Abstract
Purpose
Carbohydrate ingestion prior and during exercise attenuates exercise-induced interleukin-6. This investigation examined if an analogous effect was evident for interleukin-6 and hepcidin response when carbohydrates were ingested post-exercise.
Methods
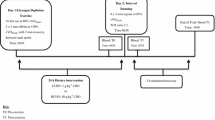
In a crossover design, 11 well-trained endurance athletes completed two experimental trials. Participants completed an 8 × 3 min interval running session at 85 % vVO2peak followed by 5 h of monitored recovery. During this period, participants were provided with two 1.2 g kg−1 carbohydrate beverages at either an early feeding time (immediately post-exercise and 2 h post-exercise) or delayed feeding time (2 h post-exercise and 4 h post-exercise). Venous blood samples were collected pre-, immediately post-, 3 and 5 h post-exercise. Samples were analysed for Interleukin-6, serum iron, serum ferritin and hepcidin.
Results
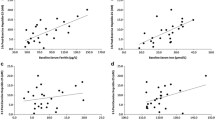
Interleukin-6 was significantly elevated (p = 0.004) immediately post-exercise compared to baseline for both trials. Hepcidin levels were significantly elevated at 3 h post-exercise (p = 0.001) and 5 h post-exercise (p = 0.002) compared to baseline levels in both trials, with no significant difference between the two conditions and any time point. Serum iron was significantly increased from baseline to immediately post-exercise (p = 0.001) for both trials, with levels decreasing by 3 h (p = 0.025) and 5 h post-exercise (p = 0.001). Serum ferritin levels increased immediately post-exercise compared to baseline (p = 0.006) in both conditions.
Conclusions
The timing and ingestion of post-exercise carbohydrate ingestion do not appear to impact post-exercise interleukin-6 and hepcidin responses; this is likely a result of the interval running task inducing an inflammatory response and subsequent up-regulation of hepcidin.
Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- BLa:
-
Blood lactate
- BM:
-
Body mass
- CHO:
-
Carbohydrate
- CO2 :
-
Carbon dioxide
- CV:
-
Coefficient of variance
- DCHO:
-
Delayed carbohydrate feeding trial
- ECHO:
-
Early carbohydrate feeding trial
- Fpn:
-
Ferroportin
- GXT:
-
Graded exercise test
- Hb:
-
Haemoglobin
- Hct:
-
Haematocrit
- HR:
-
Heart rate
- Hp:
-
Haptoglobin
- IL-6:
-
Interleukin-6
- O2 :
-
Oxygen
- RE:
-
Reticuloendothelial system
- RPE:
-
Rating of perceived exertion
- SD:
-
Standard deviation
- TLCH:
-
Train low, compete high
- VO2peak :
-
Peak oxygen uptake
- vVO2peak :
-
Velocity at peak oxygen uptake
References
Areta JL, Burke LM, Ross ML et al (2013) Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J Physiol 591:2319–2331. doi:10.1113/jphysiol.2012.244897
Badenhorst CE, Dawson B, Goodman C et al (2014) Influence of post-exercise hypoxic exposure on hepcidin response in athletes. Eur J Appl Physiol 114:951–959. doi:10.1007/s00421-014-2829-6
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14:377–381
Buchman AL, Keen C, Commisso J et al (1998) The effect of a marathon run on plasma and urine mineral and metal concentrations. J Am Coll Nutr 17:124–127
Burke LM (2010) Fueling strategies to optimize performance: training high or training low? Scand J Med Sci Sports 20(Suppl 2):48–58. doi:10.1111/j.1600-0838.2010.01185.x
Burke LM, Hawley JA, Wong SHS, Jeukendrup AE (2011) Carbohydrates for training and competition. J Sports Sci 29(Suppl 1):S17–S27. doi:10.1080/02640414.2011.585473
Cartee GD, Young DA, Sleeper MD et al (1989) Prolonged increase in insulin-stimulated glucose transport in muscle after exercise. Am J Physiol 256:E494–E499
Convertino VA, Armstrong LE, Coyle EF et al (1996) American college of sports medicine position stand. exercise and fluid replacement. Med Sci Sports Exerc 28:i–vii
Dill DB, Costill DL (1974) Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol 37:247–248
Fallon KE (2001) The acute phase response and exercise: the ultramarathon as prototype exercise. Clin J Sport Med 11:38–43
Febbraio MA, Steensberg A, Keller C et al (2003) Glucose ingestion attenuates interleukin-6 release from contracting skeletal muscle in humans. J Physiol 549:607–612. doi:10.1113/jphysiol.2003.042374
Fischer CP (2006) Interleukin-6 in acute exercise and training: what is the biological relevance? Exerc Immunol Rev 12:6–33
Goodyear LJ, Hirshman MF, King PA et al (1990) Skeletal muscle plasma membrane glucose transport and glucose transporters after exercise. J Appl Physiol 68:193–198
Helge JW, Stallknecht B, Pedersen BK et al (2003) The effect of graded exercise on IL-6 release and glucose uptake in human skeletal muscle. J Physiol 546:299–305
Hopkins W (2005) A spreadsheet for fully controlled crossovers. Sport Sci 9:3
Ivy JL, Katz AL, Cutler CL et al (1988) Muscle glycogen synthesis after exercise: effect of time of carbohydrate ingestion. J Appl Physiol 64:1480–1485
Jones AM, Doust JH (1996) A 1 % treadmill grade most accurately reflects the energetic cost of outdoor running. J Sports Sci 14:321–327. doi:10.1080/02640419608727717
Keller C, Steensberg A, Pilegaard H et al (2001) Transcriptional activation of the IL-6 gene in human contracting skeletal muscle: influence of muscle glycogen content. FASEB J 15:2748–2750. doi:10.1096/fj.01-0507fje
Kemna E, Pickkers P, Nemeth E et al (2005) Time-course analysis of hepcidin, serum iron, and plasma cytokine levels in humans injected with LPS. Blood 106:1864–1866. doi:10.1182/blood-2005-03-1159
Kroot JJC, Laarakkers CMM, Geurts-Moespot AJ et al (2010) Immunochemical and mass-spectrometry-based serum hepcidin assays for iron metabolism disorders. Clin Chem 56:1570–1579. doi:10.1373/clinchem.2010.149187
Kroot JJC, Tjalsma H, Fleming RE, Swinkels DW (2011) Hepcidin in human iron disorders: diagnostic implications. Clin Chem 57:1650–1669. doi:10.1373/clinchem.2009.140053
Laarakkers CMM, Wiegerinck ET, Klaver S et al (2013) Improved mass spectrometry assay for plasma hepcidin: detection and characterization of a novel hepcidin isoform. PLoS One 8:e75518. doi:10.1371/journal.pone.0075518
Latunde-Dada GO (2013) Iron metabolism in athletes—achieving a gold standard. Eur J Haematol 90:10–15. doi:10.1111/ejh.12026
Lymboussaki A, Pignatti E, Montosi G et al (2003) The role of the iron responsive element in the control of ferroportin1/IREG1/MTP1 gene expression. J Hepatol 39:710–715
Nemeth E, Rivera S, Gabayan V et al (2004a) IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J Clin Invest 113:1271–1276. doi:10.1172/JCI20945
Nemeth E, Tuttle MS, Powelson J et al (2004b) Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306:2090–2093. doi:10.1126/science.1104742
Nieman DC, Nehlsen-Cannarella SL, Fagoaga OR et al (1998) Influence of mode and carbohydrate on the cytokine response to heavy exertion. Med Sci Sports Exerc 30:671–678
Nieman DC, Davis JM, Henson DA et al (2003) Carbohydrate ingestion influences skeletal muscle cytokine mRNA and plasma cytokine levels after a 3-h run. J Appl Physiol 94:1917–1925. doi:10.1152/japplphysiol.01130.2002
Pattini A, Schena F, Guidi GC (1990) Serum ferritin and serum iron changes after cross-country and roller ski endurance races. Eur J Appl Physiol Occup Physiol 61:55–60
Peeling P, Dawson B, Goodman C et al (2008) Athletic induced iron deficiency: new insights into the role of inflammation, cytokines and hormones. Eur J Appl Physiol 103:381–391. doi:10.1007/s00421-008-0726-6
Peeling P, Dawson B, Goodman C et al (2009a) Effects of exercise on hepcidin response and iron metabolism during recovery. Int J Sport Nutr Exerc Metab 19:583–597
Peeling P, Dawson B, Goodman C et al (2009b) Cumulative effects of consecutive running sessions on hemolysis, inflammation and hepcidin activity. Eur J Appl Physiol 106:51–59. doi:10.1007/s00421-009-0988-7
Peeling P, Dawson B, Goodman C et al (2009c) Training surface and intensity: inflammation, hemolysis, and hepcidin expression. Med Sci Sports Exerc 41:1138–1145. doi:10.1249/MSS.0b013e318192ce58
Peeling P, Sim M, Badenhorst CE et al (2014) Iron status and the acute post-exercise hepcidin response in athletes. PLoS One 9:e93002. doi:10.1371/journal.pone.0093002
Robson-Ansley PJ, Blannin A, Gleeson M (2007) Elevated plasma interleukin-6 levels in trained male triathletes following an acute period of intense interval training. Eur J Appl Physiol 99:353–360. doi:10.1007/s00421-006-0354-y
Robson-Ansley P, Walshe I, Ward D (2011) The effect of carbohydrate ingestion on plasma interleukin-6, hepcidin and iron concentrations following prolonged exercise. Cytokine 53:196–200. doi:10.1016/j.cyto.2010.10.001
Roecker L, Meier-Buttermilch R, Brechtel L et al (2005) Iron-regulatory protein hepcidin is increased in female athletes after a marathon. Eur J Appl Physiol 95:569–571. doi:10.1007/s00421-005-0055-y
Ronsen O, Lea T, Bahr R, Pedersen BK (2002) Enhanced plasma IL-6 and IL-1ra responses to repeated vs. single bouts of prolonged cycling in elite athletes. J Appl Physiol 92:2547–2553. doi:10.1152/japplphysiol.01263.2001
Sim M, Dawson B, Landers G et al (2012) The effects of carbohydrate ingestion during endurance running on post-exercise inflammation and hepcidin levels. Eur J Appl Physiol 112:1889–1898. doi:10.1007/s00421-011-2156-0
Sim M, Dawson B, Landers GJ et al (2014) A seven day running training period increases basal urinary hepcidin levels as compared to cycling. J Int Soc Sports Nutr 11:14. doi:10.1186/1550-2783-11-14
Steensberg A, van Hall G, Osada T et al (2000) Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J Physiol 529(Pt 1):237–242
Steensberg A, Febbraio MA, Osada T et al (2001) Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol 537:633–639
Weight LM, Byrne MJ, Jacobs P (1991) Haemolytic effects of exercise. Clin Sci (Lond) 81:147–152
Yeo WK, Paton CD, Garnham AP et al (2008) Skeletal muscle adaptation and performance responses to once a day versus twice every second day endurance training regimens. J Appl Physiol 105:1462–1470. doi:10.1152/japplphysiol.90882.2008
Acknowledgments
The authors wish to acknowledge the participants who volunteered their time for this study.
Conflict of interest
The authors report no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Fabio Fischetti.
Rights and permissions
About this article
Cite this article
Badenhorst, C.E., Dawson, B., Cox, G.R. et al. Timing of post-exercise carbohydrate ingestion: influence on IL-6 and hepcidin responses. Eur J Appl Physiol 115, 2215–2222 (2015). https://doi.org/10.1007/s00421-015-3202-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-015-3202-0