance to vesicular stomatitis virus: role of HIF in antiviral response. J. Virol. 80, 10712–10723.
Irizarry, R.A., Bolstad, B.M., Collin, F., Cope, L.M., Hobbs, B., and Speed, T.P. (2003). Summaries of affymetrix genechip probe level data. Nucleic Acids Res. 31, e15.
Kilani, M.M., Mohammed, K.A., Nasreen, N., Tepper, R.S., and Antony, V.B. (2004). RSV causes HIF-1 alpha stabilization via NO release in primary bronchial epithelial cells. Inflammation 28, 245–251.
Kim, K.S., Rajagopal, V., Gonsalves, C., Johnson, C., and Kalra, V.K. (2006). A novel role of hypoxia-inducible factor in cobalt chloride- and hypoxia-mediated expression of IL-8 chemokine in human endothelial cells. J. Immunol. 177, 7211–7224.
Kim, H.A., Kim, K., Kim, S.W., and Lee, M. (2007a). Transcriptional and post-translational regulatory system for hypoxia specific gene expression using the erythropoietin enhancer and the oxygen-dependent degradation domain. J. Control. Release 121, 218–224.
Kim, H.Y., Kim, Y.H., Nam, B.H., Kong, H.J., Kim, H.H., Kim, Y.J., An, W.G., and Cheong, J. (2007b). HIF-1alpha expression in response to lipopolysaccaride mediates induction of hepatic inflammatory cytokine TNFalpha. Exp. Cell Res. 373, 1866–1876.
Lee, M.S., and Kim, Y.J. (2007). Pattern-recognition receptor signaling initiated from extracellular, membrane, and cytoplasmic space. Mol. Cells 23, 1–10.
Liu, X.H., Yu, E.Z., Li, Y.Y., and Kagan, E. (2006). HIF-1alpha has an anti-apoptotic effect in human airway epithelium that is mediated via Mcl-1 gene expression. J. Cell. Biochem. 97, 755–765.
Manalo, D.J., Rowan, A., Lavoie, T., Natarajan, L., Kelly, B.D., Ye, S.O., Garcia, J.G., and Semenza, G.L. (2005). Transcriptional regulation of vascular endothelial cell responses to hypoxia by HIF-1. Blood 705, 659–669.
Masson, N., Willam, C., Maxwell, P.H., Pugh, C.W., and Ratcliffe, P.J. (2001). Independent function of two destruction domains in hypoxia-inducible factor-alpha chains activated by prolyl hydroxylation. EMBO J. 20, 5197–5206.
Nasimuzzaman, M., Waris, G., Mikolon, D., Stupack, D.G., and Siddiqui, A. (2007). Hepatitis C virus stabilizes hypoxia-inducible factor 1 alpha and stimulates the synthesis of vascular endothelial growth factor. J. Virol. 81, 10249–10257.
Peyssonnaux, C., Cejudo-Martin, P., Doedens, A., Zinkernagel, A.S., Johnson, R.S., and Nizet, V. (2007). Cutting edge: Essential role of hypoxia inducible factor-1 alpha in development of lipopolysaccharide-induced sepsis. J. Immunol. 773, 7516–7519.
Samanta, M., Iwakiri, D., Kanda, T., Imaizumi, T., and Takada, K. (2006). EB virus-encoded RNAs are recognized by RIG-I and activate signaling to induce type IIFN. EMBO J. 25, 4207–4214.
Sonna, L.A., Cullivan, M.L., Sheldon, H.K., Pratt, R.E., and Lilly, C.M. (2003). Effect of hypoxia on gene expression by human hepatocytes (HepG2). Physiol. Genomics 12, 195–207.
Ui-Tei, K., Naito, Y., Takahashi, F., Haraguchi, T., Ohki-Hamazaki, H., Juni, A., Ueda, R., and Saigo, K. (2004). Guidelines for the selection of highly effective siRNA sequences for mammalian and chick RNA interference. Nucleic Acids Res. 32, 936–948.
van Uden, P., Kenneth, N.S., and Rocha, S. (2008). Regulation of hypoxia-inducible factor-1 alpha by NF-kappaB. Biochemistry 412, 477–484.
Walmsley, S.R., Print, C., Farahi, N., Peyssonnaux, C., Johnson, R.S., Cramer, T., Sobolewski, A., Condliffe, A.M., Cowburn, A.S., Johnson, N., et al. (2005). Hypoxia-induced neutrophil survival is mediated by HIF-1 alpha-dependent NF-kappaB activity. J. Exp. Med. 207, 105–115.
Wang, G.L., and Semenza, G.L. (1993). General involvement of hypoxia-inducible factor 1 in transcriptional response to hypoxia. Proc. Natl. Acad. Sei. USA 90, 4304–4308.
Wang, G.L., Jiang, B.H., Rue, E.A., and Semenza, G.L. (1995). Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular 02 tension. Proc. Natl. Acad. Sei. USA 92, 5510–5514.
Wykoff, C.C., Beasley, N.J., Watson, P.H., Turner, K.J., Pastorek, J., Sibtain, A., Wilson, G.D., Turley, H., Talks, K.L., Maxwell, P.H., et al. (2000). Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res. 60, 7075–7083.
Yoo, J.W., Hong, S.W., Kim, S., and Lee, D.K. (2006). Inflammatory cytokine induction by siRNAs is cell type- and transfection reagent-specific. Biochem. Biophys. Res. Commun. 347, 1053–1058.
Zamanian-Daryoush, M., Der, S.D., and Williams, B.R. (1999). Cell cycle regulation of the double stranded RNA activated protein kinase, PKR. Oncogene 18, 315–326.
Zhou, J., Schmid, T., and Brune, B. (2003). Tumor necrosis factor-alpha causes accumulation of a ubiquitinated form of hypoxia inducible factor-1 alpha through a nuclear factor-kappaB-dependent pathway. Mol. Biol. Cell 14, 2216–2225.
Zinkernagel, A.S., Johnson, R.S., and Nizet, V. (2007). Hypoxia inducible factor (HIF). function in innate immunity and infection. J. Mol. Med. 85, 1339–1346.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Hong, S.W., Yoo, J.W., Kang, H.S. et al. HIF-1α-dependent gene expression program during the nucleic acid-triggered antiviral innate immune responses. Mol Cells 27, 243–250 (2009). https://doi.org/10.1007/s10059-009-0030-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0030-2