Abstract
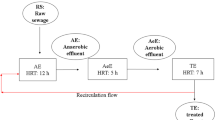
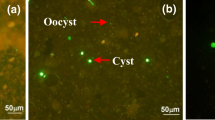
This study investigates the fate of Cryptosporidium parvum and C. hominis oocysts and Giardia duodenalis cysts at four Irish municipal wastewater treatment plants (i.e., Plant A, B, C, and D) that utilize sludge activation or biofilm-coated percolating filter systems for secondary wastewater treatment. The fate of these pathogens through the sewage treatment processes was determined based on their viable transmissive stages, i.e., oocysts for Cryptosporidium and cysts for Giardia. Analysis of final effluent indicated that over 97% of viable oocysts and cysts were eliminated, except at Plant C, which achieved only 64% of oocyst removal. A significant correlation between the removal of oocysts and cysts was found at Plants A, B, and D (R = 0.98, P < 0.05). All sewage sludge samples were positive for C. parvum and C. hominis, and G. duodenalis, with maximum concentrations of 20 oocysts and eight cysts per gram in primary sludge indicating the need for further sludge sanitization treatments. This study provides evidence that C. parvum and C. hominis oocysts and G. duodenalis cysts are present throughout the wastewater processes and in end-products, and can enter the aquatic environment with consequent negative implications for public health.


Similar content being viewed by others
References
Ahmad RA, Lee E, Tan ITL, Mohamad-Kamel AG (1997) Occurrence of Giardia cysts and Cryptosporidium oocysts in raw and treated water from two water treatment plants in Selangor, Malaysia. Water Res 31:3132–3136
Al-Saed R (2007) Pathogens assessment in reclaimed effluent used for industrial crops irrigation. Int J Environ Res Pub Hlth 4:68–75
Brianesco R, Bonadonna L (2005) An Italian study on Cryptosporidium and Giardia in wastewater, fresh water and treated water. Environ Monit Assess 104:445–457
Caccio SM, De Giacomo M, Aulicino FA, Pozio E (2003) Giardia cysts in wastewater treatment plants in Italy. Appl Envion Microbiol 69:3393–3398
Chalmers RM, Sturdee AP, Mellors P, Nicholson V, Lawlor F, Kenny F, Timpson P (1997) Cryptosporidium parvum in environmental samples in the Sligo area, Republic of Ireland: a preliminary report. Lett Appl Microbiol 25:380–384
Charles K, Ashbolt N, Ferguson C, Roser D, McGuinness R, Deere D (2003) Centralised versus decentralized sewage systems: a comparison of pathogen and nutrient loads released into Sydney’s drinking water catchments. Water Sci Technol 48:53–60
Cole D, Todd L, Wing S (2000) Concentrated swine feeding operations and public health: a review of occupational and community health effects. Environ Health Perspect 108:685–699
Council of the European Community (CEC) (1986) The protection of the environment and in particular of the soil, when sewage sludge is used in agriculture. Directive 86/278/EEC
Council of the European Community (CEC) (1991) Urban wastewater treatment directive. Directive 91/271/EEC
Deere D, Vesey G, Ashbolt N, Davies KA, Williams KL, Veal D (1989) Evaluation of fluorochromes for flow cytometric detection of Cryptosporidium parvum oocysts labelled by fluorescent in situ hybridization. Lett Appl Microbiol 27:352–356
Dorn RC, Reddy CS, Lamphere DN, Gaeuman JV, Lanese R (1985) Municipal sewage sludge application on Ohio farms: health effects. Environ Res 38:332–359
Dorsch MR, Veal DA (2001) Oligonucleotide probes for specific detection of Giardia lamblia cysts by fluorescent in situ hybridization. J Appl Microbiol 90:836–842
DuPont HL, Chappell L, Sterlig L, Okhuysen C, Rose C, Jakubowski W (1995) The infectivity of C. parvum in healthy volunteers. N Eng J Med 332:855–859
Gale P (2005) Land application of treated sewage sludge: quantifying pathogen risk from consumption of crops. J Appl Microbiol 98:380–396
Graczyk TK, Conn DB, Lucy F, Minchin D, Tamang L, Moura LNS, DaSilva AJ (2004) Human waterborne parasites in zebra mussels (Dreissena polymorpha) from the Shannon River drainage area, Ireland. Parasitol Res 93:385–391
Graczyk TK, Lucy FE, Tamang L, Miraflor A (2007) Human enteropathogen load in activated sewage sludge and corresponding sewage sludge-end products. Appl Environ Microbiol 73:2013–2015
Graczyk TK, Majewska AC, Schwab KJ (2008a) The role of aquatic birds in dissemination of human waterborne enteropathogens. Trends Parasitol 24:55–59
Graczyk TK, Kacprzak M, Neczaj E, Tamang L, Graczyk H, Lucy FE, Girouard AS (2008b) Occurrence of Cryptosporidium and Giardia in sewage sludge and solid waste landfill leachate and quantitative comparative analysis of sanitization treatments on pathogen inactivation. Environ Res 106:27–33
Gray NF (2004) Biology of wastewater treatment. Imperial College Press, London, UK
Hutchison ML, Walters LD, Moore A, Avery SM (2005) Decline of zoonotic agents in liquid livestock wastes stored in batches on-farm. J Appl Mcirobiol 99:58–65
Ingallinella AM, Sanguinelli G, Koottatep T, Montangero A, Strauss M (2002) The challenge of faecal sludge management in urban areas- strategies and treatment options. Water Sci Technol 46:285–294
Irish Environmental Protection Agency (IEPA) (2000) The hardness of Irish drinking waters: a simple guide based on conductivity data. Environmental Protection Agency, Wexford, Ireland
Irish Environmental Protection Agency (IEPA) (2007) Urban waste water discharges in Ireland. A report for the years of 2004 and 2005. Environmental Protection Agency, Wexford, Ireland
Lemos V, Graczyk TK, Aves M, Lobo ML, Neto CS, Sousa MC, Antunes F, Matos O (2005) Identification and determination of the viability of Giardia lamblia cysts. Cryptosporidium parvum and C. hominis oocysts in human fecal samples and water supply samples by fluorescent in situ hybridization and monoclonal antibodies. Parasitol Res 98:48–53
Lonigro A, Pollice A, Spinelli R, Berrilli F, DiCave D, D’Orazi C, Cavallo P, Brandonisio O (2006) Giardia cysts and Cryptosporidium oocysts in membrane-filtered municipal wastewater used for irrigation. Appl Environ Microbiol 72:7916–7918
Lowery CJ, Moore JE, Millar BC, McCorry KAJ, Xu J, Rooney PJ, Dooley JSG (2001) Occurrence and molecular genotyping of Cryptosporidium spp. in surface waters in Northern Ireland. J Appl Microbiol 91:774–779
Lucy FE, Graczyk TK, Tamang L, Miraflor A, Minchin D (2008) Biomonitoring of surface and coastal water for Cryptosporidium, Giardia and human-virulent microsporidia using molluscan shellfish. Parasitol Res 103:1369–1375
MacKenzie WR, Hoxie NJ, Proctor ME, Gradus MS, Blair KA, Peterson DE, Kazmierczak JJ, Addiss DG, Fox KR, Rose JB, Davis JP (1994) A massive outbreak in Milwaukee of Cryptosporidium infection transmitted through the public water supply. New Engl J Med 331:161–167
Montemayor M, Valero F, Jofre J, Lucena F (2005) Occurrence of Cryptosporidium spp. oocysts in raw and treated sewage and river water in north-eastern Spain. J Appl Microbiol 99:1455–1462
Pelly H, Cormican M, O’Donovan D, Chalmers RH, Hanahoe B, Cloughley R, McKeowan P, Corbett-Feeney G (2007) A large outbreak of cryptosporidiosis in western Ireland linked to public water supply: a preliminary report. Euro Surveill 12:3
Pollice A, Lopez A, Laera G, Rubino G, Lonigro A (2004) Tertiary filtered municipal wastewater as alternative water source in agriculture: a field investigation in southern Italy. Sci Total Environ 324:201–210
Rimhanen-Finne R, Vuorinen A, Marmo S, Malmberg S, Hanninen ML (2004) Comparative analysis of Cryptosporidium, Giardia and indicator bacteria during sewage sludge hygienization in various composting processes. Lett Appl Microbiol 38:301–305
Robertson LJ, Paton CA, Campbell AT, Smith PG, Jackson MH, Gilmour RA, Black SE, Stevenson DA, Smith HV (2000) Giardia cysts and Cryptosporidium oocysts at sewage treatment works in Scotland, UK. Water Res 34:2310–2322
Robertson LJ, Hermansen L, Gjerde BK (2006) Occurrence of Cryptosporidium oocysts and Giardia cysts in sewage in Norway. Appl Environ Microbiol 72:5297–5303
Skerrett HE, Holland CV (2000) The occurrence of Cryptosporidium in environmental waters in the greater Dublin area. Water Res 34:3755–3760
Smith HV, Grimason AM (2003) Giardia and Cryptosporidium in water and wastewater. In: Mara D, Horan N (eds) The handbook of water and wastewater microbiology. Elsevier, Oxford, UK, pp 619–781
Smith JJ, Gunasekera TS, Barardi CRM, Veal D, Vesey G (2004) Detection of Cryptosporidium parvum oocyst viability by fluorescence in situ hybridization using a ribosomal RNA- directed probe. J Appl Microbiol 96:409–417
Suwa M, Suzuki Y (2001) Occurrence of Cryptosporidium in Japan and counter-measures in wastewater treatment plants. Water Sci Technol 43:183–186
Tamburini A, Pozio E (1999) Long-term survival of Cryptosporidium parvum oocysts in seawater and in experimentally infected mussels (Mytilus galloprovincialis). Int J Parasitol 29:711–715
Veronica A (2008) Proper sanitization of sewage sludge: a critical issue for a sustainable society. Appl Environ Microbiol 74:5276–5275
Wolfe MS (1992) Giardiasis. Clin Microbiol Rev 5:93–100
Zintl A, Mulcahy G, deWaal T, deWaele V, Byrne C, Clyne M, Holden N, Fanning S (2006) An Irish perspective on Cryptosporidium. Irish Vet J 59:442–447
Acknowledgments
The study was supported by the Irish EPA Strive PhD grant 2007-PhD-EH-3, Fulbright Senior Specialist Fellowship (grant no. 2225 Graczyk), Johns Hopkins Center in Urban Environmental Health (grant no. P30 ES03819), School of Science Institute of Technology, Sligo, Ireland, and the U.S. Environmental Protection Agency Science to Achieve Results (STAR) Program (grant no. RD83300201). The views expressed herein have not been subjected to the U.S. EPA review and therefore do not necessarily reflect the views of the agency, and no official endorsement should be inferred.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, HW.A., Lucy, F.E., Graczyk, T.K. et al. Fate of Cryptosporidium parvum and Cryptosporidium hominis oocysts and Giardia duodenalis cysts during secondary wastewater treatments. Parasitol Res 105, 689–696 (2009). https://doi.org/10.1007/s00436-009-1440-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-009-1440-y