Abstract
Purpose
Aluminum potassium sulfate and tannic acid (ALTA) is an effective sclerosing agent for internal hemorrhoids. However, it is contraindicated for patients with chronic renal failure on dialysis, because the aluminum in ALTA can cause aluminum encephalopathy when it is not excreted effectively. We conducted this study to measure the serum aluminum concentrations and observe for symptoms relating to aluminum encephalopathy in dialysis patients after ALTA therapy.
Methods
Ten dialysis patients underwent ALTA therapy for hemorrhoids. We measured their serum aluminum concentrations and observed them for possible symptoms of aluminum encephalopathy.
Results
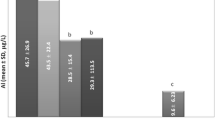
The total injection volume of ALTA solution was 31 mL (24–37). The median serum aluminum concentration before ALTA therapy was 9 μg/L, which increased to 741, 377, and 103 μg/L, respectively, 1 h, 1 day, and 1 week after ALTA therapy. These levels decreased rapidly, to 33 μg/L by 1 month and 11 μg/L by 3 months after ALTA therapy. No patient suffered symptoms related to aluminum encephalopathy.
Conclusions
Although the aluminum concentrations increased temporarily after ALTA therapy, dialysis patients with levels below 150 μg/L by 1 week and thereafter are considered to be at low risk of the development of aluminum encephalopathy.

Similar content being viewed by others
References
Shi Z. Treatment of hemorrhoid of III, IV stage by four steps injection of Xiaozhiling. Zhongguo Zhong Xi Yi Jie He Za Zhi. 1998;18:201–3.
Takamura H, Inatsugu H, Yoshikawa S, Masuda T. Treatment of internal hemorrhoids with Xiaozhiling injection. J Jpn Soc Coloproctol. 2001;54:910–4 (Japanese with English abstract).
Takano M, Iwadare J, Ohba H, Takamura H, Masuda Y, Matsuo K, et al. Sclerosing therapy of internal hemorrhoids with a novel sclerosing agent. Comparison with ligation and excision. Int J Colorectal Dis. 2006;21:44–51.
Yamamoto Y, Miwa M. Visualization of diffusion of the drug solution during aluminum potassium tannic acid injection therapy: a pilot study. Surg Today. 2013;43:698–701.
Ono T, Goto K, Takagi S, Iwasaki S, Komatsu H. Sclerosing effect of OC-108, a novel agent for hemorrhoids, is associated with granulomatous inflammation induced by aluminum. J Pharmacol Sci. 2005;99:353–63.
Sheikh F, Khubchandani IT, Rosen L, Sheets JA, Stasik JJ. Is anorectal surgery on chronic dialysis patients risky? Dis Colon Rectum. 1992;35:56–8.
Kosugi M, Nakashima H, Tabata S. A case of sclerotherapy using a novel agent (ALTA, ZIONER) for prolapsed internal hemorrhoids with renal failure under hemodialysis and its clinical significance. J Jpn Soc Coloproctol. 2008;61:71–5 (Japanese with English abstract).
Rob PM, Niederstadt C, Reusche E. Dementia in patients undergoing long-term dialysis: aetiology, differential diagnoses, epidemiology and management. CNS Drugs. 2001;15:691–9.
Hachiro Y, Kunimoto M, Abe T, Kitada M, Ebisawa Y. Aluminum potassium sulfate and tannic acid (ALTA) injection as the mainstay of treatment for internal hemorrhoids. Surg Today. 2011;41:806–9.
Bondy SC. The neurotoxicity of environmental aluminum is still an issue. Neurotoxicology. 2010;31:575–81.
Priest ND, Talbot RJ, Newton D, Day JP, King SJ, Fifield LK. Uptake by man of aluminium in a public water supply. Hum Exp Toxicol. 1988;17:296–301.
Harris WR, Wang Z, Hamada YZ. Competition between transferrin and the serum ligands citrate and phosphate for the binding of aluminum. Inorg Chem. 2003;42:3262–73.
Yokel RA, Rhineheimer SS, Sharma P, Elmore D, McNamara PJ. Entry, half-life, and desferrioxamine accelerated clearance of brain aluminum after a single Al exposure. Toxicol Sci. 2001;64:77–82.
Flendrig JA, Kruis H, Das HA. Letter: aluminum and dialysis dementia. Lancet. 1976;5(1(7971)):1235.
Jaffe JA, Liftman C, Glickman JD. Frequency of elevated serum aluminum levels in adult dialysis patients. Am J Kidney Dis. 2005;46:316–9.
Alfrey AC, LeGendre GR, Kaehny WD. The dialysis encephalopathy syndrome. Possible aluminum intoxication. N Engl J Med. 1976;22(294):184–8.
EgbunaOI Bosde A. Acute aluminium neurotoxicity secondary to treatment of severe hyperphosphatemia of acute renal failure and the K/DOQI guidelines: a case report and review of the literature. Internet J Nephrol. 2005;2:2.
Malaki M. Acute encephalopathy following the use of aluminum hydroxide in a boy affected with chronic kidney disease. J Pediatr Neurosci. 2013;8:81–2.
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42:S1–201.
Chazan JA, Abuelo JG, Blonsky SL. Plasma aluminum levels (unstimulated and stimulated): clinical and biochemical findings in 185 patients undergoing chronic hemodialysis for 4 to 95 months. Am J Kidney Dis. 1989;13:284–9.
Takamura H, Takano M, Ohba H, Fukano M, Iwadare J, Koganezawa S, et al. Investigation of efficacy, safety and pharmacokinetics with OC-108, a novel agent for sclerosing therapy of internal hemorrhoids—early phase study. Jpn Pharmacol Ther. 2004;32:355–65 (Japanese with English abstract).
Deguchi H. A clinical study using a novel agent (aluminium potassium sulfate hydrate tannic acid: ALTA) to measure changes in pre- and post-sclerotherapy serum aluminium levels in patients with internal hemorrhoids. J Hyogo Med Assoc. 2010;52:48–52 (Japanese with English abstract).
Nakamura H, Rose PG, Blumer JL, Reed MD. Acute encephalopathy due to aluminium toxicity successfully treated by combined intravenous deferoxamine and hemodialysis. J Clin Pharmacol. 2000;40:296–300.
Kirschbaum BB, Schoolwerth AC. Acute aluminium toxicity associated with oral citrate and aluminium-containing antacids. Am J Med Sci. 1989;297:9–11.
Conflict of interest
Akira Tsunoda and his co-authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsunoda, A., Nakagi, M., Kano, N. et al. Serum aluminum levels in dialysis patients after sclerotherapy of internal hemorrhoids with aluminum potassium sulfate and tannic acid. Surg Today 44, 2314–2317 (2014). https://doi.org/10.1007/s00595-014-0914-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-014-0914-4