Abstract
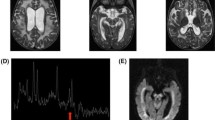
Leigh syndrome (LS) is an early-onset progressive neurodegenerative disorder characterized by unique, bilateral neuropathological findings in brainstem, basal ganglia, cerebellum and spinal cord. LS is genetically heterogeneous, with the majority of the causative genes affecting mitochondrial malfunction, and many cases still remain unsolved. Here, we report male sibs affected with LS showing ketonemia, but no marked elevation of lactate and pyruvate. To identify their genetic cause, we performed whole exome sequencing. Candidate variants were narrowed down based on autosomal recessive and X-linked recessive models. Only one hemizygous missense mutation (c.665G>C, p.W222S) in glycogenin-2 (GYG2) (isoform a: NM_001079855) in both affected sibs and a heterozygous change in their mother were identified, being consistent with the X-linked recessive trait. GYG2 encodes glycogenin-2 (GYG2) protein, which plays an important role in the initiation of glycogen synthesis. Based on the structural modeling, the mutation can destabilize the structure and result in protein malfunctioning. Furthermore, in vitro experiments showed mutant GYG2 was unable to undergo the self-glucosylation, which is observed in wild-type GYG2. This is the first report of GYG2 mutation in human, implying a possible link between GYG2 abnormality and LS.





Similar content being viewed by others
References
Amaral AI (2012) Effects of hypoglycaemia on neuronal metabolism in the adult brain: role of alternative substrates to glucose. J Inherit Metab Dis. doi:10.1007/s10545-012-9553-3
Antonicka H, Ostergaard E, Sasarman F, Weraarpachai W, Wibrand F, Pedersen AM, Rodenburg RJ, van der Knaap MS, Smeitink JA, Chrzanowska-Lightowlers ZM, Shoubridge EA (2010) Mutations in C12orf65 in patients with encephalomyopathy and a mitochondrial translation defect. Am J Hum Genet 87:115–122. doi:10.1016/j.ajhg.2010.06.004
Barbetti F, Rocchi M, Bossolasco M, Cordera R, Sbraccia P, Finelli P, Consalez GG (1996) The human skeletal muscle glycogenin gene: cDNA, tissue expression and chromosomal localization. Biochem Biophys Res Commun 220:72–77. doi:10.1006/bbrc 1996.0359
Benke PJ, Parker JC Jr, Lubs ML, Benkendorf J, Feuer AE (1982) X-linked Leigh’s syndrome. Hum Genet 62:52–59
Bollen M, Keppens S, Stalmans W (1998) Specific features of glycogen metabolism in the liver. Biochem J 336:19–31
Brown GK, Squier MV (1996) Neuropathology and pathogenesis of mitochondrial diseases. J Inherit Metab Dis 19:553–572
Cao Y, Mahrenholz AM, DePaoli-Roach AA, Roach PJ (1993) Characterization of rabbit skeletal muscle glycogenin. Tyrosine 194 is essential for function. J Biol Chem 268:14687–14693
Chaikuad A, Froese DS, Berridge G, von Delft F, Oppermann U, Yue WW (2011) Conformational plasticity of glycogenin and its maltosaccharide substrate during glycogen biogenesis. Proc Natl Acad Sci USA 108:21028–21033. doi:10.1073/pnas.1113921108
Debray FG, Morin C, Janvier A, Villeneuve J, Maranda B, Laframboise R, Lacroix J, Decarie JC, Robitaille Y, Lambert M, Robinson BH, Mitchell GA (2011) LRPPRC mutations cause a phenotypically distinct form of Leigh syndrome with cytochrome c oxidase deficiency. J Med Genet 48:183–189. doi:10.1136/jmg.2010.081976
Farina L, Chiapparini L, Uziel G, Bugiani M, Zeviani M, Savoiardo M (2002) MR findings in Leigh syndrome with COX deficiency and SURF-1 mutations. AJNR Am J Neuroradiol 23:1095–1100
Finsterer J (2008) Leigh and Leigh-like syndrome in children and adults. Pediatr Neurol 39:223–235. doi:10.1016/j.pediatrneurol.2008.07.013
Garber AJ, Menzel PH, Boden G, Owen OE (1974) Hepatic ketogenesis and gluconeogenesis in humans. J Clin Invest 54:981–989. doi:10.1172/JCI107839
Gibbons BJ, Roach PJ, Hurley TD (2002) Crystal structure of the autocatalytic initiator of glycogen biosynthesis, glycogenin. J Mol Biol 319:463–477. doi:10.1016/S0022-2836(02)00305-4
Guerois R, Nielsen JE, Serrano L (2002) Predicting changes in the stability of proteins and protein complexes: a study of more than 1000 mutations. J Mol Biol 320:369–387. doi:10.1016/S0022-2836(02)00442-4
Khan S, Vihinen M (2010) Performance of protein stability predictors. Hum Mutat 31:675–684. doi:10.1002/humu.21242
Krisman CR, Barengo R (1975) A precursor of glycogen biosynthesis: alpha-1,4-glucan-protein. Eur J Biochem 52:117–123
Laffel L (1999) Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab Res Rev 15:412–426
Larner J (1953) The action of branching enzymes on outer chains of glycogen. J Biol Chem 202:491–503
Leigh D (1951) Subacute necrotizing encephalomyelopathy in an infant. J Neurol Neurosurg Psychiatry 14:216–221
Lomako J, Lomako WM, Whelan WJ (1988) A self-glucosylating protein is the primer for rabbit muscle glycogen biosynthesis. FASEB J 2:3097–3103
Lomako J, Lomako WM, Whelan WJ (2004) Glycogenin: the primer for mammalian and yeast glycogen synthesis. Biochim Biophys Acta 1673:45–55. doi:10.1016/j.bbagen.2004.03.017
Lopez LC, Schuelke M, Quinzii CM, Kanki T, Rodenburg RJ, Naini A, Dimauro S, Hirano M (2006) Leigh syndrome with nephropathy and CoQ10 deficiency due to decaprenyl diphosphate synthase subunit 2 (PDSS2) mutations. Am J Hum Genet 79:1125–1129. doi:10.1086/510023
Magistretti PJ, Pellerin L (1999) Cellular mechanisms of brain energy metabolism and their relevance to functional brain imaging. Philos Trans R Soc Lond B Biol Sci 354:1155–1163. doi:10.1098/rstb 1999.0471
Martin MA, Blazquez A, Gutierrez-Solana LG, Fernandez-Moreira D, Briones P, Andreu AL, Garesse R, Campos Y, Arenas J (2005) Leigh syndrome associated with mitochondrial complex I deficiency due to a novel mutation in the NDUFS1 gene. Arch Neurol 62:659–661. doi:10.1001/archneur.62.4.659
Medina L, Chi TL, DeVivo DC, Hilal SK (1990) MR findings in patients with subacute necrotizing encephalomyelopathy (Leigh syndrome): correlation with biochemical defect. AJR Am J Roentgenol 154:1269–1274
Moslemi AR, Lindberg C, Nilsson J, Tajsharghi H, Andersson B, Oldfors A (2010) Glycogenin-1 deficiency and inactivated priming of glycogen synthesis. N Engl J Med 362:1203–1210. doi:10.1056/NEJMoa0900661
Mu J, Roach PJ (1998) Characterization of human glycogenin-2, a self-glucosylating initiator of liver glycogen metabolism. J Biol Chem 273:34850–34856
Mu J, Skurat AV, Roach PJ (1997) Glycogenin-2, a novel self-glucosylating protein involved in liver glycogen biosynthesis. J Biol Chem 272:27589–27597
Naess K, Freyer C, Bruhn H, Wibom R, Malm G, Nennesmo I, von Dobeln U, Larsson NG (2009) MtDNA mutations are a common cause of severe disease phenotypes in children with Leigh syndrome. Biochim Biophys Acta 1787:484–490. doi:10.1016/j.bbabio.2008.11.014
Ostergaard E, Hansen FJ, Sorensen N, Duno M, Vissing J, Larsen PL, Faeroe O, Thorgrimsson S, Wibrand F, Christensen E, Schwartz M (2007) Mitochondrial encephalomyopathy with elevated methylmalonic acid is caused by SUCLA2 mutations. Brain 130:853–861. doi:10.1093/brain/awl383
Picardi E, Pesole G (2012) Mitochondrial genomes gleaned from human whole-exome sequencing. Nat Methods 9:523–524. doi:10.1038/nmeth.2029
Pitcher J, Smythe C, Cohen P (1988) Glycogenin is the priming glucosyltransferase required for the initiation of glycogen biogenesis in rabbit skeletal muscle. Eur J Biochem 176:391–395
Quinonez SC, Leber SM, Martin DM, Thoene JG, Bedoyan JK (2013) Leigh syndrome in a girl with a novel DLD mutation causing E3 deficiency. Pediatr Neurol 48:67–72. doi:10.1016/j.pediatrneurol.2012.09.013
Rahman S, Blok RB, Dahl HH, Danks DM, Kirby DM, Chow CW, Christodoulou J, Thorburn DR (1996) Leigh syndrome: clinical features and biochemical and DNA abnormalities. Ann Neurol 39:343–351. doi:10.1002/ana.410390311
Randle PJ, Newsholme EA, Garland PB (1964) Regulation of glucose uptake by muscle. 8. Effects of fatty acids, ketone bodies and pyruvate, and of alloxan-diabetes and starvation, on the uptake and metabolic fate of glucose in rat heart and diaphragm muscles. Biochem J 93:652–665
Smythe C, Caudwell FB, Ferguson M, Cohen P (1988) Isolation and structural analysis of a peptide containing the novel tyrosyl-glucose linkage in glycogenin. EMBO J 7:2681–2686
van Erven PM, Cillessen JP, Eekhoff EM, Gabreels FJ, Doesburg WH, Lemmens WA, Slooff JL, Renier WO, Ruitenbeek W (1987) Leigh syndrome, a mitochondrial encephalo(myo)pathy. A review of the literature. Clin Neurol Neurosurg 89:217–230
Viskupic E, Cao Y, Zhang W, Cheng C, DePaoli-Roach AA, Roach PJ (1992) Rabbit skeletal muscle glycogenin. Molecular cloning and production of fully functional protein in Escherichia coli. J Biol Chem 267:25759–25763
Zhai L, Schroeder J, Skurat AV, Roach PJ (2001) Do rodents have a gene encoding glycogenin-2, the liver isoform of the self-glucosylating initiator of glycogen synthesis? IUBMB Life 51:87–91. doi:10.1080/15216540117315
Acknowledgments
We thank all the patients and their families for participating in this work. We deeply appreciate Dr. Toshiyuki Fukao, who is the professor of Department of Pediatrics, Graduate School of Medicine, Gifu University, for the enzyme assay for 3-ketothiolase deficiency and succinyl-CoA:3-oxoacid CoA transferase. We would like to thank Dr. Yasushi Okazaki at Research Center for Genomic Medicine, Saitama Medical University for helpful discussion. We also thank Ms. Y. Yamashita, S. Sugimoto and K. Takabe for their technical assistance. This work was supported by research grants from the Ministry of Health, Labour, and Welfare (H. Saitsu, N. Matsumoto and N. Miyake), the Japan Science and Technology Agency (N. Matsumoto), the Strategic Research Program for Brain Sciences (to N. Matsumoto), a Grant-in-Aid for Scientific Research on Innovative Areas (Transcription Cycle) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (N. Matsumoto), a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (to N. Matsumoto), a Grant-in-Aid for Young Scientists from the Japan Society for the Promotion of Science (H. Saitsu and N. Miyake), a grant from the 2012 Strategic Research Promotion of Yokohama City University (N. Matsumoto), and research grants from the Japan Epilepsy Research Foundation (H. Saitsu) and the Takeda Science Foundation (N. Matsumoto and N. Miyake).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Imagawa, E., Osaka, H., Yamashita, A. et al. A hemizygous GYG2 mutation and Leigh syndrome: a possible link?. Hum Genet 133, 225–234 (2014). https://doi.org/10.1007/s00439-013-1372-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-013-1372-6