Abstract
Purpose
Rabbit antithymocyte globulin (ATG) is commonly used before allogeneic hematopoietic stem cell transplantation (allo-HSCT) to prevent graft-versus-host disease. Studies comparing the effect of different ATG preparations and dosages on immune reconstitution and risk for Epstein–Barr virus (EBV)-mediated post-transplant lymphoproliferative disorder (PTLD) are rare.
Methods
In this retrospective study, we determined T and B cell subsets by flow cytometry after allo-HSCT in children, who received ATG-Genzyme (ATG-G, n = 15), ATG-Fresenius (ATG-F, n = 25) or no-ATG treatment (n = 19). Additionally, PCR-quantified EBV-genome copy counts were correlated with incidence of PTLD.
Results
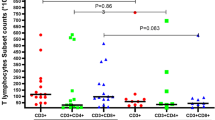
We could confirm a dose-dependent impairment of CD8+ and CD4+ T cell regeneration by ATG-G, including naïve and memory CD4+ T cells. No differences were seen between the currently applied dosages of 5–10 mg/kg ATG-G and 20–60 mg/kg ATG-F. Significantly delayed T cell subset reconstitution was determined only at high dosages of 20–60 mg/kg ATG-G compared to ATG-F. B cell reconstitution was comparably impaired in ATG-G- and ATG-F-treated patients. Although the incidence of EBV reactivation was similar in both ATG groups, EBV copy counts of >104 copies/105 peripheral blood mononuclear cells and the occurrence of PTLD were only found in ATG-G-treated patients.
Conclusions
We conclude that high, but importantly not currently applied low dosages of ATG-G, impair thymic T cell regeneration and memory T cell immunity to a greater extent than ATG-F in pediatric patients. In addition, our results suggest an increased risk for EBV-PTLD when treated with ATG-G. Prospective studies are warranted to compare different ATG preparations with regard to the immune reconstitution and EBV-PTLD.




Similar content being viewed by others
References
Bacigalupo A (2005a) Antilymphocyte/thymocyte globulin for graft versus host disease prophylaxis: efficacy and side effects. Bone Marrow Transplant 35(3):225–231
Bacigalupo A (2005b) Antithymocyte globulin for prevention of graft-versus-host disease. Curr Opin Hematol 12(6):457–462
Baker MB, Riley RL, Podack ER, Levy RB (1997) Graft-versus-host-disease-associated lymphoid hypoplasia and B cell dysfunction is dependent upon donor T cell-mediated Fas-ligand function, but not perforin function. Proc Natl Acad Sci USA 94(4):1366–1371
Basara N, Baurmann H, Kolbe K, Yaman A, Labopin M, Burchardt A et al (2005) Antithymocyte globulin for the prevention of graft-versus-host disease after unrelated hematopoietic stem cell transplantation for acute myeloid leukemia: results from the multicenter German cooperative study group. Bone Marrow Transplant 35(10):1011–1018
Bollard CM, Rooney CM, Heslop HE (2012) T-cell therapy in the treatment of post-transplant lymphoproliferative disease. Nat Rev Clin Oncol 9(9):510–519
Bomberger C, Singh-Jairam M, Rodey G, Guerriero A, Yeager AM, Fleming WH et al (1998) Lymphoid reconstitution after autologous PBSC transplantation with FACS-sorted CD34+ hematopoietic progenitors. Blood 91(7):2588–2600
Brunstein CG, Weisdorf DJ, DeFor T, Barker JN, Tolar J, van Burik JA et al (2006) Marked increased risk of Epstein–Barr virus-related complications with the addition of antithymocyte globulin to a nonmyeloablative conditioning prior to unrelated umbilical cord blood transplantation. Blood 108(8):2874–2880
Clave E, Busson M, Douay C, Peffault de Latour R, Berrou J, Rabian C et al (2009) Acute graft-versus-host disease transiently impairs thymic output in young patients after allogeneic hematopoietic stem cell transplantation. Blood 113(25):6477–6484
Copelan EA (2006) Hematopoietic stem-cell transplantation. N Engl J Med 354(17):1813–1826
Fehse N, Fehse B, Kroger N, Zabelina T, Freiberger P, Kruger W et al (2003) Influence of anti-thymocyte globulin as part of the conditioning regimen on immune reconstitution following matched related bone marrow transplantation. J Hematother Stem Cell Res 12(2):237–242
Gruhn B, Meerbach A, Hafer R, Zell R, Wutzler P, Zintl F (2003) Pre-emptive therapy with rituximab for prevention of Epstein–Barr virus-associated lymphoproliferative disease after hematopoietic stem cell transplantation. Bone Marrow Transplant 31(11):1023–1025
Heitger A, Neu N, Kern H, Panzer-Grumayer ER, Greinix H, Nachbaur D et al (1997) Essential role of the thymus to reconstitute naive (CD45RA+) T-helper cells after human allogeneic bone marrow transplantation. Blood 90(2):850–857
Heller KN, Upshaw J, Seyoum B, Zebroski H, Munz C (2007) Distinct memory CD4+ T-cell subsets mediate immune recognition of Epstein Barr virus nuclear antigen 1 in healthy virus carriers. Blood 109(3):1138–1146
Khanna R, Burrows SR (2000) Role of cytotoxic T lymphocytes in Epstein–Barr virus-associated diseases. Annu Rev Microbiol 54:19–48
Kumar A, Mhaskar AR, Reljic T, Mhaskar RS, Kharfan-Dabaja MA, Anasetti C et al (2012) Antithymocyte globulin for acute-graft-versus-host-disease prophylaxis in patients undergoing allogeneic hematopoietic cell transplantation: a systematic review. Leukemia 26(4):582–588
Landais E, Saulquin X, Houssaint E (2005) The human T cell immune response to Epstein–Barr virus. Int J Dev Biol 49(2–3):285–292
Landgren O, Gilbert ES, Rizzo JD, Socie G, Banks PM, Sobocinski KA et al (2009) Risk factors for lymphoproliferative disorders after allogeneic hematopoietic cell transplantation. Blood 113(20):4992–5001
Mautner J, Bornkamm GW (2012) The role of virus-specific CD4+ T cells in the control of Epstein–Barr virus infection. Eur J Cell Biol 91(1):31–35
Meerbach A, Gruhn B, Egerer R, Reischl U, Zintl F, Wutzler P (2001) Semiquantitative PCR analysis of Epstein–Barr virus DNA in clinical samples of patients with EBV-associated diseases. J Med Virol 65(2):348–357
Meerbach A, Wutzler P, Hafer R, Zintl F, Gruhn B (2008) Monitoring of Epstein–Barr virus load after hematopoietic stem cell transplantation for early intervention in post-transplant lymphoproliferative disease. J Med Virol 80(3):441–454
Meijer E, Bloem AC, Dekker AW, Verdonck LF (2003) Effect of antithymocyte globulin on quantitative immune recovery and graft-versus-host disease after partially T-cell-depleted bone marrow transplantation: a comparison between recipients of matched related and matched unrelated donor grafts. Transplantation 75(11):1910–1913
Mohty M (2007) Mechanisms of action of antithymocyte globulin: T-cell depletion and beyond. Leukemia 21(7):1387–1394
Na IK, Lu SX, Yim NL, Goldberg GL, Tsai J, Rao U et al (2010) The cytolytic molecules Fas ligand and TRAIL are required for murine thymic graft-versus-host disease. J Clin Invest 120(1):343–356
Na IK, Wittenbecher F, Dziubianau M, Herholz A, Mensen A, Kunkel D et al (2013) Rabbit antithymocyte globulin (Thymoglobulin(R)) impairs the thymic output of both conventional and regulatory CD4+ T cells after allogeneic hematopoietic stem cell transplantation in adult patients. Haematologica 98(1):23–30
Nakai K, Mineishi S, Kami M, Saito T, Hori A, Kojima R et al (2003) Antithymocyte globulin affects the occurrence of acute and chronic graft-versus-host disease after a reduced-intensity conditioning regimen by modulating mixed chimerism induction and immune reconstitution. Transplantation 75(12):2135–2143
Preville X, Flacher M, LeMauff B, Beauchard S, Davelu P, Tiollier J et al (2001) Mechanisms involved in antithymocyte globulin immunosuppressive activity in a nonhuman primate model. Transplantation 71(3):460–468
Scheinberg P, Melenhorst JJ, Brenchley JM, Hill BJ, Hensel NF, Chattopadhyay PK et al (2009) The transfer of adaptive immunity to CMV during hematopoietic stem cell transplantation is dependent on the specificity and phenotype of CMV-specific T cells in the donor. Blood 114(24):5071–5080
Schleuning M, Gunther W, Tischer J, Ledderose G, Kolb HJ (2003) Dose-dependent effects of in vivo antithymocyte globulin during conditioning for allogeneic bone marrow transplantation from unrelated donors in patients with chronic phase CML. Bone Marrow Transplant 32(3):243–250
Storek J, Wells D, Dawson MA, Storer B, Maloney DG (2001) Factors influencing B lymphopoiesis after allogeneic hematopoietic cell transplantation. Blood 98(2):489–491
Storek J, Geddes M, Khan F, Huard B, Helg C, Chalandon Y et al (2008) Reconstitution of the immune system after hematopoietic stem cell transplantation in humans. Semin Immunopathol 30(4):425–437
Terasako K, Sato K, Sato M, Kimura S, Nakasone H, Okuda S et al (2010) The effect of different ATG preparations on immune recovery after allogeneic hematopoietic stem cell transplantation for severe aplastic anemia. Hematology 15(3):165–169
van Esser JW, van der Holt B, Meijer E, Niesters HG, Trenschel R, Thijsen SF et al (2001) Epstein–Barr virus (EBV) reactivation is a frequent event after allogeneic stem cell transplantation (SCT) and quantitatively predicts EBV-lymphoproliferative disease following T-cell–depleted SCT. Blood 98(4):972–978
Welniak LA, Blazar BR, Murphy WJ (2007) Immunobiology of allogeneic hematopoietic stem cell transplantation. Annu Rev Immunol 25:139–170
Acknowledgments
The technical assistance of Doris Glaser and Sabine Becker is acknowledged. The nurses and physicians of the Stem Cell Transplant Unit are thanked for their care of patients and assistance in obtaining patient samples.
Conflict of interest
We declare that we have no conflict of interest.
Ethical standard
The study was approved by the institutional review board and patients and parents gave informed consent.
Author information
Authors and Affiliations
Corresponding author
Additional information
Angela Mensen, Il-Kang Na have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mensen, A., Na, IK., Häfer, R. et al. Comparison of different rabbit ATG preparation effects on early lymphocyte subset recovery after allogeneic HSCT and its association with EBV-mediated PTLD. J Cancer Res Clin Oncol 140, 1971–1980 (2014). https://doi.org/10.1007/s00432-014-1742-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1742-z